6636-78-8 Usage
Description
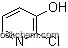
2-Chloro-3-hydroxypyridine is an organic compound that features a chloro substituent at the 2nd position and a hydroxyl group at the 3rd position on a pyridine ring. It is a versatile intermediate in organic synthesis, particularly for the preparation of various heterocyclic compounds and pharmaceuticals.
Uses
Used in Pharmaceutical and Organic Synthesis:
2-Chloro-3-hydroxypyridine is used as a key intermediate in the synthesis of various heterocyclic compounds and pharmaceuticals. Its unique structure allows for further functionalization and modification, making it a valuable building block in the development of new drugs and organic compounds.
Used in the Preparation of 3-Hydroxypyridine-2(1H)-Selenone:
2-Chloro-3-hydroxypyridine is used as a starting material in the preparation of 3-hydroxypyridine-2(1H)-selenone, a compound with potential applications in various fields, such as pharmaceuticals and materials science.
Used in the Synthesis of Benzo[4,5]furopyridine Tricyclic Heterocycles:
2-Chloro-3-hydroxypyridine is used as a precursor in the synthesis of all four possible benzo[4,5]furopyridine tricyclic heterocycles, including benzo[4,5]furo[2,3-b]pyridine, benzo[4,5]furo[2,3-c]pyridine, benzo[4,5]furo[3,2-c]pyridine, and benzo[4,5]furo[3,2-b]pyridine. The synthesis process involves α and γ-activation of chloropyridines, as well as palladium-mediated reactions, which are crucial for the formation of these complex heterocyclic structures. These tricyclic heterocycles have potential applications in various fields, such as medicinal chemistry and drug discovery.
Check Digit Verification of cas no
The CAS Registry Mumber 6636-78-8 includes 7 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 4 digits, 6,6,3 and 6 respectively; the second part has 2 digits, 7 and 8 respectively.
Calculate Digit Verification of CAS Registry Number 6636-78:
(6*6)+(5*6)+(4*3)+(3*6)+(2*7)+(1*8)=118
118 % 10 = 8
So 6636-78-8 is a valid CAS Registry Number.
InChI:InChI=1/C5H4ClNO/c6-5-4(8)2-1-3-7-5/h1-3,8H
6636-78-8Relevant articles and documents
Preparation method of 2,3-dichloropyridine
-
Paragraph 0052-0053, (2018/06/15)
The invention provides a preparation method of 2,3-dichloropyridine. The 2,3-dichloropyridine is prepared by a one-pot method, pyridine alkyl ester serves as a raw material, and synthesis is conductedby a mode of performing chlorination, performing hydrolysis and performing chlorination again; the chlorination is substitution reaction, substitution on a benzene ring is relatively easy, the alkylester group can protect the site of the first-time chlorination reaction to be at 2 position, the hydrolysis reaction is mild in condition and rapid in reaction compared with the prior art, the second-time chlorination is to substitute the hydroxyl, the reaction is easy and the reaction condition is relatively mild. The preparation method provided by the invention is simple in operation, few threewastes are generated in the production process, requirements on equipment are low and the yield is high.
Cesium carbonate mediated aryl triflate esters' deprotection
Green, Alice E.,Agouridas, Vangelis,Deniau, Eric
supporting information, p. 7078 - 7079 (2013/12/04)
A variety of diversely substituted aryl triflate esters were efficiently deprotected to the parent phenols by exposure to cesium carbonate in toluene. This procedure proved highly compatible with existing functional groups.
Method for producing an optically active 1-substituted 2-propanol
-
, (2008/06/13)
A method for producing an optically active 1-substituted 2-propanol of the following formula 1, which comprises reacting a hydroxy aromatic compound of the following formula 2 with an optically active propylene oxide in the presence of a catalyst:AOH??Formula 2CH3C*H(OH)CH2OA??Formula 1wherein A is a univalent aromatic group, and C* is an asymmetric carbon atom.