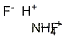1341-49-7 Usage
Description
Ammonium hydrogen difluoride, also known as ammonium bifluoride, is a white crystalline solid that can be dissolved in water. It is corrosive to metals and tissue and is commonly used in various industries due to its unique properties.
Uses
Used in Chemical Analysis and Antimicrobial Applications:
Ammonium hydrogen difluoride is used as a chemical reagent in analysis and serves as an antiseptic. It is effective in sterilizing dairy and other food equipment, ensuring cleanliness and safety in food processing.
Used in Ceramics and Electroplating Industries:
Ammonium hydrogen difluoride is utilized in the ceramics industry and as an etchant in electroplating processes. Its ability to etch glass and its corrosive nature make it a valuable component in these applications.
Used in Glass and Porcelain Industries:
In the glass and porcelain industries, ammonium hydrogen difluoride acts as a mordant for aluminum, enhancing the adhesion of dyes and pigments to these materials.
Used in Metal Manufacturing:
Ammonium hydrogen difluoride is employed in the manufacture of magnesium (Mg) and magnesium alloys, as well as in the brightening of aluminum. Its role in these processes contributes to the production of high-quality metal products.
Used in Food Processing Equipment Disinfection:
Ammonium hydrogen difluorid serves as a disinfectant for various parts of beer-dispensing apparatus, tubes, and other food processing equipment, ensuring the cleanliness and safety of the equipment.
Used in Laundering Applications:
In laundering, ammonium hydrogen difluoride acts as a "sour" agent, helping to remove stains and dirt from fabrics during the washing process.
Used in the Production of Hydrofluoric Acid:
Ammonium hydrogen difluoride is used as an intermediate in the production of hydrofluoric acid from hexafluorosilicic acid, which is an important industrial chemical with various applications.
Physical Properties:
Ammonium hydrogen difluoride forms orthorhombic or tetragonal crystals and is deliquescent. It has a density of 1.50 g/cm3, a refractive index of 1.390, and melts at 125.6°C. Ammonium hydrogen difluoride is very soluble in water and slightly soluble in alcohol.
Air & Water Reactions
Dissolves in water and forms a weak solution of hydrofluoric acid.
Reactivity Profile
AMMONIUM BIFLUORIDE reacts violently with bases. In presence of moisture will corrode glass, cement, and most metals. Flammable hydrogen gas may collect in enclosed spaces. Do not use steel, nickel, or aluminum containers [USCG, 1999].
Hazard
Corrosive to skin.
Health Hazard
TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated.
Flammability and Explosibility
Nonflammable
Safety Profile
Caustic poison and strong irritant by all routes. See also HYDROFLUORIC ACID. When heated to decomposition it emits very toxic fumes of F-, NO,, and NH3.
Potential Exposure
It is used as a sterilizer, in dairy and
brewery operations; in the ceramic, glass, and electroplating
industries; as a laundry sour.
Shipping
Administer oxygen if breathing is difficult. Remove and isolate
contaminated clothing and shoes. In case of contact with
substance, immediately flush skin or eyes with running
water for at least 20 minutes. For minor skin contact, avoid
spreading material on unaffected skin. Keep victim warm
and quiet. Effects of exposure (inhalation, ingestion or skin
contact) to substance may be delayed. Ensure that medical
personnel are aware of the material(s) involved and take precautions
to protect themselves. Medical observation is
recommended for 24 to 48 hours after breathing overexposure,
as pulmonary edema may be delayed. As first aid for
pulmonary edema, a doctor or authorized paramedic may
consider administering a drug or other inhalation therapy.
Incompatibilities
Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explosions.
Keep away from alkaline materials, strong bases,
strong acids, oxoacids, epoxides. In the presence of moisture
corrodes concrete, metals, glass.
Waste Disposal
May be buried in a specially
designated chemical landfill. Aqueous wastes may be reacted with an excess of lime followed by lagooning and
either recovery or land disposal of the separated calcium
fluoride.
Check Digit Verification of cas no
The CAS Registry Mumber 1341-49-7 includes 7 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 4 digits, 1,3,4 and 1 respectively; the second part has 2 digits, 4 and 9 respectively.
Calculate Digit Verification of CAS Registry Number 1341-49:
(6*1)+(5*3)+(4*4)+(3*1)+(2*4)+(1*9)=57
57 % 10 = 7
So 1341-49-7 is a valid CAS Registry Number.
InChI:InChI=1/FH.H3N/h1H;1H3/p+1