Hangzhou Think Chemical Co. Ltd
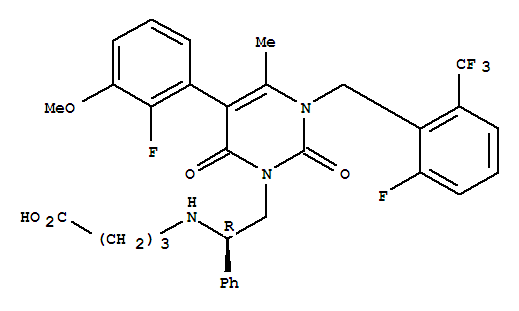
Elagolix CAS No.:834153-87-6 Name: Elagolix Synonyms: 4-[[(1R)-2-[5-(2-Fluoro-3-methoxyphenyl)-3-[[2-fluoro-6-(trifluoromethyl)phenyl]methyl]-3,6-dihydro-4-methyl-2,6-dioxo-1(2H)-pyrimidin
Cas:834153-87-6
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Other
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:834153-87-6
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryEnke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new api, intermedi
Cas:834153-87-6
Min.Order:1 Kilogram
FOB Price: $5.0 / 10.0
Type:Manufacturers
inquiryShanghai Seasonsgreen Chemical Co.,Ltd
Shanghai Seasonsgreen Chemical is a high-tech research and development, production, sale and custom synthesis set in one high-tech chemical products enterprises. Our sales and marketing division is located in Shanghai, serving international pharmaceu
Cas:834153-87-6
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate. In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so o
Cas:834153-87-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryLIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:834153-87-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:834153-87-6
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Cas:834153-87-6
Min.Order:10 Kilogram
Negotiable
Type:Trading Company
inquiryHebei yanxi chemical co.,LTD.
hebei yanxi chemical co., LTD who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and developmen
Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis according to the contract research and development services for the fine chemicals, ph
HANWAYS CHEMPHARM CO.,LIMITED
1.Prompt Goods; 2.Flexible payment terms; 3.Documents & Certificate support. Name: Elagolix Structure: Formula: C32H30F5N3O5 Molecular Weight : 631.59 Synonyms: elagolix;4-[[(1R)-2-[5-(2-Fluoro-3-methoxyphenyl)-3-[[2-fluoro-6-(trifluorome
Cas:834153-87-6
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
elagolix CAS:834153-87-6 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates,
Cas:834153-87-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:834153-87-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou JINLAN Pharm-Drugs Technology Co., Ltd
We can provide GMP validation service that complies with SFDA, FDA, WHO and EU EMPA.Excellent registration team could help us easlily to register our products in different countries.If you and your customer are interested in some products or need C
Cas:834153-87-6
Min.Order:1 Gram
Negotiable
Type:Trading Company
inquiryHebei Mojin Biotechnology Co.,Ltd
Hebei Mojin Biotechnology Co., Ltd,Our business covers more than 30 countries, most of the big customers come from Europe, America and other countries in the world, we can guarantee the quality and price. In recent decades, with the efforts of all em
Cas:834153-87-6
Min.Order:1 Gram
FOB Price: $100.0
Type:Trading Company
inquiryTaizhou Crene Biotechnology co.ltd
Our company provides one-stop services of research - development - production for a variety of special prouducts. Not only do we make effective use of our strong technological strength, but also establish of cooperative relations with several well-
Cas:834153-87-6
Min.Order:100 Milligram
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:834153-87-6
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryShanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:834153-87-6
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
SHANGHAI SYSTEAM BIOCHEM CO., LTD
We are one of a few suppliers that can offer custom synthesis service of this product We are specialized in custom synthesis, chemical/pharmaceutical/ pesticides outsourcing and contract research. We are committed to prov
TaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
Siwei Development Group Ltd.
Product name: Elagolix CAS No.:834153-87-6 Molecule Formula:C32H30F5N3O5 Molecule Weight:631.59 Purity: 99.0% Package: 25kg/drum Description:White powder Manufacture Standards:Enterprise Standard TESTING ITEMS SPE
Cas:834153-87-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
1. DMF is available. 2. Under registerring in Russia and Turkey. Hangzhou Huarong Pharm Co., Ltd. established since 2009 , has been always focusing on supplying products and services to our clients in the field of small molecule drug. Huaro
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Henan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China with ISO certificate, Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED
Cas:834153-87-6
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
elagolix Chemical Properties Boiling point 728.6±70.0 °C(Predicted) density 1.350 pka 4.40±0.10(Predicted) Appearance:powder Storage:
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Synthetic route
-
-
832720-84-0
4-((R)-2-[5-(2-fluoro-3-methoxyphenyl)-3-(2-fluoro-6-trifluoromethylbenzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidine-1-yl]-1-phenylethylamino)butyric acid ethyl ester

