-
Name
(3Z,7Z)-3,7-Dimethyl-10-propan-2-ylidene-cyclodeca-3,7-dien-1-one
- EINECS
- CAS No. 6902-91-6
- Article Data2
- CAS DataBase
- Density 0.916 g/cm3
- Solubility
- Melting Point 55-58 °C
- Formula C15H22O
- Boiling Point 330.274 °C at 760 mmHg
- Molecular Weight 218.339
- Flash Point 148.419 °C
- Transport Information
- Appearance
- Safety 23-24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 3,7-Cyclodecadien-1-one,3,7-dimethyl-10-(1-methylethylidene)-, (E,E)-;Germacra-3,7(11),9-trien-6-one,(E,E)- (8CI);Germacrone (6CI,7CI);(E,E)-Germacrone;Germacron;
- PSA 17.07000
- LogP 4.35850
Synthetic route
-
-
87920-50-1
(3E,7E)-1-Hydroxy-10-isopropylidene-3,7-dimethyl-cyclodeca-3,7-dienecarbonitrile

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| With sodium hydroxide In diethyl ether for 0.333333h; Yield given; |
-
-
37905-03-6
8-hydroxygeranyl acetate

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 13 steps 2: 65 percent / triphenylphosphine / palladium acetate / tetrahydrofuran / 5 h / Heating 3: 95 percent / sodium hydride / diethyl ether / 0 °C 4: 70 percent / diethyl ether / -10 °C 5: 90 percent / triphenylphosphine, carbon tetrachloride / 12 h / Heating 6: diisobutylaluminium hydride / tetrahydrofuran / -40 °C 7: manganese dioxide / hexane / Ambient temperature 8: KCN, 18-crown-6 / 1 h / 0 °C 9: trimethylbenzylammonium fluoride / tetrahydrofuran; H2O / 0 °C 11: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 12: p-toluenesulfonic acid / methanol / 1 h / 0 °C 13: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87920-44-3
(4E,8E)-10-Chloro-2-isopropylidene-5,9-dimethyl-deca-4,8-dien-1-ol

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: manganese dioxide / hexane / Ambient temperature 2: KCN, 18-crown-6 / 1 h / 0 °C 3: trimethylbenzylammonium fluoride / tetrahydrofuran; H2O / 0 °C 5: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 6: p-toluenesulfonic acid / methanol / 1 h / 0 °C 7: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87920-45-4
(4E,8E)-10-Chloro-2-isopropylidene-5,9-dimethyl-deca-4,8-dienal

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: KCN, 18-crown-6 / 1 h / 0 °C 2: trimethylbenzylammonium fluoride / tetrahydrofuran; H2O / 0 °C 4: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 5: p-toluenesulfonic acid / methanol / 1 h / 0 °C 6: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87920-42-1
(4E,8E)-10-Hydroxy-2-isopropylidene-5,9-dimethyl-deca-4,8-dienoic acid methyl ester

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1: 90 percent / triphenylphosphine, carbon tetrachloride / 12 h / Heating 2: diisobutylaluminium hydride / tetrahydrofuran / -40 °C 3: manganese dioxide / hexane / Ambient temperature 4: KCN, 18-crown-6 / 1 h / 0 °C 5: trimethylbenzylammonium fluoride / tetrahydrofuran; H2O / 0 °C 7: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 8: p-toluenesulfonic acid / methanol / 1 h / 0 °C 9: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87920-43-2
(4E,8E)-10-Chloro-2-isopropylidene-5,9-dimethyl-deca-4,8-dienoic acid methyl ester

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: diisobutylaluminium hydride / tetrahydrofuran / -40 °C 2: manganese dioxide / hexane / Ambient temperature 3: KCN, 18-crown-6 / 1 h / 0 °C 4: trimethylbenzylammonium fluoride / tetrahydrofuran; H2O / 0 °C 6: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 7: p-toluenesulfonic acid / methanol / 1 h / 0 °C 8: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87920-47-6
(5E,9E)-11-Chloro-2-hydroxy-3-isopropylidene-6,10-dimethyl-undeca-5,9-dienenitrile

