-
Name
Aluminum bromide
- EINECS 231-779-7
- CAS No. 7727-15-3
- Article Data70
- CAS DataBase
- Density 3.205 g/mL at 25 °C(lit.)
- Solubility decomposes in water
- Melting Point 94-98 °C(lit.)
- Formula AlBr3
- Boiling Point 265 °C
- Molecular Weight 266.694
- Flash Point 268°C subl.
- Transport Information UN 3264 8/PG 2
- Appearance white crystalline powder
- Safety 9-16-26-29-36/37/39-43-45-60-61-62-28-27
- Risk Codes 23/24/25-34-22-14-67-65-50/53-11
-
Molecular Structure
-
Hazard Symbols
 C,
C, Xi,
Xi, N,
N, F
F
- Synonyms Aluminumbromide (8CI);Aluminum tribromide;Aluminum tribromide (AlBr3);NSC 87608;Aluminum bromide(AlBr3);
- PSA 0.00000
- LogP 2.53680
Synthetic route
-
-
3530-39-0
phenylaluminium dichloride

-
-
106-95-6
allyl bromide

-
A
-
300-57-2
allylbenzene

-
B
-
92-52-4
biphenyl

-
C
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| bis(η3-allyl-μ-chloropalladium(II)) In diethyl ether Ar atmosphere; stirring (20°C, 5 mole-% catalyst, 8 h); | A 94% B <1 C n/a |
| tetrakis(triphenylphosphine) palladium(0) In diethyl ether Ar atmosphere; stirring (20°C, 5 mole-% catalyst, 20 h); | A <1 B <1 C n/a |

-
-
6591-30-6
diphenylaluminum chloride

-
-
106-95-6
allyl bromide

-
A
-
300-57-2
allylbenzene

-
B
-
92-52-4
biphenyl

-
C
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) In diethyl ether Ar atmosphere; stirring (20°C, 5 mole-% catalyst, 20 h); | A 37% B <1 C n/a |



| Conditions | Yield |
|---|---|
| Erhitzen des Reaktionsprodukts im Vakuum auf 200grad; |
-
-
1333-84-2, 1344-28-1
aluminum oxide

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| With bromine; pyrographite In not given heating; | |
| With bromine; pyrographite heating; | |
| With sulfur dibromide In solid 350°C, with vaporous sulfur bromide; |

| Conditions | Yield |
|---|---|
| reaction started at 350°C and accelerated by raising of temp.; excess of S2Br2 removed with HBr or nitrogen; | |
| reaction started at 350°C and accelerated by raising of temp.; excess of S2Br2 removed with HBr or nitrogen; |

| Conditions | Yield |
|---|---|
| With aluminum(III) sulfate In water heating on water bath with small amt. of Br2 and H2O; | |
| With bisilicates; carbon heating; | |
| With carbon; silicates heating; |

| Conditions | Yield |
|---|---|
| In not given heating on water bath with small amt. of Br2; | |
| In not given heating on water bath with small amt. of Br2; |

| Conditions | Yield |
|---|---|
| In gas Al and Br2 in vac. were vaporized separately and reacted in gaseous state; distd. in vac.; | |
| With air; water in moist air containing Br2 condensation of droplets of a soln. of AlBr3 occurs on a gray layer that has formed first;; | |
| In neat (no solvent) start of react. by heating;; |

| Conditions | Yield |
|---|---|
| With bromine reacted in the presence of AlBr3; | |
| With bromine Al heated in combustion tube; with H2 or N2 deluted Br2 lead over Al; | |
| With bromine Al sheet heated in combustion tube; vaporous Br2 leaded over Al; AlBr3 flowed out from tube; |

| Conditions | Yield |
|---|---|
| In ethylene dibromide | |
| In ethylene dibromide |


| Conditions | Yield |
|---|---|
| In not given heating of Al with SnBr4 for 30 min gives AlBr3;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction with SnBr2 under formation of AlBr3 above 216°C;; differentialthermoanalytic examinations;; |

| Conditions | Yield |
|---|---|
| at higher temp.;; |


-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| byproducts: Br2, CS2; decompn. at 150°C; | |
| byproducts: Br2, CS2; decompn. at 150°C; |

| Conditions | Yield |
|---|---|
| at 200°C; | |
| at 200°C; |

| Conditions | Yield |
|---|---|
| 120°C; | |
| 120°C; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) mixt. of the starting materials heated in evacuated sealed quartz tubes to 500-900°C; decompn. of educts ocurrs, AlBr3 sublimates off; | A 0% B n/a |


| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction at 100°C;; | |
| In neat (no solvent) reaction at 100°C;; |

