-
Name
DIPROPYLENETRIAMINE
- EINECS 200-261-2
- CAS No. 56-18-8
- Article Data28
- CAS DataBase
- Density 0.912 g/cm3
- Solubility Completely miscible in water
- Melting Point -14 °C(lit.)
- Formula C6H17N3
- Boiling Point 242.1 °C at 760 mmHg
- Molecular Weight 131.221
- Flash Point 118.3 °C
- Transport Information UN 2269 8/PG 3
- Appearance colourless to yellow liquid
- Safety 26-28-36/37/39-45
- Risk Codes 22-24-26-35-43
-
Molecular Structure
-
Hazard Symbols
 T+,
T+, C
C
- Synonyms 1,3-Propanediamine,N-(3-aminopropyl)- (9CI);Dipropylamine, 3,3'-diamino- (6CI,8CI);1,5,9-Triazanonane;1,7-Diamino-4-azaheptane;1-Propanamine, 3,3'-iminobis-;3,3'-Diaminodipropylamine;3,3'-Iminobis(propylamine);3,3'-Iminodi(propylamine);4-Aza-1,7-diaminoheptane;4-Azaheptamethylenediamine;4-Azaheptane-1,7-diamine;Bis(3-aminopropyl)amine;Caldine;Di(3-aminopropyl)amine;Dipropylenetriamine;N-(3-Aminopropyl)-1,3-propanediamine;N-3-Aminopropyl-1,3-diaminopropane;NSC7773;Norspermidine;P 2 (hardener);sym-Norspermidine;
- PSA 64.07000
- LogP 1.06510
Synthetic route
-
-
503-29-7
azetidine

-
-
109-76-2
Trimethylenediamine

-
A
-
56-18-8
bis(3-aminopropyl)amine

-
B
-
54262-75-8
N-γ-aminopropyltrimethyleneimine

| Conditions | Yield |
|---|---|
| palladium at 170℃; | A 84% B 1% |
| palladium Product distribution; |


| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen; nickel In ethanol under 2068.6 Torr; | 81% |
| With sodium hydroxide; sodium tetrahydroborate; nickel In methanol at 50℃; for 0.5h; | 80% |
| With sodium-potassium alloy; butan-1-ol | |
| With ammonia; nickel at 90 - 125℃; under 36775.4 - 183877 Torr; Hydrogenation; |

-
-
56-18-8
bis(3-aminopropyl)amine

| Conditions | Yield |
|---|---|
| With nitrogen(II) oxide In acetonitrile | 55% |

| Conditions | Yield |
|---|---|
| With ethanol; ammonia; water |

| Conditions | Yield |
|---|---|
| With ammonia |
-
-
35513-93-0
N1-(cyanoethyl)-1,3-propanediamine

-
-
56-18-8
bis(3-aminopropyl)amine

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen; nickel In ethanol |
-
-
2664-61-1
3,3',3''-nitrilotripropanamide

-
A
-
56-18-8
bis(3-aminopropyl)amine

-
B
-
4963-47-7
Tris(3-aminopropyl)amine

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride |
-
-
54262-75-8
N-γ-aminopropyltrimethyleneimine

-
-
56-18-8
bis(3-aminopropyl)amine

| Conditions | Yield |
|---|---|
| With ammonia at 120℃; Rate constant; Thermodynamic data; ΔH(excit.), ΔS(excit.);; | |
| With ammonium hydroxide at 50℃; Rate constant; Thermodynamic data; E(a); |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide at 50℃; Rate constant; Thermodynamic data; E(a); |
-
-
504-63-2
trimethyleneglycol

-
A
-
56-18-8
bis(3-aminopropyl)amine

-
B
-
71-23-8
propan-1-ol

-
C
-
109-76-2
Trimethylenediamine

-
D
-
156-87-6
propan-1-ol-3-amine

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen; carbon monoxide at 210℃; under 101258 Torr; Product distribution; Further Variations:; Catalysts; Temperatures; Pressures; amination; |

-
-
7664-41-7
ammonia

-
-
109-64-8
1,3-dibromo-propane

-
A
-
56-18-8
bis(3-aminopropyl)amine

-
B
-
109-76-2
Trimethylenediamine

| Conditions | Yield |
|---|---|
| at 25℃; |


-
-
56-18-8
bis(3-aminopropyl)amine

| Conditions | Yield |
|---|---|
| With ammonia |
-
-
111-94-4
3,3'-Iminodipropionitrile

