Chemvon Biotechnology Co. Ltd.
Chemvon Biotechnology is one of the leading high-technology manufacturer in the field of pharmaceutical and fine chemical industry. From origins as a research group in technology service, chemvon has progressed into an integrated company with a k
Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:394730-60-0
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySinoway Industrial Co., Ltd.
We are very compeitive on Boceprevir.Our mfr is GMP certified for this item with DMF document. Product Name: Boceprevir Synonyms: Boceprevir (1R,2S,5S)-N-(4-Amino-1-cyclobutyl-3,4-dioxobu
Cas:394730-60-0
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
he company has advanced technology, as well as a large number of excellent R & D team, to provide customers from the grams to one hundred kilograms and tons of high-quality products, competitive prices and quality se T rvice Appearance:
Hangzhou JINLAN Pharm-Drugs Technology Co., Ltd
We can provide GMP validation service that complies with SFDA, FDA, WHO and EU EMPA.Excellent registration team could help us easlily to register our products in different countries.If you and your customer are interested in some products or need C
Cas:394730-60-0
Min.Order:1 Gram
Negotiable
Type:Manufacturers
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:394730-60-0
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Zhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:394730-60-0
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China with ISO certificate, Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED
Cas:394730-60-0
Min.Order:1 Gram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:394730-60-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Maytime Bio-Tech Co.,Ltd.
good quality ,competitive price. Storage:keep from moisture,store in tight container Application: Anti - hepatitis c Transportation:by air or by courier
Qingdao Beluga Import and Export Co., LTD
Boceprevir CAS:394730-60-0 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates
Cas:394730-60-0
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryWuhan Wonda Pharm Limited
1.high quality: quality is life. quality is the most important element for all goods. we have a lab doing research in wuhan china. hplc and nmr is available if needed. 2.reasonable price: we provide high quality products wi
Cas:394730-60-0
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Lonwin Chemical Group Limited
Boceprevir CAS: 394730-60-0 Specification Item Standard Identification A.H-NMR:Comply with the structure B.LC-MS:Comply with the structure C.The IR spectrum of sa
Hebei Quanhe Biotechnology Co. LTD
1. Timely and efficient service to ensure communication with customers 2. Produce products of different specifications and sizes according to your requirements. 3. Quality procedures and standards recognized by SGS. Advanced plant equipment ensures s
Cas:394730-60-0
Min.Order:1 Kilogram
FOB Price: $100.0 / 150.0
Type:Lab/Research institutions
inquiryTaizhou Crene Biotechnology co.ltd
Our company provides one-stop services of research - development - production for a variety of special prouducts. Not only do we make effective use of our strong technological strength, but also establish of cooperative relations with several well-
Cas:394730-60-0
Min.Order:100 Milligram
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:394730-60-0
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHangzhou Lingrui Chemical Co.,Ltd.
BoceprevirAppearance:Pls see the Details Storage:Keep away of light, hot, water, Store in dry, dark and ventilated place Package:according to customers' requirements Application:Steroids, Cosmetics Ingredients, APIs, Intermediates, OLED&Battery Ingre
Hebei yanxi chemical co.,LTD.
Additive Chinese name to allow Chinese name using the additive of food additive function maximum permissible usage (g/kg) maximum allowable residue (g/kg) Butyl acetate food food prepared with spices used to flavor various spices composition shall
Cas:394730-60-0
Min.Order:10 Metric Ton
FOB Price: $850.0 / 1100.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
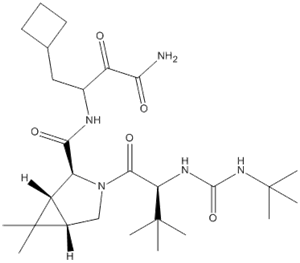
Product Name: Boceprevir Synonyms: Boceprevir (1R,2S,5S)-N-(4-Amino-1-cyclobutyl-3,4-dioxobutan-2-yl)-3-[(2S)-2-(tert-butylcarbamoylamino)-3,3-dimethylbutanoyl]-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2
Cas:394730-60-0
Min.Order:1 Metric Ton
FOB Price: $20.0
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: Boceprevir CAS No.:394730-60-0 Molecule Formula:C27H45N5O5 Molecule Weight:519.68 Purity: 99.0% Package: 25kg/drum Description:White powder Manufacture Standards:Enterprise Standard TESTING ITEMS SPE
Cas:394730-60-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:394730-60-0
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryJiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:394730-60-0
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryTAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:394730-60-0
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquirySynthetic route
-
-
394735-28-5
(1R,2S,5S)-N-(4-amino-1-cyclobutyl-3-hydroxy-4-oxobutan-2-yl)-3-((S)-2-(3-(tert-butyl)ureido)-3,3-dimethylbutanoyl)-6,6-dimethyl-3-azabicylo[3.1.0]-hexane-2-carboxamide