-
-
10097-32-2
bromine

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| With sulfur introducing a vapor of a soln. of S in Br2 over Al oxide; | |
| With S introducing a vapor of a soln. of S in Br2 over Al oxide; |
-
-
119484-32-1
AsBr4(1+)*AlBr4(1-)={AsBr4}{AlBr4}

-
A
-
7726-95-6
bromine

-
B
-
7727-15-3
aluminium bromide

-
C
-
7784-33-0
arsenic(III) bromide

| Conditions | Yield |
|---|---|
| equilibrium at room temp.; |


-
B
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| With copper(l) chloride In tetrahydrofuran byproducts: Cu, LiCl; to a soln. of Ti-complex in THF at 0°C is added CuCl; pptn. of Cu, solvent is removed in vac., the residue is dissolved in toluene, pptd. LiCl is centrifuged off, the soln. is vacuum-evaporated, from the residue at 90-110°C is distilled the complex, crystn. at 20°C, elem. anal.; |

-
A
-
14741-34-5, 21255-52-7
tricarbonyl bis(triphenylphosphine) iron

-
B
-
51744-69-5, 15020-31-2, 84622-99-1
bis(triphenylphosphine)dicarbonyldibromoiron(II)

-
C
-
7726-95-6
bromine

-
D
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran | A <1 B n/a C n/a D n/a |
| In methanol | A <1 B n/a C n/a D n/a |
| In dichloromethane | A <1 B n/a C n/a D n/a |

-
-
361447-05-4
tetraiodophosphonium tetrabromoaluminate

-
A
-
7727-15-3
aluminium bromide

-
B
-
7789-33-5
iodine(I) bromide

-
C
-
13455-01-1
phosphorous triiodide

| Conditions | Yield |
|---|---|
| In carbon disulfide under N2, anaerobic and anhyd. conditions; dissolved in CS2; |


-
A
-
22359-97-3
aluminium(I) bromide

-
B
-
136684-48-5
AlBr2

-
C
-
74-90-8, 6914-07-4
hydrogen isocyanide

-
D
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| In neat (no solvent, gas phase); solid matrix byproducts: CH4, H2C=CH2, H2C=CHCH3; N2 atmosphere, high vacuum (1E-6 to 1E-7 mbar) thermolysis at 1000°C, trapping in argon at 15 K; further organic byproducts: H2C=N(CH3)2 and H2CCHCH2; IR-monitoring; |


| Conditions | Yield |
|---|---|
| In solid heating at 400°C; |
-
-
16853-85-3
lithium aluminium tetrahydride

-
-
124-11-8
non-1-ene

-
-
7727-15-3
aluminium bromide

-
-
180334-67-2
1-dibromoaluminiononane

| Conditions | Yield |
|---|---|
| In diethyl ether; benzene Ar, LiAlH4 in ether added to benzene under stirring, most of the solvent removed, benzene and AlBr3 in benzene added, stirred at 20°C for 1 h, org. compound in benzene added dropwise under stirring, warmed to30-40°C for 5-10 min; NMR; | 100% |

-
-
7727-15-3
aluminium bromide

-
-
920-36-5, 90269-51-5
(2-methylpropyl)lithium

| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: LiBr; to stirred mixt. of MgBr2, AlBr3 and toluene at room temp., soln. of iso-C4H9Li in hexane added (Ar; ratio MgBr2, AlBr3, RLi: 1:2:8) within 20 min; react. for 3.0 h at 80°C; ppt. filtered off (Ar); (1)H NMR of soln.; | 100% |


