Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem’s R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantiti
Lonwin Chemical Group Limited
uick Details Place of Origin: Shanghai, China Brand Name: Lonwin Model Number: 148-24-3 Type: Agrochemical Intermediates,
Cas:148-24-3
Min.Order:1000 Kilogram
Negotiable
Type:Other
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Ality Chemical Corporation
8-Hydroxyquinoline Basic information Product Name: 8-Hydroxyquinoline Synonyms: 8-hydroxy-quinolin;8-OQ;8-Oxyquinolin;8-Quinol;8-Quinolol;Fennosan;Fennosan H 30;fennosanh30 CAS: 148-24-3 MF: C9H7NO MW: 145.16 EINECS: 205-711-1 Product Categori
Cas:148-24-3
Min.Order:1 Gram
Negotiable
Type:Other
inquiryAntimex Chemical Limied
ProName: 8-Hydroxyquinoline CasNo: 148-24-3 Molecular Formula: C9H7NO Appearance: White or light yellow crystalline pow... Application: It is an important raw material and in... DeliveryTime: prompt PackAge: according to
Cas:148-24-3
Min.Order:1 Metric Ton
FOB Price: $5.2
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:148-24-3
Min.Order:0 Metric Ton
FOB Price: $1.0 / 2.0
Type:Lab/Research institutions
inquiryGolden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Hangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance Off white to light brown crystal powder Light yellow crystal powder Assay
Cas:148-24-3
Min.Order:1 Kilogram
FOB Price: $15.0 / 25.0
Type:Manufacturers
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:148-24-3
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryJinan Finer Chemical Co., Ltd
Product Description Product Name 8-Hydroxyquinoline CAS No. 148-24-3 Appearance White to yellowish powder Assay ≥99.5% Capacity 500mt/year Min.packing 100gra
Cas:148-24-3
Min.Order:1 Gram
FOB Price: $12.0
Type:Lab/Research institutions
inquiryHebei yanxi chemical co.,LTD.
Hebei yanxi chemical co., LTD is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low carb
Cas:148-24-3
Min.Order:1 Kilogram
FOB Price: $1.0 / 3.0
Type:Manufacturers
inquiryWuhan Fortuna Chemical Co.,Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; ...... Appearance:Almost white to light brown crystalline powder Storage:Store
Cas:148-24-3
Min.Order:25 Kilogram
FOB Price: $15.0 / 25.0
Type:Trading Company
inquiryXi'an Quanao Biotech Co., Ltd.
Xi’an Quanao Biotech Co., Ltd. with 18 years experience in plant extract filed. Product grade: Medicine, Health care product, Cosmetics, Food, Beverage, Feed. Hot selling market: Europe, North America, South America, Middle East, Asia Pacifi
Cas:148-24-3
Min.Order:1 Kilogram
FOB Price: $124.0 / 143.0
Type:Manufacturers
inquiryHefei TNJ chemical industry co.,ltd
CAS No.: 148-24-3 Other Names: 8-Hydroxyquinoline MF: C9H7NO EINECS No.: 205-711-1 Place of Origin: Anhui, China (Mainland) Type: Dyestuff Intermediates, Pharmaceutical Intermediates Purity: 99.5%min Brand Name: TNJ
Cas:148-24-3
Min.Order:1 Metric Ton
FOB Price: $20000.0
Type:Trading Company
inquiryHangzhou Think Chemical Co. Ltd
8-Hydroxyquinoline CAS No.:148-24-3 Name 8-Hydroxyquinoline Synonyms 8-Quinolinol; Quinolin-8-ol; Oxine Molecular Structure Molecular Formula C9H7NO Molecular Weight 145.16 CAS Registry Number 148-24
Cas:148-24-3
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Other
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:148-24-3
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryEnke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new ap
Nanjing Yuance Industry&Trade Co., Ltd.
Grade: 99% Excellent quality with competitive price Fast delivery Appearance:White to pale yellow crystalline powder Storage:Store in cool dry pladce Package:25 kg/bag Application:used as complexing agent in electroplating baths, to complex met
Cas:148-24-3
Min.Order:1 Metric Ton
Negotiable
Type:Trading Company
inquiryANQING CHICO PHARMACEUTICAL CO.,LTD.
High quality. Factory supply. In stock. Best price.1.Quick response within 24 hours;2.Best quality in your requirement;?3.We pay more attention on delivery time, and usually ship on time;4.Under the premise of safety and effectiveness, we can produce
Shanghai Seasonsgreen Chemical Co.,Ltd
Shanghai Seasonsgreen Chemical is a high-tech research and development, production, sale and custom synthesis set in one high-tech chemical products enterprises. Our sales and marketing division is located in Shanghai, serving international pharmaceu
Cas:148-24-3
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryHubei DiBo chemical co., LTD
Name::8-Hydroxyquinoline The alias:8-hydroxy-quinolin;8-OQ;8-Oxyquinolin;8-Quinol;8-Quinolol;Fennosan;Fennosan H 30;fennosanh30 CAS NO:148-24-3 Molecular formula:C9H7NO Molecular weight:145.16 Product Quality 12 years of chemical raw materi
Cas:148-24-3
Min.Order:25 Kilogram
FOB Price: $1.0 / 2.0
Type:Other
inquiryHenan Tianfu Chemical Co., Ltd.
TIANFUCHEM--148-24-3--8-Hydroxyquinoline in stock Our company was built in 2009 with an ISO certificate.In the past 10 years, we have grown up as a famous fine chemicals supplier in China And we had established stable business relations
Cas:148-24-3
Min.Order:1 Metric Ton
FOB Price: $2000.0
Type:Lab/Research institutions
inquiryLeader Biochemical Group
About Product Details Mol File: 148-24-3.mol
Cas:148-24-3
Min.Order:1 Kilogram
FOB Price: $3.0 / 10.0
Type:Lab/Research institutions
inquiryLIDE PHARMACEUTICALS LIMITED
LIDE PHARMACEUTICALS LIMITED is a professional chemicals and APIs leading manufacturer in China. Our core business line covers APIs, Intermediates, Herb extract, etc. We are specialized in chemical synthesis, process development of pharmaceu
Cas:148-24-3
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:148-24-3
Min.Order:1 Gram
FOB Price: $12.0 / 15.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:148-24-3
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Molecular formula: C9H7NO Molecular weight: 145.16 CAS NO.: 148-24-3 Appearance:
Cas:148-24-3
Min.Order:5 Metric Ton
FOB Price: $1.0 / 2.0
Type:Other
inquirySynthetic route