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Stage #1: (1R,2S,5S)-N-(4-amino-1-cyclobutyl-3-hydroxy-4-oxobutan-2-yl)-3-((S)-2-(3-(tert-butyl)ureido)-3,3-dimethylbutanoyl)-6,6-dimethyl-3-azabicylo[3.1.0]-hexane-2-carboxamide With sodium hypochlorite solution; sodium acetate; potassium bromide; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In tert-butyl methyl ether at 10 - 20℃; Stage #2: With water; acetic acid In tert-butyl methyl ether for 2.25h; Stage #3: With sodium hypochlorite solution; ascorbic acid Product distribution / selectivity; more than 3 stages; | 94% |
| With sodium permanganate; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; acetic acid In tert-butyl methyl ether; water at 12℃; | 91% |
| With Dess-Martin periodane In ethyl acetate at 5 - 18℃; for 5.25h; Reagent/catalyst; | 40% |
-
-
817170-37-9
C27H46N5O8S(1-)*Na(1+)

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| With Oxone In tert-butyl methyl ether; water at 25℃; for 1h; Purification / work up; | 94% |
| With dipotassium peroxodisulfate In tert-butyl methyl ether; water at 25℃; for 0.75h; Purification / work up; | 88% |
| With sodium chloride In tert-butyl methyl ether; water at 30℃; for 0.5h; Purification / work up; | 84% |
| With Glyoxilic acid In water at 8 - 25℃; for 4h; Purification / work up; | 65% |
| With sodium glyoxylate In water at 8 - 25℃; for 4.33333h; Purification / work up; | 52% |
-
-
816444-90-3
(1R,2S,5S)-3-((S)-2-(3-tert-butylureido)-3,3-dimethylbutanoyl)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid

-
-
817169-86-1
C8H14N2O2*ClH

-
A
-
394730-60-0
boceprevir

-
B
-
3651-67-0
N-methylmorpholine hydrochloride

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; isobutyl chloroformate In ethyl acetate at 0 - 10℃; for 1h; | A 90% B n/a |

-
-
816444-90-3
(1R,2S,5S)-3-((S)-2-(3-tert-butylureido)-3,3-dimethylbutanoyl)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid

-
-
817169-86-1
C8H14N2O2*ClH

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In acetonitrile at 15 - 20℃; for 1h; | 85% |
-
-
394735-19-4
(2-cyclobutyl-1-formylethyl)carbamic acid tert-butyl ester

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: Et3N / CH2Cl2 / 12 h / 20 °C 2: H2O2; LiOH / methanol / 3 h / 0 °C 3: HCl / dioxane / 2 h / 20 °C 4: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 5: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 8 steps 1: Et3N / CH2Cl2 / 12 h / 20 °C 2: HCl / 24 h / Heating 3: Et3N / CH2Cl2 / 20 °C 4: LiOH*H2O / tetrahydrofuran; H2O / 2 h / 20 °C 5: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 6: HCl / dioxane / 2 h / 20 °C 7: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 8: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
394735-20-7
(2-cyano-1-cyclobutylmethyl-2-hydroxyethyl)carbamic acid tert-butyl ester