| Conditions | Yield |
|---|---|
| In benzene under N2, dropwise addn. of thiol soln. to a suspension of AlBr3 in benzene, dissoln. of AlBr3, stirred for 15 min at room temp.; evapn. of solvent, dried in vac. at room temp. for 3h, elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| With tetraallyltin In benzene byproducts: SnBr4, propene; 2,6-diphenylphenol (12 equiv.) treated with AlBr3 (4 equiv.) in benzene at room temp., tetraallyltin (3 equiv.) added, stirred at room temp. for1 h, solvent evapd. in vacuo for 3 h with concomitant removal of SnBr4 and propene; | 100% |

| Conditions | Yield |
|---|---|
| In benzene under N2, CH3SH introduced in a suspension of AlBr3 for 7 min, dissoln.of AlBr3, stirred for 15 min at room temp.; evapn. of solvent, dried in vac. at room temp. for 3h, elem. anal.; | 100% |
-
-
3908-55-2
Trimethyl(methylthio)silane

-
-
7727-15-3
aluminium bromide

-
-
112908-14-2
aluminium tribromide*trimethylsilyl methyl sulphide

| Conditions | Yield |
|---|---|
| In benzene a benzene soln. of Me3SiSMe was added dropwise under N2 to a suspn. of AlBr3 in benzene, mixt. was stirred for 15 min at room temp.; solvent was evapd.; elem. anal.; | 100% |
-
-
5573-62-6
(ethylthio)trimethylsilane

-
-
7727-15-3
aluminium bromide

-
-
112908-15-3
aluminium tribromide*trimethylsilyl ethyl sulphide

| Conditions | Yield |
|---|---|
| In benzene a benzene soln. of Me3SiSEt was added dropwise under N2 to a suspn. of AlBr3 in benzene, mixt. was stirred for 15 min at room temp.; solvent was evapd.; elem. anal.; | 100% |
-
-
4551-15-9
phenylthiotrimethylsilane

-
-
7727-15-3
aluminium bromide

-
-
112908-16-4
aluminium tribromide*trimethylsilyl phenyl sulphide

| Conditions | Yield |
|---|---|
| In benzene a benzene soln. of Me3SiSPh was added dropwise under N2 to a suspn. of AlBr3 in benzene, mixt. was stirred for 15 min at room temp.; solvent was evapd.; elem. anal.; | 100% |

-
-
7727-15-3
aluminium bromide

-
-
374569-92-3
supersilylaluminum dibromide

| Conditions | Yield |
|---|---|
| In n-heptane Kinetics; all manipulations with exclusion of air and moisture; compd. prepd. fromAlBr3 and org. compd. reacted in molar ratio of 1:1 at room temp. for 2 4 d (yield depended on react. time); controlled by NMR; volatiles evapd. by oil pump, residue dissolved in heptane, NaBr filtered; | 100% |
-
-
7789-65-3
selenium tetrabromide

-
-
7727-15-3
aluminium bromide

-
-
69951-93-5
tribromoselenium(IV) tetrabromoaluminate

| Conditions | Yield |
|---|---|
| In neat (no solvent) Ar atm. (300 mbar); heating (150-200°C, several days); | 100% |
| In dichloromethane addn. of AlBr3 to SeBr4, stirring (2 h); filtration (under dry N2), drying (vac.); elem. anal.; |

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| In dichloromethane; ethylene dibromide | 100% |

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| In dichloromethane; ethylene dibromide | 100% |

-
-
7727-15-3
aluminium bromide

-
-
79560-37-5
bis(η5-cyclopentadienyl)tungsten dihydride * AlBr3

| Conditions | Yield |
|---|---|
| In diethyl ether stoich. amts. (pptn.); | 99% |
-
-
55664-33-0
bis(tricyclohexylphosphine)platinum(0)

-
-
7727-15-3
aluminium bromide

-
-
960294-94-4
[(Cy3P)2Pt(AlBr3)]

| Conditions | Yield |
|---|---|
| In benzene AlBr3 was added to a soln. of Pt-complex in benzene, the soln. was kept at room temp. for 24 h; | 99% |
| In toluene (Ar); Al salt added to a soln. of Pt complex; evapd. (vac.); elem. anal.; | 98% |

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) (Ar); solid state reaction; equimolar mixt. of TlBr, and AlBr3 sealed under vac. in glass ampoules; heated with 10°C/h to 150°C for 48 h; cooled to 25°C with 2.degre.C/h; detn. by XRD; | 99% |
-
-
110-71-4
1,2-dimethoxyethane