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 2: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 3: p-toluenesulfonic acid / methanol / 1 h / 0 °C 4: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87920-46-5
(5E,9E)-11-Chloro-3-isopropylidene-6,10-dimethyl-2-trimethylsilanyloxy-undeca-5,9-dienenitrile

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: trimethylbenzylammonium fluoride / tetrahydrofuran; H2O / 0 °C 3: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 4: p-toluenesulfonic acid / methanol / 1 h / 0 °C 5: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87920-40-9
(4E,8E)-2-Acetyl-10-(1-ethoxy-ethoxy)-5,9-dimethyl-deca-4,8-dienoic acid methyl ester

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1: 95 percent / sodium hydride / diethyl ether / 0 °C 2: 70 percent / diethyl ether / -10 °C 3: 90 percent / triphenylphosphine, carbon tetrachloride / 12 h / Heating 4: diisobutylaluminium hydride / tetrahydrofuran / -40 °C 5: manganese dioxide / hexane / Ambient temperature 6: KCN, 18-crown-6 / 1 h / 0 °C 7: trimethylbenzylammonium fluoride / tetrahydrofuran; H2O / 0 °C 9: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 10: p-toluenesulfonic acid / methanol / 1 h / 0 °C 11: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87920-48-7
(5E,9E)-11-Chloro-2-(1-ethoxy-ethoxy)-3-isopropylidene-6,10-dimethyl-undeca-5,9-dienenitrile

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 2: p-toluenesulfonic acid / methanol / 1 h / 0 °C 3: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87920-49-8
(3E,7E)-1-(1-Ethoxy-ethoxy)-10-isopropylidene-3,7-dimethyl-cyclodeca-3,7-dienecarbonitrile

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: p-toluenesulfonic acid / methanol / 1 h / 0 °C 2: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87920-41-0
(4E,8E)-2-[1-(Diethoxy-phosphoryloxy)-eth-(E)-ylidene]-10-hydroxy-5,9-dimethyl-deca-4,8-dienoic acid methyl ester

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 70 percent / diethyl ether / -10 °C 2: 90 percent / triphenylphosphine, carbon tetrachloride / 12 h / Heating 3: diisobutylaluminium hydride / tetrahydrofuran / -40 °C 4: manganese dioxide / hexane / Ambient temperature 5: KCN, 18-crown-6 / 1 h / 0 °C 6: trimethylbenzylammonium fluoride / tetrahydrofuran; H2O / 0 °C 8: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 9: p-toluenesulfonic acid / methanol / 1 h / 0 °C 10: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
87064-43-5
Acetic acid (2E,6E)-8-(1-ethoxy-ethoxy)-3,7-dimethyl-octa-2,6-dienyl ester

-
-
6902-91-6
germacrone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 12 steps 1: 65 percent / triphenylphosphine / palladium acetate / tetrahydrofuran / 5 h / Heating 2: 95 percent / sodium hydride / diethyl ether / 0 °C 3: 70 percent / diethyl ether / -10 °C 4: 90 percent / triphenylphosphine, carbon tetrachloride / 12 h / Heating 5: diisobutylaluminium hydride / tetrahydrofuran / -40 °C 6: manganese dioxide / hexane / Ambient temperature 7: KCN, 18-crown-6 / 1 h / 0 °C 8: trimethylbenzylammonium fluoride / tetrahydrofuran; H2O / 0 °C 10: 79 percent / sodium bis(trimethylsilyl)amide / tetrahydrofuran / 0.5 h / 56 °C 11: p-toluenesulfonic acid / methanol / 1 h / 0 °C 12: 2percent aqueous sodium hydroxide / diethyl ether / 0.33 h View Scheme |
-
-
6902-91-6
germacrone

-
-
90988-53-7
(3E,7E)-3,7-dimethyl-10-(1-methylethylidene)-3,7-cyclodecanedien-1-ol

| Conditions | Yield |
|---|---|
| With methanol; sodium tetrahydroborate at 0℃; for 4h; Sealed tube; | 98.3% |
| With lithium aluminium tetrahydride In tetrahydrofuran at -10℃; for 0.166667h; Inert atmosphere; | 94% |
| With sodium tetrahydroborate In methanol at 4℃; for 1h; | 83% |
-
-
6902-91-6
germacrone

