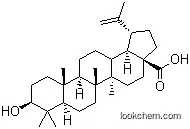
472-15-1Relevant articles and documents
A SAPONIN FROM ASPARAGUS GONOCLADUS
Mandloi, Dilip,Sant, P. G.
, p. 1687 - 1688 (1981)
The ethanolic extract of the aerial part of Asparagus gonocladus yielded a new saponin.It was identified as lup-20(29)-en-28-oic-3-O-α-L-rhamnopyranosyl-(2->1)-O-β-D-glucopyranoside. - Keywords: Asparagus gonocladus; Liliaceae; lup-20(29)-en-28-oic-3-O-α-L-rhamnopyranosyl-(2->1)-O-β-D-glucopyranoside.
α-L-RHAMNOPYRANOSYL-3β-HYDROXY-LUP-20(29)-EN-28-OIC ACID FROM THE STEM OF DILLENIA PENTAGYNA
Tiwari, Kamala P.,Srivastava, Savitri D.,Srivastava, Santosh K.
, p. 980 - 981 (1980)
Key Word Index: Dillenia pentagyna; Dilleniaceae; α-L-rhamnopyranosyl-3β-hydroxy-lup-20(29)-en-28-oic acid; a new saponin.
Design, synthesis and evaluation of antiproliferative activity of fluorinated betulinic acid
Li, Jizhen,Chang, Ling-Chu,Hsieh, Kan-Yen,Hsu, Pei-Ling,Capuzzi, Stephen J.,Zhang, Ying-Chao,Li, Kang-Po,Morris-Natschke, Susan L.,Goto, Masuo,Lee, Kuo-Hsiung
, p. 2871 - 2882 (2019)
Betulinic acid (BA), a pentacyclic triterpenoid, exhibits broad spectrum antiproliferative activity, but generally with only modest potency. To improve BA's pharmacological properties, fluorine was introduced as a single atom at C-2, creating two diastereomers, or in a trifluoromethyl group at C-3. We evaluated the impact of these groups on antiproliferative activity against five human tumor cell lines. A racemic 2-F-BA (compound 6) showed significantly improved antiproliferative activity, while each diastereomer exhibited similar effects. We also demonstrated that 2-F-BA is a topoisomerase (Topo) I and IIα dual inhibitor in cell-based and cell-free assays. A hypothetical mode of binding to the Topo I-DNA suggested a difference between the hydrogen bonding of BA and 2-F-BA to DNA, which may account for the difference in bioactivity against Topo I.
A BETULINIC ACID GLYCOSIDE FROM SCHEFFLERA VENULOSA
Purohit, M. C.,Pant, G.,Rawat, M. S. M.
, p. 2419 (1991)
A new betulinic acid glycoside, lup-20(29)-en-28-oic-3-O-β-D-glucopyranosyl (2->1)-O-β-D-glucopyranoside has been characterized from the leaves of Schefflera venulosa.
Genotoxic and mutagenic properties of synthetic betulinic acid and betulonic acid
Frolova,Kukina,Sinitsyna
, p. 409 - 413 (2015)
Abstract Betulinic acid was synthesized from birch bark extract (Betula pendula), with betulonic acid being an intermediate of the synthesis. Both compounds were isolated with a purity of 95%. Genotoxicity and mutagenicity of the prepared compounds was analyzed by the Ames test and SOS chromotest and it was found that the substances show no mutagenic and genotoxic activity.
Catalytic oxidative transformation of betulin to its valuable oxo-derivatives over gold supported catalysts: Effect of support nature
Kolobova,M?ki-Arvela,Grigoreva,Pakrieva,Carabineiro,Peltonen,Kazantsev,Bogdanchikova,Pestryakov,Murzin, D.Yu.
, p. 95 - 110 (2020/08/26)
Liquid-phase oxidation of betulin extracted from birch bark was studied over gold catalysts supported on a range of supports (hydrotalcite, ZrO2, ZnO, MgO, CeO2, La2O3, HMS and various alumina) with different morphology and properties. Gold catalysts as well as the corresponding supports were characterized by XRD, BET, ICP-OES, XPS and TEM. It was found that the nature of the support plays a decisive role in betulin oxidation over gold-based catalysts, expressed in the influence on the average size and distribution of gold nanoparticles and, thereby, on their catalytic performance (activity and selectivity) in betulin oxidation. Moreover, it was revealed that betulin oxidation catalyzed by gold is a structure sensitive reaction, requiring an optimal size of gold nanoparticles of ca. 3.3 nm. The most suitable support for gold was found to be alumina. Kinetic studies allowed determination of reaction orders and conditions favorable for selective formation of a particular oxo-derivative of betulin (betulone, betulinic and betulonic aldehydes, betulinic acid).
Rhodamine B-based fluorescent probes for molecular mechanism study of the anti-influenza activity of pentacyclic triterpenes
Chen, Yingying,Li, Man,Ma, Wenxiao,Ran, Fuxiang,Xiao, Sulong,Yuan, Lan,Zhang, Lihe,Zhou, Demin
supporting information, (2020/08/05)
The antiviral activity of pentacyclic triterpenes has attracted increasing attention. However, the detailed antiviral mechanism remains fully unclear. In the present study, four C28 or C30 modified pentacyclic triterpene probes via conjugating with rhodamine B were designed and synthesized, and their anti-influenza virus activity was evaluated. The results indicated that two compounds 14 and 23 showed significant antiviral activity to influenza A/WSN/33 (H1N1) virus in Madin-Darby canine kidney (MDCK) cells with IC50 values of 8.36 and 8.24 μM, respectively. The mechanism of action studies of representative probe 23 indicated that it could inhibit the membrane fusion by binding with influenza virus hemagglutinin (HA), and the apparent dissociation constant (KD) value for probe 23-HA interaction was successfully evaluated (1.78 × 10?5 M) using surface plasmon resonance spectroscopy. In addition, the subcellular localization of probe 23 in MDCK cells was determined by confocal microscopy and flow cytometry, and the results suggested that fluorescent probe 23 was rapidly taken up in MDCK cells and accumulated in cytoplasm, but no antiviral activity was observed after its entry into cells. The present study further confirmed our previous finding that pentacyclic triterpenes could tightly bind to the viral envelope HA protein, thus blocking the virus entry into host cells.