-
Name
1,7-Heptanediol
- EINECS 211-085-0
- CAS No. 629-30-1
- Article Data230
- CAS DataBase
- Density 0.951 g/cm3
- Solubility Soluble in water, slightly soluble in ether.
- Melting Point 17-19 °C(lit.)
- Formula C7H16O2
- Boiling Point 262 °C at 760 mmHg
- Molecular Weight 132.203
- Flash Point 120.9 °C
- Transport Information
- Appearance yellowish liquid
- Safety 24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 1,7-Dihydroxyheptane;NSC 3821;a,w-Heptanediol;w-Heptanediol;
- PSA 40.46000
- LogP 0.92150
Synthetic route

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; for 0.5h; | 100% |
-
-
766-77-8
Dimethylphenylsilane

-
-
81036-11-5
7-(2-tetrahydro-2H-pyranyloxy)heptan-1-ol

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane at 0℃; chemoselective reaction; | 100% |
-
-
81036-11-5
7-(2-tetrahydro-2H-pyranyloxy)heptan-1-ol

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With acide phosphorique at 180 - 200℃; | 98.6% |

| Conditions | Yield |
|---|---|
| With sodium bis(2-methoxyethoxy)aluminium dihydride In 2-methyltetrahydrofuran at 45℃; for 5h; Inert atmosphere; | 98% |
| With samarium diiodide; water; triethylamine In tetrahydrofuran at 20℃; for 20h; Inert atmosphere; | 87% |
| With ethanol; sodium |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 5 - 20℃; for 2.5h; Large scale; | 95.3% |
| With ethanol; sodium | |
| With sodium; butan-1-ol | |
| With lithium aluminium tetrahydride; diethyl ether |

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With carbon tetrabromide In methanol for 3h; Deacetylation; ether cleavage; Heating; | 74% |
-
-
1192601-85-6
7-hydroxyheptyl phenyl telluride

-
A
-
629-30-1
1,7-heptandiol

-
B
-
4117-10-6
6-hepten-1-ol

-
C
-
22054-13-3
7-hydroxyheptanal

| Conditions | Yield |
|---|---|
| With oxygen In hexane for 0.0333333h; UV-irradiation; | A 16 %Chromat. B 6 %Chromat. C 72% |

-
-
6485-79-6
chlorotriisopropylsilane

-
-
81036-11-5
7-(2-tetrahydro-2H-pyranyloxy)heptan-1-ol

-
A
-
629-30-1
1,7-heptandiol

-
B
-
859509-85-6
7-((triisopropylsilyl)oxy)heptan-1-ol

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In toluene at 0℃; chemoselective reaction; | A n/a B 58% |


| Conditions | Yield |
|---|---|
| Stage #1: 1,5-dibromo-pentane With magnesium In tetrahydrofuran at 200℃; for 2h; Inert atmosphere; Stage #2: formaldehyd In tetrahydrofuran for 12h; | 50% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; samarium diiodide In tetrahydrofuran; water for 0.01h; Ambient temperature; | 25% |
| With lithium aluminium tetrahydride; diethyl ether | |
| With copper-aluminium-zinc catalyst at 280℃; under 147102 Torr; Hydrogenation; | |
| Multi-step reaction with 2 steps 1: concentrated sulfuric acid 2: sodium; alcohol View Scheme | |
| Multi-step reaction with 2 steps 1: toluene-4-sulfonic acid / 7 h / Reflux; Large scale 2: lithium aluminium tetrahydride / tetrahydrofuran / 2.5 h / 5 - 20 °C / Large scale View Scheme |

| Conditions | Yield |
|---|---|
| With hydrogen In butan-1-ol at 150℃; under 7500.75 Torr; for 4h; Autoclave; | 9% |

| Conditions | Yield |
|---|---|
| With hydrogen In butan-1-ol at 150℃; under 7500.75 Torr; for 8h; Autoclave; | A 7% B 6% |
-
-
137333-33-6
1,7-dimethoxyheptane

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With hydrogen iodide |

| Conditions | Yield |
|---|---|
| With silver(I) acetate Destillierung das Diacetat ueber Natronkalk; |
-
-
84143-36-2
3-heptene-1,7-diol

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| Hydrogenation; |
-
-
102547-94-4
hept-3-yne-1,7-diol

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With methanol; nickel under 22065.2 - 36775.4 Torr; Hydrogenation; |
-
-
503-30-0
trimethylene oxide

-
-
58766-02-2
4-<(tetrahydropyran-2-yl)oxy>butylmagnesium chloride

-
-
629-30-1
1,7-heptandiol
-
-
66997-43-1
2,8-dihydroxy-octanoic acid

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With chromium(VI) oxide |

| Conditions | Yield |
|---|---|
| With lithium triethylborohydride In various solvent(s) for 0.416667h; Thermodynamic data; ΔH; |
-
-
3070-53-9
1,6-heptadiene