-
-
20303-60-0
(4S)-trans-β-elemenone

| Conditions | Yield |
|---|---|
| With bis(benzonitrile)palladium(II) dichloride In toluene for 1h; Cope rearrangement; Reflux; Inert atmosphere; | 98% |
-
-
6902-91-6
germacrone

-
-
1443277-02-8
C15H22O

| Conditions | Yield |
|---|---|
| With sulfuric acid In chloroform at 20℃; for 1h; Temperature; Time; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With sodium ethanolate at 20℃; for 48h; Inert atmosphere; | 86% |
-
-
6902-91-6
germacrone

-
B
-
32179-17-2, 70680-71-6, 92691-35-5
(4S,5S)-(+)-germacrone 4,5-epoxide

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane for 6h; Inert atmosphere; | A 19% B 79% |
-
-
6902-91-6
germacrone

-
A
-
54707-47-0
selina-4(14),7(11)-dien-8-one

-
B
-
1443277-02-8
C15H22O

-
C
-
97530-59-1
eudesma-4,7(11)-dien-8-one

| Conditions | Yield |
|---|---|
| With sulfuric acid In chloroform at 25℃; for 0.5h; Inert atmosphere; | A 5% B 78% C 10% |

-
-
6902-91-6
germacrone

-
-
52061-45-7, 106862-05-9, 142794-64-7
1,10-epoxygermacrone

-
-
32179-17-2, 70680-71-6, 92691-35-5
(+/-)-germacrone-4,5-epoxide

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 20℃; | A 19% B 72% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In dichloromethane at 20℃; for 0.25h; Inert atmosphere; | A 53% B 19% |
-
-
6902-91-6
germacrone

-
A
-
54707-47-0
selina-4(14),7(11)-dien-8-one

-
B
-
1443277-02-8
C15H22O

-
C
-
97530-59-1
eudesma-4,7(11)-dien-8-one

| Conditions | Yield |
|---|---|
| With sulfuric acid In chloroform at 25℃; for 0.0833333h; Inert atmosphere; | A 5% B 21% C 42% D 24% |

-
-
6902-91-6
germacrone

-
A
-
5975-50-8, 92465-08-2, 92465-09-3, 108667-42-1, 116713-46-3
isogermacrone <(2Z,7E)-3,7-dimethyl-10-(1-methylethylidene)-2,7-cyclodecadien-1-one>

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; sodium hydroxide In ethanol at 20℃; for 960h; | A 41% B 20% |

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid at -78℃; for 1h; Inert atmosphere; | A 39% B 27% C 9% D 4% E 1% |
| With chlorosulfonic acid at -78℃; for 0.166667h; Inert atmosphere; | A 16% B 16% C 5% D 21% E 15% |

-
-
5707-04-0
Phenylselenyl chloride

-
-
6902-91-6
germacrone

-
A
-
1443277-05-1
C21H26OSe

-
B
-
1443277-06-2
C21H26OSe

-
C
-
1443277-08-4
C21H26OSe

-
D
-
1443277-09-5
C21H26OSe

-
E
-
1443277-07-3
C21H26OSe

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 0.5h; Inert atmosphere; | A 37% B 20% C 14% D 6% E 7% |


| Conditions | Yield |
|---|---|
| With N-chloro-succinimide In dichloromethane at 20℃; for 30h; Inert atmosphere; | A 35% B 6% C 31% |


| Conditions | Yield |
|---|---|
| With chlorosulfonic acid at -78℃; for 0.166667h; Inert atmosphere; | A 24% B 35% C 8% |

-
-
6902-91-6
germacrone

-
-
54707-46-9, 73788-48-4, 130466-24-9
6-isopropylidene-4,8aβ-dimethyl-trans-1,2,5,6,8,8a-hexahydronaphthalene-7-(4aH)-one

| Conditions | Yield |
|---|---|
| With gallium(III) trichloride In dichloromethane at 20℃; for 0.666667h; | A 15% B 24% |
-
-
6902-91-6
germacrone