| Conditions | Yield |
|---|---|
| With RhCl(PPh3)3 In benzene at 40℃; for 1h; Product distribution; primer deut. isotop eff.; | A 100% B n/a |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In decane at 150℃; for 36h; Inert atmosphere; Schlenk technique; | 98% |
| With iron oxide surrounded by nitrogen doped graphene shell immobilized on carbon support In acetonitrile at 100℃; under 11251.1 Torr; for 24h; Autoclave; | 93% |
| With perylene diimide covalent immobilized to SiO2 nanospheres; air In N,N-dimethyl acetamide at 20℃; UV-irradiation; | 93% |
-
-
52968-25-9
Bis-<8-chinolinyl>-carbonat

-
-
81494-56-6
2-Amino-1,1,3,3-tetramethoxypropan

-
A
-
148-24-3
8-quinolinol

| Conditions | Yield |
|---|---|
| In chloroform 1.) r. t., 12 h, 2.) reflux, 10 h; | A 97% B 74% |


| Conditions | Yield |
|---|---|
| Stage #1: glycerol With sulfuric acid; copper(II) sulfate; calcium oxide for 1h; Reflux; Stage #2: 2-amino-phenol for 0.333333h; Skraup Quinoline Synthesis; Stage #3: With 2-hydroxynitrobenzene at 130 - 140℃; Concentration; | 96.31% |
| Stage #1: 2-amino-phenol With sulfuric acid; 2-hydroxynitrobenzene at 70℃; for 0.5h; Stage #2: glycerol at 150℃; for 4h; Temperature; | 91% |
| With sulfuric acid In water at 36 - 200℃; for 0.25h; Microwave irradiation; Sealed tube; Green chemistry; | 34% |
| With sulfuric acid; iodine | |
| Stage #1: 2-amino-phenol; glycerol With sulfuric acid Stage #2: With nitrobenzene |

| Conditions | Yield |
|---|---|
| Stage #1: anthranilic acid; acrolein With hydrogenchloride; acetic acid; hydroquinone at 95 - 100℃; for 3h; Stage #2: With chloropyridinecobaloxime(III); Eosin Y In water; acetonitrile at 20℃; for 3h; Microwave irradiation; | 96.2% |

| Conditions | Yield |
|---|---|
| With formic acid at 95 - 100℃; for 2h; Concentration; Molecular sieve; | 95.6% |


| Conditions | Yield |
|---|---|
| With silica gel at 110℃; for 0.25h; isomerization; | A 5% B 95% |