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: H2O2; LiOH / methanol / 3 h / 0 °C 2: HCl / dioxane / 2 h / 20 °C 3: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 4: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 7 steps 1: HCl / 24 h / Heating 2: Et3N / CH2Cl2 / 20 °C 3: LiOH*H2O / tetrahydrofuran; H2O / 2 h / 20 °C 4: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 5: HCl / dioxane / 2 h / 20 °C 6: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 7: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
565456-75-9
2-(tert-butoxycarbonylamino)-3-cyclobutylpropanoic acid

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 15.0 g / BOP; N-methylmorpholine / CH2Cl2 / 16 h / 20 °C 2: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 3: Et3N / CH2Cl2 / 12 h / 20 °C 4: H2O2; LiOH / methanol / 3 h / 0 °C 5: HCl / dioxane / 2 h / 20 °C 6: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 7: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 10 steps 1: 15.0 g / BOP; N-methylmorpholine / CH2Cl2 / 16 h / 20 °C 2: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 3: Et3N / CH2Cl2 / 12 h / 20 °C 4: HCl / 24 h / Heating 5: Et3N / CH2Cl2 / 20 °C 6: LiOH*H2O / tetrahydrofuran; H2O / 2 h / 20 °C 7: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 8: HCl / dioxane / 2 h / 20 °C 9: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 10: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
394735-18-3
tert-butyl 3-cyclobutyl-1-(methoxy(methyl)amino)-1-oxopropan-2-ylcarbamate

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 2: Et3N / CH2Cl2 / 12 h / 20 °C 3: H2O2; LiOH / methanol / 3 h / 0 °C 4: HCl / dioxane / 2 h / 20 °C 5: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 6: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 9 steps 1: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 2: Et3N / CH2Cl2 / 12 h / 20 °C 3: HCl / 24 h / Heating 4: Et3N / CH2Cl2 / 20 °C 5: LiOH*H2O / tetrahydrofuran; H2O / 2 h / 20 °C 6: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 7: HCl / dioxane / 2 h / 20 °C 8: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 9: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
394735-21-8
methyl 3-tert-butoxycarbonylamino-4-cyclobutyl-2-hydroxybutyrate

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: LiOH*H2O / tetrahydrofuran; H2O / 2 h / 20 °C 2: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 3: HCl / dioxane / 2 h / 20 °C 4: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 5: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
864765-28-6
3-(tert-butoxycarbonylamino)-4-cyclobutyl-2-hydroxybutanoic acid

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 2: HCl / dioxane / 2 h / 20 °C 3: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 4: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
394735-17-2
ethyl 2-amino-3-cyclobutylpropanoate

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1: CH2Cl2 / 12 h / 20 °C 2: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 3: 15.0 g / BOP; N-methylmorpholine / CH2Cl2 / 16 h / 20 °C 4: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 5: Et3N / CH2Cl2 / 12 h / 20 °C 6: H2O2; LiOH / methanol / 3 h / 0 °C 7: HCl / dioxane / 2 h / 20 °C 8: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 9: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 12 steps 1: CH2Cl2 / 12 h / 20 °C 2: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 3: 15.0 g / BOP; N-methylmorpholine / CH2Cl2 / 16 h / 20 °C 4: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 5: Et3N / CH2Cl2 / 12 h / 20 °C 6: HCl / 24 h / Heating 7: Et3N / CH2Cl2 / 20 °C 8: LiOH*H2O / tetrahydrofuran; H2O / 2 h / 20 °C 9: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 10: HCl / dioxane / 2 h / 20 °C 11: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 12: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
569679-06-7
3-(tert-butyl) 2-methyl (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2,3-dicarboxylate

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: HCl / dioxane 2: BOP; N-methylmorpholine / CH2Cl2; dimethylformamide / 24 h / 20 °C 3: HCl / dioxane / 3 h / 20 °C 4: CH2Cl2 / 20 °C 5: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 6: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 7: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
816430-02-1
ethyl 2-(tert-butoxycarbonylamino)-3-cyclobutylpropanoate