-
-
7664-41-7
ammonia

-
A
-
56-18-8
bis(3-aminopropyl)amine

-
B
-
4963-47-7
Tris(3-aminopropyl)amine

-
C
-
109-76-2
Trimethylenediamine

| Conditions | Yield |
|---|---|
| at 90 - 125℃; unter Druck; |

-
-
109-76-2
Trimethylenediamine

-
A
-
56-18-8
bis(3-aminopropyl)amine

-
B
-
4605-14-5
N,N'-bis-(3-aminopropyl)-1,3-propanediamine

-
C
-
13274-42-5
N-(3-aminopropyl)-N'-<3-<(3-aminopropyl)amino>propyl>propane-1,3-diamine

| Conditions | Yield |
|---|---|
| With hydrogen at 100 - 200℃; under 0 - 15001.5 Torr; Product distribution / selectivity; |

-
-
167568-21-0
bis-(3-amino-propyl)-carbamic acid tert-butyl ester

-
-
56-18-8
bis(3-aminopropyl)amine

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 20℃; Inert atmosphere; |
-
-
107-15-3
ethylenediamine

-
-
109-76-2
Trimethylenediamine

-
A
-
56-18-8
bis(3-aminopropyl)amine

-
B
-
349636-04-0
tripropylene-tetramine

-
C
-
13531-52-7
N-(2-Aminoethyl)-1,3-propanediamine

| Conditions | Yield |
|---|---|
| With hydrogen; Ni and Re on Al2O3/SiO2 at 130 - 135℃; under 8517.48 Torr; for 6h; Autoclave; |

-
-
146724-95-0
dipropylenetriamine NONOate

-
A
-
56-18-8
bis(3-aminopropyl)amine

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| With perchloric acid In water at 25℃; pH=< 2; Kinetics; pH-value; |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
85-44-9
phthalic anhydride

-
-
102202-87-9
N,N-bis(phthalimidopropyl)amine

| Conditions | Yield |
|---|---|
| In acetic acid for 1h; Heating; | 100% |
| 99% | |
| With acetic acid for 2h; Reflux; | 98% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
383-63-1
ethyl trifluoroacetate,

-
-
252334-20-6
N-[3-(trifluoroacetamido)propyl]-N'-trifluoroacetyl-1,3-diaminopropane

| Conditions | Yield |
|---|---|
| In ethanol for 60h; Acylation; | 100% |
| In acetonitrile at 20℃; for 15h; | 100% |
| In tetrahydrofuran Acylation; Heating; | 99% |
-
-
874306-28-2
(3α,5β)-3,24-bis(3-bromophenoxy)cholane

-
-
56-18-8
bis(3-aminopropyl)amine

| Conditions | Yield |
|---|---|
| With 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane Heating; | 100% |
| With 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate; [Pd(dibenzylideneacetone)2] In 1,4-dioxane for 5h; Heating; | 90 % Spectr. |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
55550-98-6
2,2-dimethyltetrahydro-[1,3]dithiol[4,5-c]furan

-
-
869193-26-0
C20H33N3O6S4

| Conditions | Yield |
|---|---|
| In dichloromethane at 0 - 20℃; for 1h; | 100% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
4375-83-1
tris(dimethylamino)borane

-
-
1730-15-0
1,8,10,9-Triazoboradecalin

| Conditions | Yield |
|---|---|
| 100% | |
| In benzene byproducts: dimethylamine; reflux, 1h; | 75% |
| In benzene byproducts: dimethylamine; reflux, 1h; | 75% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
107-13-1
acrylonitrile

-
-
1155879-59-6
3-[{3-[{3-[bis-(2-cyanoethyl)amino]propyl}-(2-cyanoethyl)amino]propyl}-(2-cyanoethyl)amino]propionitrile

| Conditions | Yield |
|---|---|
| at 20℃; for 120h; Michael condensation; | 100% |
| In water at 20 - 110℃; Inert atmosphere; | 63% |

| Conditions | Yield |
|---|---|
| at 20℃; under 750075 Torr; for 16h; neat (no solvent); | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-methyl-2-(methyltellanyl)propanoic acid With 1-[(1-(cyano-2-ethoxy-2-oxoethylidenaminooxy)dimethylamino-morpholino)]-uronium hexafluorophosphate; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; for 0.5h; Inert atmosphere; Stage #2: bis(3-aminopropyl)amine In N,N-dimethyl-formamide at 20℃; for 3h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-Pyridinedicarboxaldehyde; zinc perchlorate In methanol at 25℃; for 1.5h; Stage #2: bis(3-aminopropyl)amine In methanol at 25℃; for 12h; | 100% |