-
-
148-24-3
8-quinolinol

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In neat (no solvent, solid phase) at 20℃; for 0.583333h; Green chemistry; | 94% |

| Conditions | Yield |
|---|---|
| With nickel(III) oxide; sulfuric acid; acetic acid at 90℃; for 5h; Temperature; | 92.7% |
| With sulfuric acid; acetic acid |

-
-
108530-08-1
quinolin-8-yl trifluoromethanesulfonate

-
-
148-24-3
8-quinolinol

| Conditions | Yield |
|---|---|
| With caesium carbonate In toluene at 80℃; for 15h; | 92% |

| Conditions | Yield |
|---|---|
| With bis(triphenyl)oxodiphosphonium trifluoromethanesulfonate salt; potassium iodide In ethanol at 20℃; for 1.16667h; | 90% |
| With lithium tetrafluoroborate In water; acetonitrile at 20℃; for 4h; Electrochemical reaction; Inert atmosphere; | 24 mg |
-
-
222713-64-6
8-((tert-butyldimethylsilyl)oxy)quinoline

-
-
148-24-3
8-quinolinol

| Conditions | Yield |
|---|---|
| With zinc tetrafluoroborate In water for 13h; Ambient temperature; | 89% |
| With potassium hydrogen difluoride In methanol at 20℃; for 2h; | 89% |

| Conditions | Yield |
|---|---|
| With piperidine; carbon monoxide; palladium diacetate; triethylamine; triphenylphosphine In N,N-dimethyl-formamide at 50℃; under 52505.3 Torr; for 24h; | 83% |

| Conditions | Yield |
|---|---|
| With water; poly(ethyleneimine) at 40℃; for 0.5h; | 82% |

-
-
148-24-3
8-quinolinol

| Conditions | Yield |
|---|---|
| With ammonia In water at 20℃; for 24h; | 80.6% |

| Conditions | Yield |
|---|---|
| With tetrachloromethane; iron(III) chloride hexahydrate at 150℃; for 8h; Inert atmosphere; Sealed tube; | 79% |
| With 2,4,6-trimethyl-pyridine; oxygen; palladium diacetate; trifluoroacetic acid at 150℃; for 16h; Schlenk technique; | 55% |

| Conditions | Yield |
|---|---|
| With i-Amyl alcohol; ozone In water at -0.1℃; | 78% |
| With dihydrogen peroxide; bis(triphenyl)oxodiphosphonium trifluoromethanesulfonate salt In ethanol; water at 20℃; for 1h; regioselective reaction; | 55% |
| findet sich im Harn von Kaninchen nach peroraler Verabreichung; |

| Conditions | Yield |
|---|---|
| With lithium salt of proline; tetrabutylammomium bromide; potassium hydroxide; copper dichloride In water at 120℃; for 0.666667h; Microwave irradiation; Green chemistry; | 78% |
-
-
16567-18-3
8-bromoquinoline

-
-
148-24-3
8-quinolinol

| Conditions | Yield |
|---|---|
| With 5-(di-tert-butylphosphino)-1′, 3′, 5′-triphenyl-1′H-[1,4′]bipyrazole; tris-(dibenzylideneacetone)dipalladium(0); cesiumhydroxide monohydrate In 1,4-dioxane at 110℃; Inert atmosphere; Glovebox; Sealed tube; | 74% |
-
-
23504-14-5
2-(prop-2-yn-1-ylamino)phenol

-
A
-
148-24-3
8-quinolinol

-
B
-
343270-95-1
2-methylidene-3,4-dihydro-2H-1,4-benzoxazine

| Conditions | Yield |
|---|---|
| With potassium carbonate; gold(I) chloride In acetonitrile for 8h; Reflux; | A 8% B 72% |
-
-
6640-50-2
1,2,3,4-tetrahydroquinolin-8-ol

-
-
98-95-3
nitrobenzene

-
A
-
148-24-3
8-quinolinol

-
B
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| With nickel-nitrogen-doped carbon framework In water at 145℃; for 18h; Inert atmosphere; Sealed tube; Green chemistry; | A 58% B 62% |

