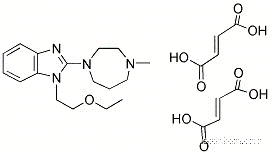
87233-62-3 Usage
Description
Emedastine Fumarate, also known as Emedastine Difumarate, is a potent H1-receptor antagonist that selectively targets histamine H1 receptors over H2, H3, and other receptors. It is used for its antiallergic effects, which are achieved through the inhibition of substance P-induced histamine release and by blocking Ca2+ release from extracellular stores and Ca2+ influx into mast cells. Emedastine Fumarate has demonstrated efficacy in improving symptoms of allergic rhinitis, urticaria, and asthma, as well as reducing vascular permeability and inhibiting the secretion of pro-inflammatory cytokines.
Used in Pharmaceutical Industry:
Emedastine Fumarate is used as an antiallergic agent for the treatment of allergic conditions such as allergic rhinitis, urticaria, and conjunctivitis. It is effective in reducing symptoms associated with these conditions by inhibiting histamine-induced responses and the release of pro-inflammatory cytokines.
Used in Ophthalmology:
Emedastine Fumarate is used as a topical ocular treatment for allergic conjunctivitis. It helps to alleviate symptoms such as itching, redness, and swelling by inhibiting histamine-induced vascular permeability and the secretion of pro-inflammatory cytokines in the conjunctiva.
Used in Respiratory Medicine:
Emedastine Fumarate is used as a therapeutic agent for bronchial asthma. In clinical trials, it has been shown to improve asthmatic symptoms in a significant percentage of patients, demonstrating its potential in managing respiratory allergic conditions.
Manufacturing Process
Preparation of 2-(4-methyl-1-piperazinyl)benzimidazole. A mixture of 2-
chlorobenzimidazole (10.00 g) and N-mehylpiperazine (20.00 g) is stirred at
125°C for 5 hours. A 10% aqueous sodium hydroxide (100 ml) is added to the
reaction mixture, and the precipitated crystals are separated by filtration. The
filtrate is extracted with chloroform, and the chloroform extract is evaporated
to dryness to give the same crystals. The crystals are combined and
recrystallized from water-methanol to give 2-(4-methyl-1-
piperazinyl)benzimidazole (7.02 g) as colorless needles, m.p. 225°-226°C.2-(4-Methyl-1-piperazinyl)benzimididazole (5.00 g) prepared as above is
dissolved in N,N-dimethylformamide (50 ml) and thereto is added sodium
hydride (concentration: 50%) (1.50 g) at room temperature, and the mixture
is stirred for 30 minutes. To the mixture is added 2-bromoethyl ethyl ether
(4.00 g), and the mixture is stirred at 70°C for 10 hours. To the reaction
mixture is added water (150 ml), and the mixture is extracted with ethyl
acetate. The extract is washed with water, dried over anhydrous magnesium
sulfate and then concentrated to give a brown oily substance (5.40 g). The
brown oily substance is treated with fumaric acid (3.26 g) in hot ethanol. The
crude crystals thus obtained are recrystallized from ethyl acetate-ethanol to
give 1-[2-(ethoxy)ethyl]-2-(4-methyl-1-piperazinyl)benzimidazole 3/2
fumarate (6.31 g) as colorless plates, melting point 167.5°-168.5°C.
Elementary analysis for C22H30N4O7: Calcd. (%): C, 57.13; H, 6.54; N, 12.11;
Found (%): C, 57.04; H, 6.44; N, 12.02.1-[2-(Ethoxy)ethyl]-2-(4-methyl-1-piperazinyl)benzimidazole can be prepared
using 2-chloro-(1-[2-(ethoxy)ethyl]benzimidazole), (last one can be produced
from 2-bromoethyl ethyl ether 2-chlorobenzimidazole) and N-methylpiperazine
and fumaric acid there are obtained crude crystals, which are recrystallized
from ethanol to give 1-[2-(ethoxy)ethyl]-2-(4-methyl-1-
piperazinyl)benzimidazole 3/2 fumarate. This product has the same physical
properties as those of the product above described.
Hazard
A poison by ingestion.
Check Digit Verification of cas no
The CAS Registry Mumber 87233-62-3 includes 8 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 5 digits, 8,7,2,3 and 3 respectively; the second part has 2 digits, 6 and 2 respectively.
Calculate Digit Verification of CAS Registry Number 87233-62:
(7*8)+(6*7)+(5*2)+(4*3)+(3*3)+(2*6)+(1*2)=143
143 % 10 = 3
So 87233-62-3 is a valid CAS Registry Number.
InChI:InChI=1/C17H26N4O.2C4H4O4/c1-3-22-14-13-21-16-8-5-4-7-15(16)18-17(21)20-10-6-9-19(2)11-12-20;2*5-3(6)1-2-4(7)8/h4-5,7-8H,3,6,9-14H2,1-2H3;2*1-2H,(H,5,6)(H,7,8)/b;2*2-1+