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 2: 15.0 g / BOP; N-methylmorpholine / CH2Cl2 / 16 h / 20 °C 3: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 4: Et3N / CH2Cl2 / 12 h / 20 °C 5: H2O2; LiOH / methanol / 3 h / 0 °C 6: HCl / dioxane / 2 h / 20 °C 7: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 8: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 11 steps 1: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 2: 15.0 g / BOP; N-methylmorpholine / CH2Cl2 / 16 h / 20 °C 3: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 4: Et3N / CH2Cl2 / 12 h / 20 °C 5: HCl / 24 h / Heating 6: Et3N / CH2Cl2 / 20 °C 7: LiOH*H2O / tetrahydrofuran; H2O / 2 h / 20 °C 8: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 9: HCl / dioxane / 2 h / 20 °C 10: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 11: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
394735-22-9
tert-butyl 4-amino-1-cyclobutyl-3-hydroxy-4-oxobutan-2-ylcarbamate

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: HCl / dioxane / 2 h / 20 °C 2: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 3: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: hydrogenchloride / isopropyl alcohol / 4 h / 50 °C / Large scale 2: benzotriazol-1-ol; 4-methyl-morpholine; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / water; ethyl acetate; 1-methyl-pyrrolidin-2-one / 16 h / 20 - 24 °C 3: acetic acid; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; sodium permanganate / tert-butyl methyl ether; water / 12 °C View Scheme |
-
-
1025799-39-6
2-(benzhydrylidene-amino)-3-cyclobutyl-propionic acid ethyl ester

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 18 g / aq. HCl / diethyl ether / 5 h / 20 °C 2: CH2Cl2 / 12 h / 20 °C 3: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 4: 15.0 g / BOP; N-methylmorpholine / CH2Cl2 / 16 h / 20 °C 5: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 6: Et3N / CH2Cl2 / 12 h / 20 °C 7: H2O2; LiOH / methanol / 3 h / 0 °C 8: HCl / dioxane / 2 h / 20 °C 9: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 10: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 13 steps 1: 18 g / aq. HCl / diethyl ether / 5 h / 20 °C 2: CH2Cl2 / 12 h / 20 °C 3: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 4: 15.0 g / BOP; N-methylmorpholine / CH2Cl2 / 16 h / 20 °C 5: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 6: Et3N / CH2Cl2 / 12 h / 20 °C 7: HCl / 24 h / Heating 8: Et3N / CH2Cl2 / 20 °C 9: LiOH*H2O / tetrahydrofuran; H2O / 2 h / 20 °C 10: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 11: HCl / dioxane / 2 h / 20 °C 12: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 13: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
394735-26-3
(1R,2S,5S)-3-((S)-2-tert-Butoxycarbonylamino-3,3-dimethyl-butyryl)-6,6-dimethyl-3-aza-bicyclo[3.1.0]hexane-2-carboxylic acid methyl ester

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: HCl / dioxane / 3 h / 20 °C 2: CH2Cl2 / 20 °C 3: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 4: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 5: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: BOP; N-methylmorpholine / CH2Cl2; dimethylformamide / 24 h / 20 °C 2: HCl / dioxane / 3 h / 20 °C 3: CH2Cl2 / 20 °C 4: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 5: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 6: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: N-ethyl-N,N-diisopropylamine; benzotriazol-1-ol / dichloromethane / 0.17 h / 0 - 5 °C 1.2: 6 h / 25 - 30 °C 2.1: water; lithium hydroxide monohydrate / tetrahydrofuran / 6 h / 25 - 30 °C 3.1: N-ethyl-N,N-diisopropylamine; benzotriazol-1-ol; dicyclohexyl-carbodiimide / dichloromethane / 5 h / 0 - 30 °C 4.1: Dess-Martin periodane / dichloromethane / 0 - 30 °C View Scheme |
-
-
394735-27-4
methyl (1R,2S,5S)-3-((S)-2-(3-(tert-butyl)-ureido)-3,3-dimethylbutanoyl)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylate

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 2: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 3: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: water; lithium hydroxide monohydrate / tetrahydrofuran / 6 h / 25 - 30 °C 2: N-ethyl-N,N-diisopropylamine; benzotriazol-1-ol; dicyclohexyl-carbodiimide / dichloromethane / 5 h / 0 - 30 °C 3: Dess-Martin periodane / dichloromethane / 0 - 30 °C View Scheme | |
| Multi-step reaction with 3 steps 1: water; sodium hydroxide / toluene; methanol / 0 - 30 °C 2: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; 4-methyl-morpholine / N,N-dimethyl-formamide; ethyl acetate / 0 - 10 °C 3: Dess-Martin periodane / ethyl acetate / 5.25 h / 5 - 18 °C View Scheme |

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: Et3N / CH2Cl2 / 20 °C 2: LiOH*H2O / tetrahydrofuran; H2O / 2 h / 20 °C 3: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 4: HCl / dioxane / 2 h / 20 °C 5: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 6: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 2: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: benzotriazol-1-ol; 4-methyl-morpholine; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / water; ethyl acetate; 1-methyl-pyrrolidin-2-one / 16 h / 20 - 24 °C 2: acetic acid; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; sodium permanganate / tert-butyl methyl ether; water / 12 °C View Scheme |
-
-
816444-90-3
(1R,2S,5S)-3-((S)-2-(3-tert-butylureido)-3,3-dimethylbutanoyl)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 2: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: N-ethyl-N,N-diisopropylamine; benzotriazol-1-ol; dicyclohexyl-carbodiimide / dichloromethane / 5 h / 0 - 30 °C 2: Dess-Martin periodane / dichloromethane / 0 - 30 °C View Scheme |

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: CH2Cl2 / 20 °C 2: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 3: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 4: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
17247-58-4
(Bromomethyl)cyclobutane

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1.1: t-BuOK / tetrahydrofuran / 1 h / 0 °C 1.2: tetrahydrofuran / 48 h / 20 °C 2.1: 18 g / aq. HCl / diethyl ether / 5 h / 20 °C 3.1: CH2Cl2 / 12 h / 20 °C 4.1: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 5.1: 15.0 g / BOP; N-methylmorpholine / CH2Cl2 / 16 h / 20 °C 6.1: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 7.1: Et3N / CH2Cl2 / 12 h / 20 °C 8.1: H2O2; LiOH / methanol / 3 h / 0 °C 9.1: HCl / dioxane / 2 h / 20 °C 10.1: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 11.1: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 14 steps 1.1: t-BuOK / tetrahydrofuran / 1 h / 0 °C 1.2: tetrahydrofuran / 48 h / 20 °C 2.1: 18 g / aq. HCl / diethyl ether / 5 h / 20 °C 3.1: CH2Cl2 / 12 h / 20 °C 4.1: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 5.1: 15.0 g / BOP; N-methylmorpholine / CH2Cl2 / 16 h / 20 °C 6.1: 14 g / LiAlH4 / tetrahydrofuran / 1 h / 20 °C 7.1: Et3N / CH2Cl2 / 12 h / 20 °C 8.1: HCl / 24 h / Heating 9.1: Et3N / CH2Cl2 / 20 °C 10.1: LiOH*H2O / tetrahydrofuran; H2O / 2 h / 20 °C 11.1: NH4Cl; EDCI; HOOBt / NMM / CH2Cl2; dimethylformamide / 72 h / 20 °C 12.1: HCl / dioxane / 2 h / 20 °C 13.1: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 14.1: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
134107-65-6
(3R,7aS)-3-phenyl-3,7a-dihydro-1H-pyrrolo[1,2-c]oxazol-5-one