-
-
6640-50-2
1,2,3,4-tetrahydroquinolin-8-ol

-
-
20651-75-6
1-butyl-4-nitro-benzene

-
A
-
148-24-3
8-quinolinol

-
B
-
104-13-2
4-Butylaniline

| Conditions | Yield |
|---|---|
| With nickel-nitrogen-doped carbon framework In water at 145℃; for 18h; Inert atmosphere; Sealed tube; Green chemistry; | A 62% B 61% |


| Conditions | Yield |
|---|---|
| With oxygen; N-ethyl-N,N-diisopropylamine; [5,6]fullerene-C70 In chloroform; toluene at 20℃; for 12h; Irradiation; | 61% |
-
-
83-73-8
5,7-diiodo-8-hydroxyquinoline

-
A
-
148-24-3
8-quinolinol

-
B
-
13207-63-1
5-iodoquinolin-8-ol

-
C
-
7385-89-9
7-iodo-8-hydroxyquinoline

| Conditions | Yield |
|---|---|
| With carbon monoxide; glycine ethyl ester hydrochloride; palladium diacetate; triethylamine; triphenylphosphine In N,N-dimethyl-formamide at 50℃; under 52505.3 Torr; for 24h; | A 51% B n/a C n/a |

-
-
6640-50-2
1,2,3,4-tetrahydroquinolin-8-ol

-
-
7369-50-8
3-ethylnitrobenzene

-
A
-
148-24-3
8-quinolinol

-
B
-
587-02-0
m-ethylaniline

| Conditions | Yield |
|---|---|
| With nickel-nitrogen-doped carbon framework In water at 145℃; for 18h; Inert atmosphere; Sealed tube; Green chemistry; | A 50% B 51% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 111℃; for 24h; Doebner-von Miller Reaction; | 41% |

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; silver trifluoromethanesulfonate In toluene at 80℃; for 4h; | 40% |
-
-
1245937-15-8
C19H15NO3

-
-
148-24-3
8-quinolinol

| Conditions | Yield |
|---|---|
| at 650℃; Flash vacuum pyrolysis; | 34% |
-
-
91-22-5
quinoline

-
A
-
580-18-7
3-hydroxyquinoline

-
B
-
148-24-3
8-quinolinol

-
C
-
118-92-3
anthranilic acid

-
D
-
112259-26-4
(5R,6S)-cis-5,6-dihydroxy-5,6-dihydroquinoline

| Conditions | Yield |
|---|---|
| With Pseudomonas putida | A 13% B 27% C 27% D 33% |
| biotransformation by Pseudomonas putida UV4; Further byproducts given; | A 11 mg B 23 mg C 22 mg D 32% |
| biotransformation by Pseudomonas putida UV4; Further byproducts given; | A 11 mg B 23 mg C 22 mg D 32 mg |


| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 24h; Heating; | 100% |
| With sodium hydroxide; tetrabutylammomium bromide In dichloromethane; water at 25℃; for 4h; | 84% |
| With potassium carbonate In ethanol at 70℃; for 10h; regioselective reaction; | 84% |
-
-
148-24-3
8-quinolinol

-
-
121-57-3
4-aminobenzene sulfonic acid

-
-
574-70-9
(E)-4-((8-hydroxyquinolin-5-yl)diazenyl)benzenesulfonic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-aminobenzene sulfonic acid With hydrogenchloride; sodium nitrite In water at -5℃; for 0.25h; Stage #2: 8-quinolinol With sodium hydroxide In water | 100% |
| With hydrogenchloride; sodium nitrite | |
| With hydrogenchloride; sodium hydroxide; ethylenediaminetetraacetic acid; sodium nitrite In ethanol for 0.0833333h; |

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane | 100% |
| With dihydrogen peroxide In water at 20℃; for 4h; Catalytic behavior; | 98% |
| With 3-chloro-benzenecarboperoxoic acid In Isopropyl acetate at 10 - 35℃; for 5.83333h; Green chemistry; | 98.2% |

| Conditions | Yield |
|---|---|
| With oleum at 20℃; | 100% |
| With sulfuric acid | |
| With sulfuric acid at 180℃; im Rohr; | |
| With sulfuric acid at 0 - 5℃; for 30h; |
-
-
148-24-3
8-quinolinol