-
-
7727-15-3
aluminium bromide

-
-
182561-85-9
aluminium tribromide-1,2-dimethoxyethane

| Conditions | Yield |
|---|---|
| In toluene (N2); stirring (1 h); filtration, washing (toluene); elem. anal.; | 98.7% |

| Conditions | Yield |
|---|---|
| In benzene (glovebox); addn. of a soln. of AlBr3 in C6H6 to a soln. of silicon compd. in C6H6 in vac. with stirring, sealing, heating to 40°C for 30min with shaking; centrifugation, decantation, evapn; elem. anal.; | A 98% B 98% |

-
-
176763-62-5
(1R,2R)-(-)-N,N'-bis(3,5-di-tert-butylsalicydene)-1,2-cyclohexanediaminocobalt(II)

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| In dichloromethane; water addn. of Co(C6H10(NCHC6H2O(t-Bu)2)2) (1 equiv.) to soln. of AlBr3 (1 equiv.) in CH2Cl2/H2O; stirring at room temp. for 1 h; concn. under reduced pressure, treatment with H2O and CH2Cl2; | 98% |

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| With I2 In dichloromethane a mixt. of B compd., I2, and AlBr3 in CH2Cl2 refluxed for 16.5 h under N2; poured into ice-cold H2O, the org. phase sepd., the aq. layer extd. (Et2O), the combined org. phase washed (Na2S2O3 soln., then H2O), dried (MgSO4), solvent-removed, recrystd. (hexane); elem. anal.; | 98% |
-
-
109-99-9
tetrahydrofuran

-
-
97355-16-3
N,N-bis(2-hydroxy-3,5-di-tert-butylphenyl)amine

-
-
7727-15-3
aluminium bromide

-
-
1384526-06-0
[bis(3,5-di-tert-butyl-2-phenol)amine(2-)]AlBr(THF)

| Conditions | Yield |
|---|---|
| With KH In tetrahydrofuran under inert atm. mixt. ligand and KH in THF were stirred at room temp. for 2 h, soln. was frozen, AlBr3 was added upon thawing, stirred at room temp. overnight; solvent was removed in vacuo, residue was co-evapd. with pentane; elem. anal.; | 98% |


| Conditions | Yield |
|---|---|
| In methyl cyclohexane addn. of 2-butyne to soln. of AlBr3, 0°C, stirred 15h at 25°C, filtered, washed with methylcyclohexane, dried in vac.; elem. anal.; | 97% |

| Conditions | Yield |
|---|---|
| In n-heptane room temp.; | 97% |
| In benzene room temp.; | 97% |
| In benzene room temp.; | 97% |
-
-
7727-15-3
aluminium bromide

-
-
653-38-3
(pentafluoro phenyl) methyl mercury

-
-
4457-90-3
(pentafluoro phenyl) dibromo alane

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating of the components ( molar ratio 1 : 1) in a closed tube at 70°C, 5 days;; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: uranium dioxide; aluminium bromide at 250℃; for 12h; Sealed tube; Stage #2: at 350℃; for 144h; Sealed tube; | 97% |

| Conditions | Yield |
|---|---|
| In diethyl ether; benzene Ar, LiAlH4 in ether added to benzene under stirring, most of the solvent removed, benzene and AlBr3 in benzene added, stirred at 20°C for 1 h, org. compound in benzene added drowise under stirring, warmed to 30-40°C for 5-10 min; NMR; | 96% |

-
-
26944-84-3
1,3-dimethyl-2-dimethylaminodiazaborolane

-
-
7727-15-3
aluminium bromide

| Conditions | Yield |
|---|---|
| under N2, at -78°C; excess of Lewis-acid used; elem. anal.; | 96% |

| Conditions | Yield |
|---|---|
| In toluene N2-atmosphere; slow addn. of Al-salt to slight excess of alcohol at 25°C, stirring for 4 h; drying (vac.), extn. into CH2Cl2, drying (vac.); elem. anal.; | 96% |

| Conditions | Yield |
|---|---|
| In benzene (glovebox); dropwise addn. of a soln. of AlBr3 in C6H6 to a soln. of silicon compd. in C6H6 over 15 min with stirring, refluxing for 7 h; centrifugation, decantation, evapn.; elem. anal.; | A 88% B 96% |

