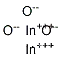1312-43-2 Usage
Description
Indium oxide, also known as indium sesquioxide (In2O3), is a light-yellow powder with a cubic crystal structure. It occurs in both amorphous and crystalline forms, with the amorphous form converting to crystalline upon heating at higher temperatures. Indium oxide is a stable, ceramic-like material that is insoluble in water and isomorphous with hematite (Fe2O3). It has a density of 7.18 g/cm3 and melts around 2,000°C. The amorphous form dissolves readily in mineral acids, while the crystalline form has low solubility in acids. As an n-type semiconductor, indium oxide is used as a resistive element in integrated circuits and can form heterojunctions with various semiconductor materials.
Uses
Used in Electronics Industry:
Indium oxide is used as a n-type semiconductor for resistive elements in integrated circuits. It is also utilized in the production of thin film coatings for optical, antistatic, and infrared reflectors. When doped with tin oxide, it is used as a transparent conductive coating.
Used in Glass Manufacturing:
Indium oxide is used in the glass industry as a color additive and in the production of specialty glass.
Used in Batteries:
Indium trioxide is employed in alkaline batteries to suppress gas formation, enhancing the battery's performance and longevity.
Used in High Current Electrical Switches and Contacts:
Indium oxide serves as an anti-arcing additive in high current electrical switches and contacts, improving their efficiency and reliability.
Used in Coatings:
Indium oxide is also used in the formulation of coatings for various applications, such as In2O3/Au/Ag coated PET film, which has chemical properties of a light green powder.
Preparation
Indium trioxide may be obtained by heating indium in air or oxygen:
4In + 3O2 → 2In2O3
or by calcination of indium hydroxide, nitrate, or carbonate at elevated tem-peratures:
2In(OH)3 → In2O3 + 3H2O
In2(CO3)3 →In2O3 + 3CO2
Hazard
See indium.
Purification Methods
Wash it with H2O and dry it below 850o. It volatilises at 850o and dissolves in hot mineral acids to form salts. Store it away from light because it darkens due to the formation of free In.
Check Digit Verification of cas no
The CAS Registry Mumber 1312-43-2 includes 7 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 4 digits, 1,3,1 and 2 respectively; the second part has 2 digits, 4 and 3 respectively.
Calculate Digit Verification of CAS Registry Number 1312-43:
(6*1)+(5*3)+(4*1)+(3*2)+(2*4)+(1*3)=42
42 % 10 = 2
So 1312-43-2 is a valid CAS Registry Number.
InChI:InChI=1/2In.3O/rIn2O3/c3-1-5-2-4