-
-
546-67-8
lead(IV) tetraacetate

-
-
115857-52-8
(1R,3aR,8aR)-7-Isopropylidene-1-methoxy-1-methyl-4-methylene-octahydro-azulen-6-one

-
B
-
115842-60-9
4,7-Diacetyl-2-isopropylidene-cyclooctanone

-
-
115842-59-6, 115939-29-2
(1S,3aR,4R,8aS)-7-Isopropylidene-1,4-dimethoxy-1,4-dimethyl-octahydro-azulen-6-one

-
-
115842-59-6, 115939-29-2
(1S,3aS,4S,8aS)-7-Isopropylidene-1,4-dimethoxy-1,4-dimethyl-octahydro-azulen-6-one

| Conditions | Yield |
|---|---|
| In methanol for 0.833333h; Ambient temperature; | A 40 mg B 20% C 20 mg D 60 mg |

-
-
6902-91-6
germacrone

-
-
69239-69-6
2-isopropylidene-5,9-dimethyl-cyclodecanone

| Conditions | Yield |
|---|---|
| With hydrogen; platinum(IV) oxide |
-
-
6902-91-6
germacrone

-
-
32663-53-9
10-isopropylidene-3,7-dimethyl-cyclodeca-3t,7t-dienone

| Conditions | Yield |
|---|---|
| With acetophenone In diethyl ether Irradiation; | |
| With acetophenone Irradiation; |
-
-
6902-91-6
germacrone

-
-
32663-51-7
3,7-dimethyl-10-isopropylidene-3(Z),7(E)-cyclodecadien-1-one

| Conditions | Yield |
|---|---|
| With acetophenone In diethyl ether Irradiation; | |
| With acetophenone Irradiation; |
-
-
6902-91-6
germacrone

-
-
470-19-9, 20303-60-0, 30824-86-3, 32663-57-3, 52153-83-0
(+/-)-4r-isopropenyl-2-isopropylidene-5c-methyl-5t-vinyl-cyclohexanone

| Conditions | Yield |
|---|---|
| at 140 - 148℃; |
-
-
67-56-1
methanol

-
-
6902-91-6
germacrone

-
-
115857-52-8
(1R,3aR,8aR)-7-Isopropylidene-1-methoxy-1-methyl-4-methylene-octahydro-azulen-6-one

-
B
-
115842-60-9
4,7-Diacetyl-2-isopropylidene-cyclooctanone

-
-
115842-59-6, 115939-29-2
(1S,3aR,4R,8aS)-7-Isopropylidene-1,4-dimethoxy-1,4-dimethyl-octahydro-azulen-6-one

-
-
115842-59-6, 115939-29-2
(1S,3aS,4S,8aS)-7-Isopropylidene-1,4-dimethoxy-1,4-dimethyl-octahydro-azulen-6-one

| Conditions | Yield |
|---|---|
| With lead(IV) acetate for 0.833333h; Ambient temperature; | A 40 mg B 40 mg C 20 mg D 60 mg |

-
-
75-11-6
diiodomethane

-
-
6902-91-6
germacrone

-
-
96235-08-4
4,5-cyclopropane-8-oxo-germacrene B

-
-
96235-09-5
1,10-cyclopropane-8-oxo-germacrene B

| Conditions | Yield |
|---|---|
| With copper(l) chloride; zinc In diethyl ether at 30℃; for 24h; |

-
-
6902-91-6
germacrone

-
-
546-67-8
lead(IV) tetraacetate

-
A
-
116046-02-7
2-hydroxy-4-methyl-ω,ω-dichloroacetophenone

-
-
115842-58-5
Acetic acid (1R,8aR)-7-isopropylidene-1,4-dimethyl-6-oxo-1,2,3,5,6,7,8,8a-octahydro-azulen-1-yl ester

-
-
115842-51-8
Acetic acid (3aR,4R)-7-isopropylidene-1,4-dimethyl-6-oxo-2,3,3a,4,5,6,7,8-octahydro-azulen-4-yl ester