-
-
1121-60-4
pyridine-2-carbaldehyde

-
-
56-18-8
bis(3-aminopropyl)amine

-
-
64739-67-9
N-pyridin-2-ylmethyl-N'-{3-[(pyridin-2-ylmethyl)amino]propyl}propane-1,3-diamine

| Conditions | Yield |
|---|---|
| Stage #1: pyridine-2-carbaldehyde; bis(3-aminopropyl)amine With acetic acid; zinc In methanol at 70 - 80℃; for 4.5h; Stage #2: With hydrogenchloride In methanol at 20℃; Stage #3: With sodium hydroxide In water | 99% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
1755-15-3
2-acetyldimedone

-
-
674297-98-4
N1,N9-bis-1-(4,4-dimethyl-2,6-dioxocyclohexylidene)ethyl-norspermidine

| Conditions | Yield |
|---|---|
| In ethanol for 2h; Heating; | 99% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
580-13-2
2-bromonaphthalene

-
-
1351684-37-1
N1-(3-aminopropyl)-N3-(naphthalen-2-yl)propane-1,3-diamine

| Conditions | Yield |
|---|---|
| With copper(l) iodide; caesium carbonate; rac-Pro-OH In acetonitrile for 24h; Ullmann reaction; Inert atmosphere; Reflux; regioselective reaction; | 99% |
| With copper(l) iodide; caesium carbonate; L-proline In acetonitrile Inert atmosphere; Reflux; | 90% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
255367-21-6
1,2-bis(4-tert-butyl-2,6-diformylphenylsulfanyl)ethane

-
-
1417170-21-8
C38H64N6S2

| Conditions | Yield |
|---|---|
| Stage #1: bis(3-aminopropyl)amine; 1,2-bis(4-tert-butyl-2,6-diformylphenylsulfanyl)ethane In ethanol; dichloromethane at 0 - 20℃; for 23h; Stage #2: With sodium tetrahydroborate In ethanol at 20℃; for 4h; | 99% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
193215-78-0
tetradecane-1,14-diol dimesylate

| Conditions | Yield |
|---|---|
| In acetonitrile for 4h; Heating; | 98% |
-
-
770738-07-3
1-(benzyloxy)-6-(2-thioxothiazolidine-3-carbonyl)pyridin-2(1H)-one

-
-
56-18-8
bis(3-aminopropyl)amine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; | 98% |
| With triethylamine In dichloromethane at 20℃; for 6h; | 98% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
501-53-1
benzyl chloroformate

-
-
183249-68-5
[3-(3-benzyloxycarbonylaminopropylamino)propyl]carbamic acid benzyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 10℃; Temperature; Reagent/catalyst; Solvent; | 97.1% |

| Conditions | Yield |
|---|---|
| In water; acetonitrile Heating; | 97% |
| In water; acetonitrile for 4h; Reflux; | 97% |
| 93% | |
| With water In acetonitrile for 3h; Heating / reflux; |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
35980-64-4
1,5,9-Tritosyl-1,5,9-triazanonane

| Conditions | Yield |
|---|---|
| With sodium hydroxide In diethyl ether; water Heating / reflux; | 96.5% |
| With potassium carbonate In water at 60℃; for 3h; | 95% |
| With sodium hydroxide In dichloromethane at 20℃; for 3h; | 90% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
630-19-3
pivalaldehyde

-
-
530-62-1
1,1'-carbonyldiimidazole

-
-
82409-02-7
bis[3-(t-butoxycarbonylamino)propyl]amine

| Conditions | Yield |
|---|---|
| Stage #1: pivalaldehyde; 1,1'-carbonyldiimidazole With potassium hydroxide In toluene at 60℃; for 3h; Stage #2: bis(3-aminopropyl)amine In toluene at 60℃; for 3h; Further stages.; | 96% |