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Stage #1: ethyl (R)-4-((2-(5-(2-fluoro-3-methoxyphenyl)-3-(2-fluoro-6-(trifluoromethyl)benzyl)-4-methyl-2,6-dioxo-3,6-dihydropyrimidin-1(2H)-yl)-1-phenylethyl)amino)butanoate With sodium hydroxide; water In ethanol at 35℃; for 2h; Stage #2: With hydrogenchloride In water at 10 - 22℃; for 17h; pH=6.1; | 91% |
| With water; sodium hydroxide In ethanol at 35℃; | 86% |
| With water; sodium hydroxide In ethanol at 50℃; for 2h; | 83.59% |

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide In ethanol at 55 - 60℃; | 89% |

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With water; potassium carbonate In tetrahydrofuran at 50℃; for 6h; Solvent; Temperature; | 82% |
-
-
830346-50-4
3-[(2R)-amino-2-phenylethyl]-5-(2-fluoro-3-methoxyphenyl)-1-[2-fluoro-6-(trifluoromethyl)benzyl]-6-methyl-2,4(1H,3H)-pyrimidinedione trifluoroacetate

-
-
692-29-5
Succinic semialdehyde

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; acetic acid In dichloromethane at 25℃; for 18h; Reagent/catalyst; Solvent; Temperature; | 65% |
-
-
1092070-97-7
methyl (R)-4-{2-[5-(2-fluoro-3-methoxyphenyl)-3-[2-fluoro-6-(trifluoromethyl)benzyl]-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl]-1-phenylethylamino}butyrate

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With water; lithium hydroxide In methanol at 45℃; |
-
-
830346-50-4
3-[(2R)-amino-2-phenylethyl]-5-(2-fluoro-3-methoxyphenyl)-1-[2-fluoro-6-(trifluoromethyl)benzyl]-6-methyl-2,4(1H,3H)-pyrimidinedione trifluoroacetate

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 52 °C 2: sodium hydroxide; water / ethanol / 35 °C View Scheme | |
| Multi-step reaction with 2 steps 1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 2: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 2 steps 1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 2: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |
-
-
1150560-59-0
5-(2-fluoro-3-methoxyphenyl)-1-{[2-fluoro-6-(trifluoromethyl)phenyl]methyl}-6-methylpyrimidine-2,4(1H,3H)-dione

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: potassium carbonate / N,N-dimethyl-formamide / 55 °C 2: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 52 °C 3: sodium hydroxide; water / ethanol / 35 °C View Scheme | |
| Multi-step reaction with 3 steps 1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 2: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 3: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 3 steps 1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 2: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 3: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |
-
-
1150560-54-5
1-{[2-fluoro-6-(trifluoromethyl)phenyl]methyl}-5-iodo-6-methylpyrimidine-2,4(1H,3H)-dione