-
-
98644-26-9, 115936-78-2
Acetic acid (1S,3aS,4S,8aS)-1-acetoxy-7-isopropylidene-1,4-dimethyl-6-oxo-decahydro-azulen-4-yl ester

| Conditions | Yield |
|---|---|
| In acetic acid for 2h; Ambient temperature; Further byproducts given; | A 50 mg B 25 mg C 25 mg D 16 mg |

-
-
6902-91-6
germacrone

-
-
546-67-8
lead(IV) tetraacetate

-
-
115842-54-1
(3aR,3bR,7aS)-6-Isopropylidene-1,3b-dimethyl-3,3a,3b,4,6,7-hexahydro-cyclopenta[1,3]cyclopropa[1,2]benzen-5-one

-
-
115842-48-3
(3aS,8aS)-5-Isopropylidene-3-methyl-8-methylene-3a,4,5,7,8,8a-hexahydro-1H-azulen-6-one

-
-
115842-47-2
(3aS,8aS)-5-Isopropylidene-3,8-dimethyl-3a,4,5,8a-tetrahydro-1H-azulen-6-one

-
-
115842-49-4
Acetic acid (3aR,4R,8aR)-7-isopropylidene-1,4-dimethyl-6-oxo-3,3a,4,5,6,7,8,8a-octahydro-azulen-4-yl ester

| Conditions | Yield |
|---|---|
| In benzene for 18h; Ambient temperature; Further byproducts given; | A 3 mg B 15 mg C 18 mg D 75 mg |

-
-
6902-91-6
germacrone

-
-
546-67-8
lead(IV) tetraacetate

-
-
115842-48-3
(3aS,8aS)-5-Isopropylidene-3-methyl-8-methylene-3a,4,5,7,8,8a-hexahydro-1H-azulen-6-one

-
-
115842-47-2
(3aS,8aS)-5-Isopropylidene-3,8-dimethyl-3a,4,5,8a-tetrahydro-1H-azulen-6-one

-
-
115842-52-9
Acetic acid (4aR,8aS)-6-isopropylidene-4,8a-dimethyl-7-oxo-1,2,4a,5,6,7,8,8a-octahydro-naphthalen-1-yl ester

-
-
115842-50-7
Acetic acid (3aR,4R,8aR)-7-isopropylidene-4-methyl-1-methylene-6-oxo-decahydro-azulen-4-yl ester

-
-
115842-49-4
Acetic acid (3aR,4R,8aR)-7-isopropylidene-1,4-dimethyl-6-oxo-3,3a,4,5,6,7,8,8a-octahydro-azulen-4-yl ester

-
-
115842-51-8
Acetic acid (3aR,4R)-7-isopropylidene-1,4-dimethyl-6-oxo-2,3,3a,4,5,6,7,8-octahydro-azulen-4-yl ester

| Conditions | Yield |
|---|---|
| In benzene for 18h; Mechanism; Product distribution; Ambient temperature; different solvents, reaction time; | A 15 mg B 18 mg C 9 mg D 12 mg E 75 mg F 9 mg |

-
-
6902-91-6
germacrone

-
-
546-67-8
lead(IV) tetraacetate

-
-
115842-48-3
(3aS,8aS)-5-Isopropylidene-3-methyl-8-methylene-3a,4,5,7,8,8a-hexahydro-1H-azulen-6-one

-
-
115842-47-2
(3aS,8aS)-5-Isopropylidene-3,8-dimethyl-3a,4,5,8a-tetrahydro-1H-azulen-6-one

-
-
115842-52-9
Acetic acid (4aR,8aS)-6-isopropylidene-4,8a-dimethyl-7-oxo-1,2,4a,5,6,7,8,8a-octahydro-naphthalen-1-yl ester

-
-
115842-49-4
Acetic acid (3aR,4R,8aR)-7-isopropylidene-1,4-dimethyl-6-oxo-3,3a,4,5,6,7,8,8a-octahydro-azulen-4-yl ester

| Conditions | Yield |
|---|---|
| In benzene for 18h; Ambient temperature; Further byproducts given; | A 15 mg B 18 mg C 9 mg D 75 mg |