-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
108530-08-1
quinolin-8-yl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; for 12h; Schlenk technique; Inert atmosphere; | 100% |
| With pyridine In dichloromethane at 0 - 20℃; | 99% |
| With pyridine In dichloromethane at 0 - 20℃; | 97% |
-
-
148-24-3
8-quinolinol

-
-
85116-37-6
(-)-diisopinocamphenylborane chloride

| Conditions | Yield |
|---|---|
| In diethyl ether | 100% |
-
-
148-24-3
8-quinolinol

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; antimony pentafluoride at 25℃; | 100% |
| With trifluorormethanesulfonic acid; cyclohexane; antimony pentafluoride at 20℃; |
-
-
148-24-3
8-quinolinol

-
-
10025-73-7
chromium(III) chloride

| Conditions | Yield |
|---|---|
| With ammonia In water prepn. from aq. CrCl3 soln. and a soln. of 8-hydroxy-quinoline (in CH3CO2H, neutralized by dropwise addn. of NH4OH (slight pptn.), cleared by heating, satd. with CO2 after cooling, adjusted to pH 4-8 with aq. NH3, heated for 1.5 h with stirring;; pptn., filtering hot, washing with hot H2O and drying at 110 °C;; | 100% |
| With NH3 In water prepn. from aq. CrCl3 soln. and a soln. of 8-hydroxy-quinoline (in CH3CO2H, neutralized by dropwise addn. of NH4OH (slight pptn.), cleared by heating, satd. with CO2 after cooling, adjusted to pH 4-8 with aq. NH3, heated for 1.5 h with stirring;; pptn., filtering hot, washing with hot H2O and drying at 110 °C;; | 100% |

| Conditions | Yield |
|---|---|
| In diethyl ether To a suspn. of complex in ether was added stoichiometric amount of ligand, stirred for 1-2 h at room temp.;; recrystd. from CH2Cl2-n-hexane; elem. anal.;; | 100% |
-
-
148-24-3
8-quinolinol

| Conditions | Yield |
|---|---|
| With triethylamine In diethyl ether; benzene byproducts: (Et3NH)Cl; N2 atmosphere, addn. of Et3N and Et2O soln. of quinolinol compound to benzene soln. of Mo complex at room temp., stirring (18 h); removement of volatiles (vacuum), extn. (pentane), centrifugation, concn. of pentane layers; elem. anal.; | 100% |
-
-
148-24-3
8-quinolinol

-
-
5893-61-8, 14690-98-3, 15305-21-2, 16842-73-2, 22992-79-6, 34729-06-1, 133386-02-4, 133386-04-6
copper(II) formate tetrahydrate

-
-
13014-03-4
copper 8-hydroxyquinolinate

| Conditions | Yield |
|---|---|
| at 80 - 100℃; under 0.000750075 Torr; for 1h; Time; Large scale; | 100% |
-
-
148-24-3
8-quinolinol

-
-
150-13-0
4-amino-benzoic acid

-
-
67940-27-6
(E)-4-((8-hydroxyquinolin-5-yl)diazenyl)benzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-amino-benzoic acid With hydrogenchloride; sodium nitrite In water at -5℃; for 0.25h; Stage #2: 8-quinolinol With sodium hydroxide In water | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 8-quinolinol With sodium hydride In tetrahydrofuran Inert atmosphere; Stage #2: di-tert-butyl-diazodicarboxylate In tetrahydrofuran at -5 - 60℃; for 1.5h; Reagent/catalyst; Solvent; Time; Temperature; Inert atmosphere; | 99.9% |

| Conditions | Yield |
|---|---|
| Stage #1: 8-quinolinol With sodium hydride In tetrahydrofuran Inert atmosphere; Stage #2: di-isopropyl azodicarboxylate In tetrahydrofuran at -5 - 60℃; for 1.5h; Reagent/catalyst; Solvent; Time; Temperature; Inert atmosphere; | 99.7% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 40 - 90℃; for 1h; | 99.3% |