-
-
56-18-8
bis(3-aminopropyl)amine

-
-
60469-45-6
1,7-di-(5-norbornen-2-yl)-4-heptanone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium borohydrid In water; isopropyl alcohol; toluene | 96% |
| With hydrogenchloride; sodium borohydrid In water; isopropyl alcohol; toluene | 96% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
13939-06-5, 199620-15-0
molybdenum hexacarbonyl

-
-
161334-70-9
fac-[(H2N(CH2)3NH(CH2)3NH2)Mo(CO)3]

| Conditions | Yield |
|---|---|
| In dibutyl ether N2-atmosphere; slight excess metal carbonyl, refluxing for 2 h (returning sublimed hexacarbonyl into reaction vessel); cooling to room temp., collection of ppt., washing (hexane), drying (vac., 50°C); | 96% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
15930-60-6
N,N-diethyl-3-iodobenzamide

-
-
1351684-33-7
3,3'-[3,3'-azanediyl-bis(propane-3,1-diyl)bis(azanediyl)]bis(N,N-diethylbenzamide)

| Conditions | Yield |
|---|---|
| With copper(l) iodide; caesium carbonate; rac-Pro-OH In propiononitrile for 24h; Ullmann reaction; Inert atmosphere; Reflux; regioselective reaction; | 96% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 5h; | 95% |
| In tetrahydrofuran at -5℃; for 24h; | 70% |

| Conditions | Yield |
|---|---|
| With sodium t-butanolate; 1,1'-bis-(diphenylphosphino)ferrocene; tris(dibenzylideneacetone)dipalladium (0) In 1,4-dioxane Substitution; Amination; Heating; | 95% |
| With 1,3-bis(diphenylphosphino)ferrocene; bis(dibenzylideneacetone)-palladium(0); sodium t-butanolate In 1,4-dioxane for 30h; Heating; | 95% |
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium t-butanolate In 1,4-dioxane for 20h; Heating; | 64% |

| Conditions | Yield |
|---|---|
| In toluene at 20℃; for 1h; | 95% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
15555-49-4, 131485-81-9, 75918-15-9
(di(3-aminopropyl)amine)trichloro cobalt(III)

| Conditions | Yield |
|---|---|
| In isopropyl alcohol refluxing a mixt. of the Co-compd. and the amine for 0.5 h; pptn. after cooling, filtering off, washing with EtOH and acetone, air-drying; | 95% |
-
-
56-18-8
bis(3-aminopropyl)amine

| Conditions | Yield |
|---|---|
| In water Cu(ClO4)2*6H2O was dissolved in water, ligand was added with stirring, soln. of NaN3 in water was added, stirred for 0.5 h; crystd. for 1 w; elem. anal.; | 95% |

-
-
56-18-8
bis(3-aminopropyl)amine

-
-
143-66-8
sodium tetraphenyl borate

-
-
90-02-8
salicylaldehyde

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| With C5H5N In methanol salicylaldehyde and 3,3'-diaminodipropylamine mixed in MeOH, Fe salt in MeOH added, C5H5N added, stirred for 1 h, Na salt added; pptd. for hours, filtered, washed (Et2O), dried at room temp., recrystd.(MeCN); elem. anal.; | 95% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 23℃; for 2h; | 95% |
-
-
56-18-8
bis(3-aminopropyl)amine

