-
Name
AFLATOXIN G2
- EINECS 230-643-4
- CAS No. 7241-98-7
- Article Data3
- CAS DataBase
- Density 1.55 g/cm3
- Solubility 15mg/L(temperature not stated)
- Melting Point 237-240oC
- Formula C17H14 O7
- Boiling Point 602.5 °C at 760 mmHg
- Molecular Weight 330.294
- Flash Point 269.6 °C
- Transport Information UN 3462 6
- Appearance
- Safety Suspected carcinogen. Acute poison by ingestion. Mutation data reported. When heated to decomposition it emits acrid smoke and irritating fumes. See also various aflatoxins.
- Risk Codes 45-26/27/28
-
Molecular Structure
-
Hazard Symbols
 T+
T+
- Synonyms 1H,12H-Furo[3',2':4,5]furo[2,3-h]pyrano[3,4-c][1]benzopyran-1,12-dione,3,4,7a,9,10,10a-hexahydro-5-methoxy-, (7aR-cis)-;1H,12H-Furo[3',2':4,5]furo[2,3-h]pyrano[3,4-c][1]benzopyran-1,12-dione, 3,4,7aa,9,10,10aa-hexahydro-5-methoxy- (8CI);Aflatoxin G2 (7CI); Dihydroaflatoxin G1
- PSA 84.20000
- LogP 1.73690
AFLATOXIN G2 Chemical Properties
Product Name: Aflatoxin G2 (CAS NO.7241-98-7)
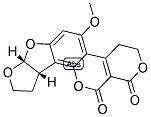
Molecular Formula: C17H14O7
Molecular Weight: 330.29g/mol
Mol File: 7241-98-7.mol
Einecs: 230-643-4
Appearance: Yellow powder
Melting Point: 237-240 °C
Boiling point: 602.5 °C at 760 mmHg
Storage Temperature: 2-8°C
Flash Point: 269.7 °C
Density: 1.55 g/cm3
Surface Tension: 66.9 dyne/cm
Enthalpy of Vaporization: 89.64 kJ/mol
Vapour Pressure: 1.8E-14 mmHg at 25°C
XLogP3-AA: 1.5
H-Bond Donor: 0
H-Bond Acceptor: 7
Product Categories: CRM and Matrix RMApplication CRMs; Food and Agriculture CRM; Other; CarcinogensCell Signaling and Neuroscience; Cancer Research; Mold; Toxins and Venoms; A;AA to ALBiotoxins; Alphabetic; Mycotoxins; Single component solutions; Biotoxins
AFLATOXIN G2 Toxicity Data With Reference
| 1. | dns-rat:lvr 10 µmol/L/1H | CNREA8 Cancer Research. 37 (1977),1845. | ||
| 2. | sce-ham:lng 3300 µg/L | CRNGDP Carcinogenesis. 1 (1980),759. | ||
| 3. | orl-dck LD50:2450 µg/kg | NATUAS Nature. 200 (1963),1101. |
AFLATOXIN G2 Consensus Reports
IARC Cancer Review: Animal Inadequate Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 1 (1972),p. 145.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) . EPA Genetic Toxicology Program.
AFLATOXIN G2 Safety Profile
Suspected carcinogen. Acute poison by ingestion. Mutation data reported. When heated to decomposition it emits acrid smoke and irritating fumes. See also various aflatoxins.
Safety Information of Aflatoxin G2 (CAS NO.7241-98-7):
Hazard Codes: T+![]() ,T
,T![]() ,Xn
,Xn![]() ,F
,F![]()
Risk Statements: 45-26/27/28-36-20/21/22-11-39/23/24/25-23/24/25-65-48/23/24/25-36/38-46
11: Highly Flammable
20: Harmful by inhalation
21: Harmful in contact with skin
22: Harmful if swallowed
23: Toxic by inhalation
24: Toxic in contact with skin
25: Toxic if swallowed
26: Very Toxic by inhalation
27: Very Toxic in contact with skin
28: Very Toxic if swallowed
36: Irritating to the eyes
38: Irritating to the skin
39: Danger of very serious irreversible effects
45: May cause cancer
46: May cause heritable genetic damage
48: Danger of serious damage to health by prolonged exposure
Safety Statements: 53-28-36/37-45-36-26-16-7-62
7: Keep container tightly closed
16: Keep away from sources of ignition - No smoking
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
28: After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
36: Wear suitable protective clothing
37: Wear suitable gloves
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
53: Avoid exposure - obtain special instruction before use
62: If swallowed, do not induce vomiting: seek medical advice immediately and show this container or label
AFLATOXIN G2 Specification
Aflatoxin G2 ,its CAS NO. is 7241-98-7,the synonyms is Pyrano(3,4-c)(1)-benzopyran-1,12-dione ; 10a-Hexahydro-5-methoxy-1(7ar-cis)- ; 12h-Furo(3',2':4,5)furo(2,3-h)pyrano(3,4-c)(1)benzopyran-1,12-dione,3,4,7a-1 ;1 h,12h-Furo(3',2':4,5)furo(2,3-h)pyrano(3,4-c)(1)benzopyran-1,12-dione,3,4,7aa ; 1H,12H-Furo[3',2':4,5]furo[2,3-h]pyrano[3,4-c][1]benzopyran-1,12-dione, 3,4,7a,9,10,10a-hexahydro-5-methoxy-, (7aR-cis)- ; 1H,12H-Furo[3',2':4,5]furo[2,3-h]pyrano[3,4-c][1]benzopyran-1,12-dione, 3,4,7aalpha,9,10,10aalpha-hexahydro-5-methoxy- ; 3,4,7Aalpha,9,10,10aalpha-hexahydro-5-methoxy-1h,12h-furo(3',2':4,5)furo(2,3-h;5-Methoxy-3,4,7a,9,10,10a-hexahydro-1H,12H-furo[3',2':4,5]furo[2,3-H]pyrano[3,4-c]chromene-1,12-dione .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View