-
Name
Allyl acetate
- EINECS 209-734-8
- CAS No. 591-87-7
- Article Data149
- CAS DataBase
- Density 0.913 g/cm3
- Solubility slightly soluble in water
- Melting Point 6 °C
- Formula C5H8O2
- Boiling Point 103.999 °C at 760 mmHg
- Molecular Weight 100.117
- Flash Point 6.667 °C
- Transport Information UN 2333
- Appearance Clear colourless liquid
- Safety 16-27-45
- Risk Codes 11-21-25
-
Molecular Structure
-
Hazard Symbols
 F,
F, T
T
- Synonyms Aceticacid, 2-propenyl ester (9CI);Acetic acid, allyl ester (8CI);Allyl alcohol,acetate (6CI);2-Propen-1-ol acetate;3-Acetoxy-1-propene;3-Acetoxypropene;Acetic acid,2-propen-1-yl ester;NSC 7612;
- PSA 26.30000
- LogP 0.73550
Allyl acetate Chemical Properties
IUPAC Name: Prop-2-enyl acetate
Molecular Formula: C5H8O2
Formula Weight: 100.12 g/mol
EINECS: 209-734-8
Melting Point: 6 °C
Boiling Point: 104 °C at 760 mmHg
Flash Point: 44 °F
Index of Refraction: 1.402
Molar Refractivity: 26.71 cm3
Molar Volume: 109.6 cm3
Surface Tension: 24.8 dyne/cm
Density: 0.913 g/cm3
Enthalpy of Vaporization: 34.31 kJ/mol
Vapour Pressure: 31.4 mmHg at 25 °C
Structure of Allyl acetate (CAS NO.591-87-7):
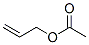
XLogP3: 1
H-Bond Acceptor: 2
Rotatable Bond Count: 3
Exact Mass: 100.05243
MonoIsotopic Mass: 100.05243
Topological Polar Surface Area: 26.3
Canonical SMILES: CC(=O)OCC=C
InChI: InChI=1S/C5H8O2/c1-3-4-7-5(2)6/h3H,1,4H2,2H3
InChIKey: FWZUNOYOVVKUNF-UHFFFAOYSA-N
Product Categories of Allyl acetate (591-87-7): Allyl monomers ; Monomers ; Polymer Science
Allyl acetate Uses
Allyl acetate (CAS NO.591-87-7) is used in organic synthesis.
Allyl acetate Production
Allyl acetate is generated by the gas phase reaction of propene, acetic acid, and oxygen over a palladium catalyst.
C3H6 + CH3COOH + ½ O2 → CH2=CHCH2OCOCH3 + H2O
Allyl acetate Toxicity Data With Reference
| 1. | skn-rbt 10 mg/24H MLD | JIHTAB Journal of Industrial Hygiene and Toxicology. 31 (1949),60. | ||
| 2. | eye-rbt 100 mg/24H MOD | 85JCAE Prehled Prumyslove Toxikologie; Organicke Latky Marhold, J.,Prague, Czechoslovakia.: Avicenum,1986,355. | ||
| 3. | orl-rat LD50:130 mg/kg | JIHTAB Journal of Industrial Hygiene and Toxicology. 31 (1949),60. | ||
| 4. | ihl-rat LC50:1000 ppm/1H | AMIHAB AMA Archives of Industrial Health. 21 (1960),28. | ||
| 5. | orl-mus LD50:170 mg/kg | FCTXAV Food and Cosmetics Toxicology. 2 (1964),327. | ||
| 6. | skn-rbt LD50:1021 mg/kg | JIHTAB Journal of Industrial Hygiene and Toxicology. 31 (1949),60. |
Allyl acetate Consensus Reports
Reported in EPA TSCA Inventory.
Allyl acetate Safety Profile
Hazard Codes:  F,
F,  T
T
Risk Statements: 11-21-23/25-36-25
R11:Highly flammable.
R21:Harmful in contact with skin.
R23/25:Toxic by inhalation and if swallowed.
R36:Irritating to eyes.
Safety Statements: 16-26-36-45-27
S16:Keep away from sources of ignition.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S27:Take off immediately all contaminated clothing.
WGK Germany: 3
HazardClass: 3
RTECS: AF1750000
PackingGroup: II
HS Code: 29153990
Allyl acetate Standards and Recommendations
DOT Classification: 3; Label: Flammable Liquid, Poison
Allyl acetate Specification
Allyl acetate (CAS NO.591-87-7) is also named as 2-Propenyl acetate ; 2-Propenyl ethanoate ; 2-Propenyl methanoate ; 3-Acetoxy-1-propene ; 3-Acetoxypropene ; 4-02-00-00180 (Beilstein Handbook Reference) ; AI3-22625 ; Acetic acid, 2-propenyl ester ; Allyl acetic acid ; BRN 1742050 ; HSDB 2697 ; NSC 7612 . Allyl acetate (CAS NO.591-87-7) is clear colourless liquid. It is insoluble in water and slightly less dense than water. Poisonous by ingestion and moderately toxic by inhalation and skin contact. Irritating to skin and eyes. Allyl acetate is an ester. Reacts with acids to liberate heat along with alcohols and acids. Generates heat with strong oxidizing acids. The reaction may be sufficiently exothermic to ignite the reaction products. Also generates heat with basic solutions. Generates flammable hydrogen with alkali metals and hydrides. Emits acrid smoke and irritating fumes when heated to decomposition. Fire will produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution. It will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion and poison hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.
Related Products
- ALLYL α-IONONE
- Allyl (2-methylbutoxy)acetate
- Allyl (3-methylbutoxy)acetate
- Allyl 2-(acetylamino)-2-deoxy-3-O-benzyl--D-glucopyranoside
- Allyl 2-acetamido-2-deoxy-beta-D-glucopyranoside
- Allyl 2-chloroethylsulfide
- Allyl 2-ethylbutyrate
- Allyl 2-oxocyclopentanecarboxylate
- Allyl 3,4-epoxy-6-methylcyclohexanecarboxylate
- Allyl 3,5,5-trimethylhexanoate
- 59188-53-3
- 5918-93-4
- 59189-97-8
- 59191-99-0
- 59194-26-2
- 5919-74-4
- 591-97-9
- 59198-70-8
- 592-02-9
- 59202-96-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View