-
-
49761-82-2
tert-butyl 1H-imidazole-1-carboxylate

-
-
82409-02-7
bis[3-(t-butoxycarbonylamino)propyl]amine

| Conditions | Yield |
|---|---|
| In toluene at 60℃; for 18h; | 95% |
| In tetrahydrofuran at 20℃; Reflux; | 80% |
| at 60℃; for 4h; | 66% |
Dipropylenetriamine Chemical Properties
Molecular Formula: C6H17N3
Molar mass: 131.22 g/mol
EINECS: 200-261-2
Density: 0.912 g/cm3
Flash Point: 118.3 °C
Index of Refraction: 1.475
Boiling Point: 242.1 °C at 760 mmHg
Vapour Pressure: 0.0346 mmHg at 25°C
Appearance: Colourless to yellow liquid
Stable:Air and light sensitive. Combustible. Incompatible with oxidizing agents
Structure of Aminobis(propylamine) (56-18-8):
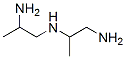
XLogP3-AA: -1.4
H-Bond Donor: 3
H-Bond Acceptor: 3
Systematic Name: N-(3-aminopropyl)propane-1,3-diamine
SMILES: NCCCNCCCN
InChI: InChI=1/C6H17N3/c7-3-1-5-9-6-2-4-8/h9H,1-8H2
InChIKey: OTBHHUPVCYLGQO-UHFFFAOYAG
Std. InChI: InChI=1S/C6H17N3/c7-3-1-5-9-6-2-4-8/h9H,1-8H2
Std. InChIKey: OTBHHUPVCYLGQO-UHFFFAOYSA-N
Dipropylenetriamine Uses
Aminobis(propylamine) (56-18-8) can be used to make soaps, dyes, and pharmaceuticals.
Dipropylenetriamine Toxicity Data With Reference
| 1. | skn-rbt 470 mg open MOD | UCDS** Union Carbide Data Sheet. (Industrial Medicine and Toxicology Dept., Union Carbide Corp., 270 Park Ave., New York, NY 10017) 6/13/68 . | ||
| 2. | eye-rbt 47 mg SEV | UCDS** Union Carbide Data Sheet. (Industrial Medicine and Toxicology Dept., Union Carbide Corp., 270 Park Ave., New York, NY 10017) 6/13/68 . | ||
| 3. | orl-rat LD50:738 mg/kg | ZHYGAM Zeitschrift fuer die Gesamte Hygiene und Ihre Grenzgebiete. 20 (1974),393. | ||
| 4. | orl-mus LD50:435 mg/kg | ZHYGAM Zeitschrift fuer die Gesamte Hygiene und Ihre Grenzgebiete. 20 (1974),393. | ||
| 5. | orl-rbt LD50:210 mg/kg | ZHYGAM Zeitschrift fuer die Gesamte Hygiene und Ihre Grenzgebiete. 20 (1974),393. | ||
| 6. | skn-rbt LDLo:110 mg/kg | AIHAAP American Industrial Hygiene Association Journal. 23 (1962),95. |
Carcinogenicity of Aminobis(propylamine) (56-18-8) hasn't been listed as a carcinogen by NTP, IARC,ACGIH, or CA Prop 65.You can see actual entry in RTECS for complete information.
Dipropylenetriamine Consensus Reports
Reported in EPA TSCA Inventory.
Dipropylenetriamine Safety Profile
Poison by skin contact. Moderately toxic by ingestion. A skin and severe eye irritant. When heated to decomposition it emits toxic fumes of NOx. An explosive.
Dipropylenetriamine Standards and Recommendations
DOT Classification: 8; Label: Corrosive
Dipropylenetriamine Specification
Aminobis(propylamine) (56-18-8) also can be called 3,3'-Iminobispropylamine ; Dipropylenetriamine ; N,N-di(3-aminopropyl)amine ; N-(3-Aminopropyl)-1,3-propanediamine ; 1,3-Propanediamine, N- (3-aminopropyl)- ; 3,3'-azanediyldi(propanamine) ; N-(3-aminopropyl)propane-1,3-diamine ; Bis(3-aminopropyl)amine and Norspermidine .It is hazardous,so the first aid measures and others should be known.Such as: When on the skin: first,should flush skin with plenty of water immediatelyfor at least 15 minutes while removing contaminated clothing. Secondly,Get shoesmedical aid . Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids.Then get medical aid soon.While ,it's Inhaled: Remove from exposure and move to fresh air immediately.Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product : Wash mouth out with water,and get medical aid immediately.Notes to physician: Treat supportively and symptomatically.
In addition, Aminobis(propylamine) (56-18-8) absorbs moisture or water from the air, and can be stable under normal temperature and pressure conditions.It is not compatible with strong oxidizing agents, acids, acid chlorides, acid anhydrides, chloroformates, and you must not take it with incompatible materials and not be exposured to moist air or water.And also prevent it to broken down into hazardous decomposition products: oxides of nitrogen, carbon dioxide, carbon monoxide.
Related Products
- Dipropylenetriamine
- 5618-84-8
- 56189-09-4
- 5618-96-2
- 56190-17-1
- 5619-04-5
- 5619-05-6
- 5619-07-8
- 5619-09-0
- 56191-23-2
- 56205-88-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View