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium hydroxide; tri tert-butylphosphoniumtetrafluoroborate / acetone; water / 0.83 h / 15 - 45 °C 2: potassium carbonate / N,N-dimethyl-formamide / 55 °C 3: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 52 °C 4: sodium hydroxide; water / ethanol / 35 °C View Scheme |
-
-
352303-67-4
(2-fluoro-3-methoxyphenyl)boronic acid

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium hydroxide; tri tert-butylphosphoniumtetrafluoroborate / acetone; water / 0.83 h / 15 - 45 °C 2: potassium carbonate / N,N-dimethyl-formamide / 55 °C 3: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 52 °C 4: sodium hydroxide; water / ethanol / 35 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: methanol; water / 24 h / 70 °C 2.1: tetra(n-butyl)ammonium hydroxide / water / 0.03 h / Inert atmosphere 2.2: 1 h / 80 °C / Inert atmosphere 3.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 4.1: toluene / 4 h / Reflux 4.2: 2 h / Reflux 5.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 6.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 7.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: methanol; water / 24 h / 70 °C 2.1: tetra(n-butyl)ammonium hydroxide / water / 0.03 h / Inert atmosphere 2.2: 1 h / 80 °C / Inert atmosphere 3.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 4.1: toluene / 4 h / Reflux 4.2: 2 h / Reflux 5.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 6.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 7.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: methanol; water / 24 h / 70 °C 2.1: tetra(n-butyl)ammonium hydroxide / water / 0.03 h / Inert atmosphere 2.2: 1 h / 80 °C / Inert atmosphere 3.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 4.1: toluene / 4 h / Reflux 4.2: 2 h / Reflux 5.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 6.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 7.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: methanol; water / 24 h / 70 °C 2.1: tetra(n-butyl)ammonium hydroxide / water / 0.03 h / Inert atmosphere 2.2: 1 h / 80 °C / Inert atmosphere 3.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 4.1: toluene / 4 h / Reflux 4.2: 2 h / Reflux 5.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 6.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 7.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |
-
-
110143-62-9
(S)-(+)-<2-<(methylsulfonyl)oxy>-1-phenylethyl>carbamic acid 1,1-dimethylethyl ester

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: potassium carbonate / N,N-dimethyl-formamide / 55 °C 2: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 52 °C 3: sodium hydroxide; water / ethanol / 35 °C View Scheme |
-
-
2900-27-8
(2S)-2-[(tert-butoxycarbonyl)amino]-2-phenylethanoic acid

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 0 - 10 °C 2: potassium carbonate / N,N-dimethyl-formamide / 55 °C 3: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 52 °C 4: sodium hydroxide; water / ethanol / 35 °C View Scheme |

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: toluene / 105 - 110 °C 1.2: 105 - 110 °C 2.1: potassium carbonate; sodium iodide / N,N-dimethyl-formamide / 90 - 100 °C 3.1: sodium hydroxide; water / ethanol / 55 - 60 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |
-
-
15018-56-1
5-bromo-6-methyluracil

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium carbonate / N,N-dimethyl-formamide / 75 - 85 °C 2: iron(III)-acetylacetonate; N,N,N,N,-tetramethylethylenediamine / tetrahydrofuran / 0 - 45 °C 3: potassium carbonate; sodium iodide / N,N-dimethyl-formamide / 90 - 100 °C 4: sodium hydroxide; water / ethanol / 55 - 60 °C View Scheme |
-
-
239087-08-2
1-(bromomethyl)-2-fluoro-6-(trifluoromethyl)benzene

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium carbonate / N,N-dimethyl-formamide / 75 - 85 °C 2: iron(III)-acetylacetonate; N,N,N,N,-tetramethylethylenediamine / tetrahydrofuran / 0 - 45 °C 3: potassium carbonate; sodium iodide / N,N-dimethyl-formamide / 90 - 100 °C 4: sodium hydroxide; water / ethanol / 55 - 60 °C View Scheme |
-
-
830346-48-0
5-bromo-1-[2-fluoro-6-(trifluoromethyl)benzyl]-6-methylpyrimidine-2,4-(1H,3H)-dione