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 12 steps 1.1: BuLi / tetrahydrofuran 1.2: tetrahydrofuran / -78 - 20 °C 2.1: LiAlH4 / tetrahydrofuran / Heating 3.1: H2 / Pd/C 4.1: acetone / 3 h / -5 °C 5.1: toluene; methanol 6.1: HCl / dioxane 7.1: BOP; N-methylmorpholine / CH2Cl2; dimethylformamide / 24 h / 20 °C 8.1: HCl / dioxane / 3 h / 20 °C 9.1: CH2Cl2 / 20 °C 10.1: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 11.1: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 12.1: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
103201-79-2
(3R,7aS)-3-phenyl-tetrahydro-pyrrolo[1,2-c]oxazol-5-one

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 13 steps 1.1: KHMDS; PhSeCl / -78 - 20 °C 1.2: H2O2; pyridine 2.1: BuLi / tetrahydrofuran 2.2: tetrahydrofuran / -78 - 20 °C 3.1: LiAlH4 / tetrahydrofuran / Heating 4.1: H2 / Pd/C 5.1: acetone / 3 h / -5 °C 6.1: toluene; methanol 7.1: HCl / dioxane 8.1: BOP; N-methylmorpholine / CH2Cl2; dimethylformamide / 24 h / 20 °C 9.1: HCl / dioxane / 3 h / 20 °C 10.1: CH2Cl2 / 20 °C 11.1: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 12.1: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 13.1: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
1026427-11-1
((1R,2S,5S)-3-Benzyl-6,6-dimethyl-3-aza-bicyclo[3.1.0]hex-2-yl)-methanol

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: H2 / Pd/C 2: acetone / 3 h / -5 °C 3: toluene; methanol 4: HCl / dioxane 5: BOP; N-methylmorpholine / CH2Cl2; dimethylformamide / 24 h / 20 °C 6: HCl / dioxane / 3 h / 20 °C 7: CH2Cl2 / 20 °C 8: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 9: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 10: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
219754-00-4
tert-butyl (1R,2S,5S)-2-(hydroxymethyl)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-3-carboxylate

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1: acetone / 3 h / -5 °C 2: toluene; methanol 3: HCl / dioxane 4: BOP; N-methylmorpholine / CH2Cl2; dimethylformamide / 24 h / 20 °C 5: HCl / dioxane / 3 h / 20 °C 6: CH2Cl2 / 20 °C 7: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 8: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 9: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
-
-
219754-02-6
(1R,2S,5S)-3-(tert-butoxycarbonyl)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: toluene; methanol 2: HCl / dioxane 3: BOP; N-methylmorpholine / CH2Cl2; dimethylformamide / 24 h / 20 °C 4: HCl / dioxane / 3 h / 20 °C 5: CH2Cl2 / 20 °C 6: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 7: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 8: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1: N-ethyl-N,N-diisopropylamine; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol / N,N-dimethyl-formamide; dichloromethane / 0 - 30 °C 2: hydrogenchloride / isopropyl alcohol / 6 h / 60 - 65 °C 3: N-ethyl-N,N-diisopropylamine; HATU / acetonitrile / 5 h / 25 - 30 °C 4: Dess-Martin periodane / dichloromethane / 0 - 30 °C View Scheme |
-
-
219753-99-8
(1S,2S,4S,7R)-6-aza-3,3-dimethyl-8-oxa-7-phenyltricyclo<4.3.0.0>nonan-5-one

-
-
394730-60-0
boceprevir

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1: LiAlH4 / tetrahydrofuran / Heating 2: H2 / Pd/C 3: acetone / 3 h / -5 °C 4: toluene; methanol 5: HCl / dioxane 6: BOP; N-methylmorpholine / CH2Cl2; dimethylformamide / 24 h / 20 °C 7: HCl / dioxane / 3 h / 20 °C 8: CH2Cl2 / 20 °C 9: LiOH*H2O / tetrahydrofuran; H2O / 3 h / 20 °C 10: EDCI; HOOBt; N-methylmorpholine / dimethylformamide; CH2Cl2 / 48 h / -20 °C 11: dichloroacetic acid; EDCI; DMSO / toluene / 4 h / 20 °C View Scheme |
Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View