-
-
6902-91-6
germacrone

-
-
546-67-8
lead(IV) tetraacetate

-
-
115842-48-3
(3aS,8aS)-5-Isopropylidene-3-methyl-8-methylene-3a,4,5,7,8,8a-hexahydro-1H-azulen-6-one

-
-
115842-47-2
(3aS,8aS)-5-Isopropylidene-3,8-dimethyl-3a,4,5,8a-tetrahydro-1H-azulen-6-one

-
-
115842-50-7
Acetic acid (3aR,4R,8aR)-7-isopropylidene-4-methyl-1-methylene-6-oxo-decahydro-azulen-4-yl ester

-
-
115842-49-4
Acetic acid (3aR,4R,8aR)-7-isopropylidene-1,4-dimethyl-6-oxo-3,3a,4,5,6,7,8,8a-octahydro-azulen-4-yl ester

| Conditions | Yield |
|---|---|
| In benzene for 18h; Ambient temperature; Further byproducts given; | A 15 mg B 18 mg C 12 mg D 75 mg |

-
-
6902-91-6
germacrone

-
-
546-67-8
lead(IV) tetraacetate

-
-
115842-48-3
(3aS,8aS)-5-Isopropylidene-3-methyl-8-methylene-3a,4,5,7,8,8a-hexahydro-1H-azulen-6-one

-
-
115842-47-2
(3aS,8aS)-5-Isopropylidene-3,8-dimethyl-3a,4,5,8a-tetrahydro-1H-azulen-6-one

-
-
115842-49-4
Acetic acid (3aR,4R,8aR)-7-isopropylidene-1,4-dimethyl-6-oxo-3,3a,4,5,6,7,8,8a-octahydro-azulen-4-yl ester

-
-
115842-56-3
Acetic acid (2E,6E)-(1S,5S)-5-acetoxy-8-isopropylidene-1,5-dimethyl-9-oxo-cyclodeca-2,6-dienyl ester

| Conditions | Yield |
|---|---|
| In benzene for 18h; Ambient temperature; Further byproducts given; | A 15 mg B 18 mg C 75 mg D 3 mg |

-
-
6902-91-6
germacrone

-
-
546-67-8
lead(IV) tetraacetate

-
-
115842-48-3
(3aS,8aS)-5-Isopropylidene-3-methyl-8-methylene-3a,4,5,7,8,8a-hexahydro-1H-azulen-6-one

-
-
115842-49-4
Acetic acid (3aR,4R,8aR)-7-isopropylidene-1,4-dimethyl-6-oxo-3,3a,4,5,6,7,8,8a-octahydro-azulen-4-yl ester

-
-
115842-58-5
Acetic acid (1R,8aR)-7-isopropylidene-1,4-dimethyl-6-oxo-1,2,3,5,6,7,8,8a-octahydro-azulen-1-yl ester

-
-
115842-51-8
Acetic acid (3aR,4R)-7-isopropylidene-1,4-dimethyl-6-oxo-2,3,3a,4,5,6,7,8-octahydro-azulen-4-yl ester

| Conditions | Yield |
|---|---|
| In acetic acid for 2h; Ambient temperature; Further byproducts given; | A 16 mg B 50 mg C 25 mg D 25 mg |

-
-
6902-91-6
germacrone

-
-
546-67-8
lead(IV) tetraacetate

-
-
115842-57-4
Acetic acid (1R,3aR,8aR)-7-isopropylidene-1-methyl-4-methylene-6-oxo-decahydro-azulen-1-yl ester

-
-
115842-49-4
Acetic acid (3aR,4R,8aR)-7-isopropylidene-1,4-dimethyl-6-oxo-3,3a,4,5,6,7,8,8a-octahydro-azulen-4-yl ester

-
-
115842-58-5
Acetic acid (1R,8aR)-7-isopropylidene-1,4-dimethyl-6-oxo-1,2,3,5,6,7,8,8a-octahydro-azulen-1-yl ester