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: iron(III)-acetylacetonate; N,N,N,N,-tetramethylethylenediamine / tetrahydrofuran / 0 - 45 °C 2: potassium carbonate; sodium iodide / N,N-dimethyl-formamide / 90 - 100 °C 3: sodium hydroxide; water / ethanol / 55 - 60 °C View Scheme |
-
-
295376-21-5
1-bromo-2-fluoro-3-methoxybenzene

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: potassium carbonate; L-proline; copper(l) iodide / dimethyl sulfoxide / 45 - 50 °C / Inert atmosphere 2.1: toluene / 105 - 110 °C 2.2: 105 - 110 °C 3.1: potassium carbonate; sodium iodide / N,N-dimethyl-formamide / 90 - 100 °C 4.1: sodium hydroxide; water / ethanol / 55 - 60 °C View Scheme |

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |
-
-
239087-06-0
(2-fluoro-6-(trifluoromethyl) phenyl)methanamine

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: hydrogenchloride / water / 3 h / Reflux 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: hydrogenchloride / water / 3 h / Reflux 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: hydrogenchloride / water / 3 h / Reflux 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |
-
-
60611-24-7
2-fluoro-6-(trifluoromethyl)benzaldehyde

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: hydroxylamine hydrochloride; sodium hydroxide / water; ethanol / 1 h 1.2: 2 h 2.1: hydrogenchloride / water / 3 h / Reflux 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: hydroxylamine hydrochloride; sodium hydroxide / water; ethanol / 1 h 1.2: 2 h 2.1: hydrogenchloride / water / 3 h / Reflux 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: hydroxylamine hydrochloride; sodium hydroxide / water; ethanol / 1 h 1.2: 2 h 2.1: hydrogenchloride / water / 3 h / Reflux 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |
-
-
830346-46-8
N-{[2-fluoro-6-(trifluoromethyl)phenyl]methyl}urea

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: toluene / 4 h / Reflux 1.2: 2 h / Reflux 2.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 3.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 4.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: tetra(n-butyl)ammonium hydroxide / water / 0.03 h / Inert atmosphere 1.2: 1 h / 80 °C / Inert atmosphere 2.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: tetra(n-butyl)ammonium hydroxide / water / 0.03 h / Inert atmosphere 1.2: 1 h / 80 °C / Inert atmosphere 2.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: tetra(n-butyl)ammonium hydroxide / water / 0.03 h / Inert atmosphere 1.2: 1 h / 80 °C / Inert atmosphere 2.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: tetra(n-butyl)ammonium hydroxide / water / 0.03 h / Inert atmosphere 1.2: 1 h / 80 °C / Inert atmosphere 2.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: 5,5-dimethyl-1,3-cyclohexadiene / 6 h / Reflux 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |
-
-
1080673-30-8
2-fluoro-1-iodo-3-methoxybenzene

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: copper(l) iodide; 4R-4-hydroxyproline; caesium carbonate / dimethyl sulfoxide / 96 h / 60 °C / Heating 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: copper(l) iodide; 4R-4-hydroxyproline; caesium carbonate / dimethyl sulfoxide / 96 h / 60 °C / Heating 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: copper(l) iodide; 4R-4-hydroxyproline; caesium carbonate / dimethyl sulfoxide / 96 h / 60 °C / Heating 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: copper(l) iodide; 4R-4-hydroxyproline; caesium carbonate / dimethyl sulfoxide / 96 h / 60 °C / Heating 2.1: toluene / 4 h / Reflux 2.2: 2 h / Reflux 3.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 4.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 5.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |
-
-
321-28-8
1-fluoro-2-methoxybenzene

