-
Name
Acetylenedicarboxylic acid monopotassium salt
- EINECS 213-169-2
- CAS No. 928-04-1
- Article Data2
- CAS DataBase
- Density
- Solubility slightly soluble in water(20 °C)
- Melting Point 133 °C
- Formula C4HKO4
- Boiling Point 362.4oC at 760 mmHg
- Molecular Weight 152.148
- Flash Point 187.2oC
- Transport Information UN 2811 6.1/PG 1
- Appearance white powder
- Safety 26-36/37/39-45
- Risk Codes 23/25-27-36/37/38
-
Molecular Structure
-
Hazard Symbols
 T+
T+
- Synonyms potassium,4-hydroxy-4-oxobut-2-ynoate
- PSA 77.43000
- LogP -2.17570
Synthetic route
-
-
67-56-1
methanol

-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

-
-
542-92-7
cyclopenta-1,3-diene

-
-
947-57-9
dimethyl bicyclo[2.2.1]hept-2,5-diene-2,3-dicarboxylate

| Conditions | Yield |
|---|---|
| Stage #1: methanol; acetylene dicarboxylic acid mono potassium salt With hydrogenchloride In 1,4-dioxane for 0.166667h; Stage #2: cyclopenta-1,3-diene In 1,4-dioxane; methanol at 20℃; Diels-Alder Cycloaddition; | 97% |

-
-
64-17-5
ethanol

-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

-
-
762-21-0
acetylenedicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid In benzene for 24h; Heating; | 95% |

| Conditions | Yield |
|---|---|
| 85% | |
| With hydrogen iodide |

| Conditions | Yield |
|---|---|
| With bromine; sodium bromide; potassium hydroxide In water at 20℃; for 24h; | 80.47% |
| Stage #1: acetylene dicarboxylic acid mono potassium salt With bromine; potassium hydroxide Stage #2: With hydrogenchloride |
-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

-
-
542-92-7
cyclopenta-1,3-diene

-
-
15872-28-3
bicyclo[2.2.1]hepta-2,5-diene-2,3-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 25℃; for 4h; Diels-Alder Cycloaddition; | 79% |
| With hydrogenchloride In water for 0.333333h; Ambient temperature; | 38% |

| Conditions | Yield |
|---|---|
| With water-d2 at 100℃; for 2h; | 78% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

-
-
78619-83-7
Acetylenedicarboxylic acid monotrimethylsilyl ester

| Conditions | Yield |
|---|---|
| In benzene 1.) cooled (freezing mixture), 10 min, 2.) reflux, 5 h; | 60% |
-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

-
-
18910-53-7
<3-2H>propiolic acid

| Conditions | Yield |
|---|---|
| With water-d2 a) from RT to reflux, 2 h, b) reflux, 1 h; | 54% |
| With water-d2 for 2h; Heating; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) for 6h; Inert atmosphere; Reflux; | 53% |


-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

| Conditions | Yield |
|---|---|
| In water; acetonitrile addn. of potassium salt in water to V-complex in MeCN, stirring (room temp., 3h), pptn.; filtration off, washing (water, MeCN, Et2O), drying (desiccator, 2 h), recrystn. (CH2Cl2/toluene 1/1 v/v); elem. anal.; | 51% |
-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

-
-
14493-42-6
2,2,3,3-tetradeuterio-succinic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide; sodium amalgam; water-d2 1.) water, room temperature, 2 d, 2.) 1 h; Multistep reaction; |
-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

-
-
82203-73-4
C4O4(2-)*Ba(2+)

| Conditions | Yield |
|---|---|
| With sodium hydroxide; barium(II) chloride Multistep reaction; |
-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

-
-
7732-18-5
water

-
A
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

-
A
-
124-38-9
carbon dioxide

-
B
-
44196-71-6
propiolate

| Conditions | Yield |
|---|---|
| In water at 80℃; under 206266 Torr; Kinetics; Activation energy; Further Variations:; Temperatures; |

-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

| Conditions | Yield |
|---|---|
| With hydrogenchloride; chlorosulfonic acid; potassium hydroxide In methanol; dichloromethane; water; Petroleum ether |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium hydrogencarbonate In water |

| Conditions | Yield |
|---|---|
| In water |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water |
-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

-
-
42369-42-6
2,4-diisopropylthiophenol

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water |
-
-
29236-93-9
2,5-Diethoxythiophenol

-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water |
-
-
42369-53-9
5-(3-methylbutoxy)-2-propylthiophenol

-
-
928-04-1
acetylene dicarboxylic acid mono potassium salt

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water |
Acetylenedicarboxylic acid monopotassium salt Consensus Reports
Reported in EPA TSCA Inventory.
Acetylenedicarboxylic acid monopotassium salt Specification
The Acetylenedicarboxylic acid monopotassium salt, with the CAS registry number 928-04-1, is also known as 2-Butynedioic acid monopotassium salt. It belongs to the product categories of Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts; Acetylenes; Acetylenic Carboxylic Acids & Their Derivatives. Its EINECS registry number is 213-169-2. This chemical's molecular formula is C4HKO4 and molecular weight is 152.15. What's more, its IUPAC name is called Potassium 4-hydroxy-4-oxobut-2-ynoate. It should be stored in a cool, dry and well-ventilated place.
Physical properties about Acetylenedicarboxylic acid monopotassium salt are: (1)#H bond acceptors: 4; (2)#H bond donors: 2; (3)#Freely Rotating Bonds: 1; (4)Polar Surface Area: 80.26 Å2.
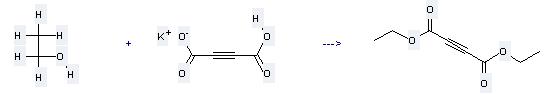
Uses of Acetylenedicarboxylic acid monopotassium salt: (1) it is used as organic intermediate; (2) it is used to produce other chemicals. For example, it can react with ethanol to get butynedioic acid diethyl ester. The reaction occurs with reagent H2SO4 and other condition of heating for 24 hours. The yield is 95 %.
When you are dealing with this chemical, you should be very careful. This chemical causes damage to health at very low levels. It is toxic by inhalation and if swallowed. This chemical is irritating to eyes, respiratory system and skin. Therefore, you should wear suitable protective clothing, gloves and eye/face protection. In case of contacting with eyes, you should rinse immediately with plenty of water and seek medical advice. And in case of accident or if you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
(1) SMILES: [K+].[O-]C(=O)C#CC([O-])=O
(2) InChI: InChI=1S/C4H2O4.K/c5-3(6)1-2-4(7)8;/h(H,5,6)(H,7,8);/q;+1/p-2
(3) InChIKey: KLLYWRUTRAFSJT-UHFFFAOYSA-L
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | skin | 25mg/kg (25mg/kg) | United States Environmental Protection Agency, Office of Pesticides and Toxic Substances. Vol. 8EHQ-1278-0263, | |
| mouse | LD50 | intraperitoneal | 32mg/kg (32mg/kg) | Toxicology and Applied Pharmacology. Vol. 17, Pg. 733, 1970. | |
| mouse | LD50 | intravenous | 89mg/kg (89mg/kg) | Toxicology and Applied Pharmacology. Vol. 17, Pg. 733, 1970. | |
| mouse | LD50 | oral | 63mg/kg (63mg/kg) | Toxicology and Applied Pharmacology. Vol. 17, Pg. 733, 1970. |
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View