-
-
115842-51-8
Acetic acid (3aR,4R)-7-isopropylidene-1,4-dimethyl-6-oxo-2,3,3a,4,5,6,7,8-octahydro-azulen-4-yl ester

| Conditions | Yield |
|---|---|
| In acetic acid for 2h; Ambient temperature; Further byproducts given; | A 16 mg B 50 mg C 25 mg D 25 mg |

(E,E)-Germacra-3,7(11),9-trien-6-one Chemical Properties
IUPAC Name: (3E,7E)-3,7-Dimethyl-10-propan-2-ylidenecyclodeca-3,7-dien-1-one
CAS: 6902-91-6
Formula : C15H22O
Molecular Weight: 218.33458
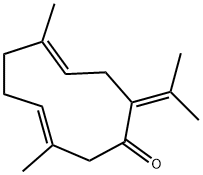
Molecular Structure of (3E,7E)-3,7-Dimethyl-10-propan-2-ylidenecyclodeca-3,7-dien-1-one (6902-91-6):
Density: 0.916 g/cm3
Flash Point: 148.4 °C
Melting Point: 55-58 °C
Boiling Point: 330.3 °C at 760 mmHg
Index of Refraction: 1.485
Molar Refractivity: 68.34 cm3
Molar Volume: 238.3 cm3
Polarizability: 27.09×10-24cm3
Surface Tension: 29.2 dyne/cm
Appearance: fanit yellow crystal
Enthalpy of Vaporization: 57.28 kJ/mol
Vapour Pressure: 0.000168 mmHg at 25°C
Water Solubility: 0.8354(mg/L)at 25°C
(E,E)-Germacra-3,7(11),9-trien-6-one Toxicity Data With Reference
| 1. | orl-mus TDLo:25 mg/kg | BIPBU* Biological & pharmaceutical bulletin. 25 (2002),627. |
(E,E)-Germacra-3,7(11),9-trien-6-one Safety Profile
A poison by ingestion. When heated to decomposition it emits acrid smoke and irritating vapors.
Safety Information about (3E,7E)-3,7-Dimethyl-10-propan-2-ylidenecyclodeca-3,7-dien-1-one (6902-91-6):
S23: Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer).
S24/25: Avoid contact with skin and eyes.
(E,E)-Germacra-3,7(11),9-trien-6-one Specification
The chemical synonyms of (3E,7E)-3,7-Dimethyl-10-propan-2-ylidenecyclodeca-3,7-dien-1-one (6902-91-6) are (3Z,7Z)-3,7-Dimethyl-10-propan-2-ylidene-cyclodeca-3,7-dien-1-one ; (E,E)-Germacra-3,7(11),9-trien-6-one ; (E,E)-3,7-Dimethyl-10-(1-methylethylidene)-3,7-cyclodecadien-1-one ; 3,7-Cyclodecadien-1-one, 3,7-dimethyl-10-(1-methylethylidene)-, (E,E)- ; 3,7-Dimethyl-10-(1-methylethylidene)-3,7-cyclodecadien-1-one ; Germacra-3,7(11),9-trien-6-one, (E,E)- ; Germacron ; Trans,trans-3,7-Dimethyl-10-isopropylidene-3,7-cyclodecadien-1-one .
Related Products
- (E)-10-Hydroxy-2-decenoic acid
- (E)-2-Bromo-4,4,4-trifluoro-2-butenoic acid ethyl ester
- (E)-2-Butenenitrile
- (E)-2-Butenoyl chloride
- (E)-2-Cyano-3-(3,4-dihydroxyphenyl)-2-propenoic acid
- (E)-2-Cyano-3-(4-methylphenyl)prop-2-enoate
- (E)-2-Hexadecenoic acid
- (E)-2-Hexadecenoic acid ethyl ester
- (E)-2-Methyl-2-pentenal
- (E)-2-Octadecenol
- 69029-35-2
- 69029-71-6
- 69029-72-7
- 69029-73-8
- 6903-14-6
- 6903-18-0
- 69033-81-4
- 69033-92-7
- 69034-12-4
- 69037-87-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View