-
-
211381-77-0
(2,2,6,6-tetramethylpiperidino)2AlBr(isoquinoline)

-
-
7727-15-3
aluminium bromide

-
-
211381-88-3
(2,2,6,6-tetramethylpiperidino)2Al(isoquinoline) tetrabromoaluminate

| Conditions | Yield |
|---|---|
| In Bromoform inert atmosphere, addn. of soln. of AlBr3 to soln of isoquinoline complex at 0°C, 30 min; reduction of volume, crystn. (-20°C, overnight); elem. anal.; | 96% |
-
-
16853-85-3
lithium aluminium tetrahydride

-
-
625-65-0
2,4-dimethyl-2-pentene

-
-
7727-15-3
aluminium bromide

-
-
180334-71-8
(CH3)2CHCH(AlBr2)CH(CH3)CH3

| Conditions | Yield |
|---|---|
| In diethyl ether; benzene Ar, LiAlH4 in ether added to benzene under stirring, most of the solvent removed, benzene and AlBr3 in benzene added, stirred at 20°C for 1 h, org. compound in benzene added drowise under stirring, warmed to 30-40°C for 5-10 min; NMR; | 96% |

Aluminium bromide Chemical Properties
IUPAC Name: Tribromoalumane
Molecular Formula: AlBr3
Formula Weight: 266.69g/mol
EINECS: 231-779-7
Melting Point: 94-98 °C(lit.)
Boiling Point: 265 °C
Density: 3.205 g/mL at 25 °C(lit.)
Vapor Pressure: 1 mm Hg ( 81.3 °C)
Flash Point: 268°C subl.
Chemical Properties: White crystalline powder
Water Solubility: Decomposes
Sensitive: Moisture sensitive
Stability: Stable, but reacts violently with water. Incompatible with aqueous solutions, alcohols, acids.
Synonyms of Aluminium bromide (7727-15-3): Aluminum bromide ; Aluminum bromide Hexahydrate ; Aluminium tribromide ; AlBr3 ; Aluminum bromide (AlBr3); Aluminum bromide , anhydrous ; Aluminumbromide (albr3)
Product Categories of Aluminium bromide (7727-15-3): Inorganics ; Aluminum salts ; Aluminummetal and ceramic science ; Aluminumsynthetic reagents ; Catalysis and inorganic chemistry ; Chemical synthesis ; Crystal grade inorganics ; Lewis acids ; Salts
Structure of Aluminium bromide (7727-15-3):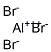
Aluminium bromide Safety Profile
Safety Statements: 9-16-26-29-36/37/39-43-45
9: Keep container in a well-ventilated place
16: Keep away from sources of ignition - No smoking
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
29: Do not empty into drains
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
43: In case of fire, use ... (indicate in the space the precise type of fire-fighting equipment. If water increases the risk add - Never use water)
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
WGK Germany: 3
Hazard Note: Irritant
Hazard Class: 8
Risk Statements: 23/24/25-34-22-14-67-65-50/53-11
23/24/25: Toxic by inhalation, in contact with skin and if swallowed
34: Causes burns
22: Harmful if swallowed
14: Reacts violently with water
67: Vapors may cause drowsiness and dizziness
65: Harmful: May cause lung damage if swallowed
50/53: Very Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
11: Highly Flammable
Aluminium bromide Specification
General Description: A white to yellowish-red lumpy solid with a pungent odor.
Related Products
- Aluminium
- Aluminium 2-ethylhexanoate
- Aluminium ammonium sulfate dodecahydrate
- Aluminium bromide
- Aluminium carboxymethyl cellulose
- Aluminium Chloride
- Aluminium chloride hexahydrate
- Aluminium dihydrogen triphosphate
- Aluminium fluoride trihydrate
- Aluminium glycinate
- 7727-18-6
- 77271-96-6
- 7727-21-1
- 7727-33-5
- 7727-37-9
- 7727-43-7
- 77275-38-8
- 7727-54-0
- 77275-51-5
- 77275-70-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View