| Conditions | Yield |
|---|---|
| With bromine; sodium hydrogencarbonate In methanol at 20℃; for 0.0833333h; | 99% |
| With bromine In methanol at 20℃; for 0.0833333h; | 97% |
| Stage #1: 8-quinolinol With bromine; sodium hydrogencarbonate In methanol at 20℃; for 0.0833333h; Stage #2: With sodium sulfite In methanol; water at 20℃; | 97% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 2-carbomethoxy-3-hydroxyquinoxaline-di-N-oxide; caesium carbonate In N,N-dimethyl-formamide at 110℃; for 12h; Schlenk technique; Inert atmosphere; | 99% |
| With potassium carbonate In dimethyl sulfoxide for 0.0833333h; Heating; microwave irradiation; | 94% |
| With PEG-400; sodium hydroxide In N,N-dimethyl-formamide for 15h; Etherification; Irradiation; microwave irradiation; | 54% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 0 - 20℃; for 4h; | 99% |
| With hydrogenchloride In water at 0 - 20℃; for 8h; | 98% |
| With hydrogenchloride In water at 0 - 20℃; for 8h; Product distribution / selectivity; | 98% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; | 99% |
| With triethylamine In dichloromethane at 0 - 20℃; | 94% |
| With triethylamine In ethyl acetate at 0 - 20℃; for 0.166667h; Green chemistry; | 85% |

| Conditions | Yield |
|---|---|
| In triethylamine byproducts: {(C2H5)3NH}Cl; sublimation at 200 °C and 10E-1 Torr;; | 99% |
| In triethylamine byproducts: {(C2H5)3NH}Cl; sublimation at 200 °C and 10E-1 Torr;; | 99% |
| In triethylamine byproducts: {(C2H5)3NH}Cl; | 68% |
| In triethylamine byproducts: {(C2H5)3NH}Cl; | 68% |
-
-
148-24-3
8-quinolinol

-
-
10025-69-1
tin(II) chloride dihdyrate

-
-
12148-41-3
bis(8-hydroxyquinolinate)tin(II)

| Conditions | Yield |
|---|---|
| In ethanol N2-atmosphere; pptn. on dropwise addn. of SnCl2 to ligand (room temp., stirring); washing (EtOH, water), drying (vac., 1 h); | 99% |
-
-
148-24-3
8-quinolinol

-
-
15120-50-0
pentamethylantimony

-
-
22784-64-1
tetramethyl(8-oxyquinolinato)antimony(V)

| Conditions | Yield |
|---|---|
| In toluene byproducts: CH4; refluxing Me5Sb and oxine in toluene for 24 h; evapn. of solvent in vacuo, elem. anal.; | 99% |
-
-
148-24-3
8-quinolinol

-
-
632323-52-5
nitridobis(1,3-diphenylpropane-1,3-dionate)chromium(V)

-
-
632323-51-4
nitridobis(8-hydroxyquinolinate)chromium(V)

| Conditions | Yield |
|---|---|
| In acetonitrile addn. of chromium complex to a soln. of ligand in acetonitrile, reflux for 5 min; hot filtration, cooling; elem. anal.; | 99% |
-
-
148-24-3
8-quinolinol

-
-
55171-05-6, 13814-75-0
molybdenum(VI) tetrachloride oxide

| Conditions | Yield |
|---|---|
| In tetrachloromethane MoOCl4 and equiv. amt. of 8-hydroxyquinoline in CCl4 at room temp.; | 99% |
-
-
148-24-3
8-quinolinol

-
-
55171-05-6, 13814-75-0
molybdenum(VI) tetrachloride oxide

| Conditions | Yield |
|---|---|
| In tetrachloromethane MoOCl4 and equiv. amt. of 8-hydroxyquinoline in CCl4 at room temp.; | 99% |

| Conditions | Yield |
|---|---|
| In tetrachloromethane WOCl4 and equiv. amt. of 8-hydroxyquinoline in CCl4 at room temp.; | 99% |

| Conditions | Yield |
|---|---|
| In tetrachloromethane WOCl4 and equiv. amt. of 8-hydroxyquinoline in CCl4 at room temp.; | 99% |
-
-
148-24-3
8-quinolinol

-
-
511243-86-0
5-[(6-Bicyclo[2.2.1]hept-5-en-2-yl-hexylamino)-methyl]-quinolin-8-ol

-
-
557-20-0
diethylzinc

-
-
796882-43-4
(C7H9(CH2)6NHCH2C9H5NO)zinc(8-hydroxyquinoline)

| Conditions | Yield |
|---|---|
| In not given 1:1:1 molar ratio, ligand added to ZnEt2, followed by quinoline; | 99% |


| Conditions | Yield |
|---|---|
| In chloroform for 72h; Reflux; Schlenk technique; Inert atmosphere; | 99% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View





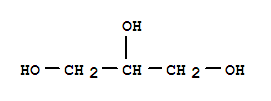









 Xn
Xn