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: N,N,N',N'',N'''-pentamethyldiethylenetriamine; n-butyllithium / tetrahydrofuran / 1 h / -78 °C 1.2: 2 h / -78 °C 2.1: copper(l) iodide; 4R-4-hydroxyproline; caesium carbonate / dimethyl sulfoxide / 96 h / 60 °C / Heating 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: N,N,N',N'',N'''-pentamethyldiethylenetriamine; n-butyllithium / tetrahydrofuran / 1 h / -78 °C 1.2: 2 h / -78 °C 2.1: copper(l) iodide; 4R-4-hydroxyproline; caesium carbonate / dimethyl sulfoxide / 96 h / 60 °C / Heating 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: N,N,N',N'',N'''-pentamethyldiethylenetriamine; n-butyllithium / tetrahydrofuran / 1 h / -78 °C 1.2: 2 h / -78 °C 2.1: copper(l) iodide; 4R-4-hydroxyproline; caesium carbonate / dimethyl sulfoxide / 96 h / 60 °C / Heating 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: N,N,N',N'',N'''-pentamethyldiethylenetriamine; n-butyllithium / tetrahydrofuran / 1 h / -78 °C 1.2: 2 h / -78 °C 2.1: copper(l) iodide; 4R-4-hydroxyproline; caesium carbonate / dimethyl sulfoxide / 96 h / 60 °C / Heating 3.1: toluene / 4 h / Reflux 3.2: 2 h / Reflux 4.1: potassium carbonate / N,N-dimethyl-formamide / 7 h / 100 °C 5.1: N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 2 h / 60 °C 6.1: sodium hydroxide; water / ethanol / 2 h / 50 °C View Scheme |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With phosphoric acid In ethyl acetate at 0 - 10℃; | 93.6% |
| Multi-step reaction with 2 steps 1: sodium hydroxide / water / 1 h / 60 °C 2: phosphoric acid / water / 25 °C View Scheme |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With dmap; di-tert-butyl dicarbonate In acetonitrile | 92% |
| Stage #1: 4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid With triethylamine In dichloromethane at 20℃; for 1h; Stage #2: With 1,1'-carbonyldiimidazole In dichloromethane at 20℃; for 24h; | 64.8% |
| In butanone at 25℃; for 168h; | 93.03 %Chromat. |
| Multi-step reaction with 2 steps 1: sodium hydroxide / water / 1 h / 60 °C 2: butanone / 168 h / 25 °C View Scheme |
-
-
50-00-0
formaldehyd

-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With borane pyridine complex In methanol at 20℃; | |
| With borane pyridine In methanol |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With Dowex MSC-1 | 1.58 g |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With water-d2; sodium carbonate for 15h; Reflux; |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium carbonate; water-d2 / 15 h / Reflux 2: sodium carbonate; water-d2 / 15 h / Reflux View Scheme |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydroxide / water / 1 h / 60 °C 2: hydrogenchloride / water / 25 °C View Scheme |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydroxide / water / 1 h / 60 °C 2: hydrogen bromide / water / 25 °C View Scheme |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydroxide / water / 1 h / 60 °C 2: nitric acid / water / 25 °C View Scheme |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydroxide / water / 1 h / 60 °C 2: sulfuric acid / water / 25 °C View Scheme | |
| With sulfuric acid In acetonitrile at 27℃; Solvent; Temperature; |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water at 60℃; for 1h; | 3.7 g |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

| Conditions | Yield |
|---|---|
| With calcium acetate In water at 60℃; for 1h; | 3.5 g |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

-
-
130-85-8
Pamoic acid

| Conditions | Yield |
|---|---|
| In methanol at 25 - 60℃; for 19h; | 1.5 g |
-
-
834153-87-6
4-((R)-2-[5-(2- fluoro-3-methoxy-phenyl)-3-(2-fluoro-6-trifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl ]-1- phenylethylamino)-butyric acid

-
-
98-11-3
benzenesulfonic acid

| Conditions | Yield |
|---|---|
| In methanol at 20 - 60℃; for 26h; | 1.1 g |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View