-
A
-
629-30-1
1,7-heptandiol

-
B
-
60096-09-5
heptane-1,5-diol

-
C
-
23418-82-8
cis-1,5-cyclooctanediol

| Conditions | Yield |
|---|---|
| With 9-borabicyclo[3.3.1]nonane dimer; dimethylsulfide borane complex Product distribution; |


-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With copper chromite; ethanol at 200℃; under 73550.8 - 147102 Torr; Hydrogenation; |

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With copper oxide-chromium oxide at 210℃; under 760000 Torr; Hydrogenation; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) magnesium / 1.) THF, reflux, 1.5 h, 2.) reflux, 1 h. 2: 98.6 percent / acide phosphorique / 180 - 200 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 2: 1.) magnesium / 1.) THF, reflux, 1.5 h, 2.) reflux, 1 h. 3: 98.6 percent / acide phosphorique / 180 - 200 °C View Scheme | |
| Multi-step reaction with 2 steps 1: (i) /BRN= 4652394/, DMSO, (ii) H2, Raney-Ni, semicarbazide*HCl, aq. EtOH, (iii) /BRN= 4652394/, aq. HCl, dioxane, (iv) (heating) 2: CrO3 View Scheme |
-
-
21890-96-0
7-(tetrahydro-2H-pyran-2-yloxy)hept-3-yn-1-ol

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: methanol; toluene-4-sulfonic acid 2: Raney nickel; methanol / 22065.2 - 36775.4 Torr / Hydrogenation View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: phosphoryl chloride; sodium amide; liquid ammonia / Reagens Nr. 4: Aether 2: methanol; toluene-4-sulfonic acid 3: Raney nickel; methanol / 22065.2 - 36775.4 Torr / Hydrogenation View Scheme |
-
-
60579-50-2
B-methoxyborocane

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water oxidation with alkaline H2O2;; |

| Conditions | Yield |
|---|---|
| Stage #1: C6H11OPol; carbon monoxide With acetylacetonatodicarbonylrhodium(l); hydrogen In toluene at 60℃; under 30003 Torr; for 24h; Inert atmosphere; Autoclave; solid phase reaction; Stage #2: With sodium tetrahydroborate Inert atmosphere; solid phase reaction; Stage #3: With trifluoroacetic acid In dichloromethane at 20℃; for 0.166667h; Inert atmosphere; solid phase reaction; |


| Conditions | Yield |
|---|---|
| With Ethyl trichloroacetate; triphenylphosphine In acetonitrile at 10 - 15℃; for 3h; | 100% |
| With pyridine; thionyl chloride | |
| (i) COCl2, (ii) Ph3P; Multistep reaction; | |
| With pyridine; thionyl chloride |

| Conditions | Yield |
|---|---|
| A 100% B n/a |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; dimethyl sulfoxide at 100℃; under 760 Torr; for 5h; | 99% |
| With sodium hydroxide; cesium fluoride In dimethyl sulfoxide at 100℃; for 2.5h; atmospheric pressure; | 87% |

| Conditions | Yield |
|---|---|
| With iron(III) chloride; 1-(n-butyl)-3-methylimidazolium triflate at 40℃; for 3.16667h; Ionic liquid; | 99% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 15h; Steglich Esterification; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With C24H33IrN4O3; water; sodium hydroxide for 18h; Reflux; | 97% |

| Conditions | Yield |
|---|---|
| With hafnium(IV) trifluoromethanesulfonate In toluene at 110℃; for 48h; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With 1-(ω-sulfonic acid)propyl-3-methylimidazolium bromide at 100℃; Inert atmosphere; | 95% |
| With dibromotriphenylphosphorane | 89% |
| With phosphorus tribromide; triethylamine In diethyl ether at 0℃; | 48% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; Inert atmosphere; | 95% |
-
-
629-30-1
1,7-heptandiol

-
-
109216-50-4
(1S,5R,7S)-1,5,7-Trimethyl-2,4-dioxo-3-aza-bicyclo[3.3.1]nonane-7-carbonyl chloride

-
-
129786-97-6
C31H46N2O8

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane Heating; | 94% |

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; for 0.5h; | 100% |
-
-
766-77-8
Dimethylphenylsilane

-
-
81036-11-5
7-(2-tetrahydro-2H-pyranyloxy)heptan-1-ol

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane at 0℃; chemoselective reaction; | 100% |
-
-
81036-11-5
7-(2-tetrahydro-2H-pyranyloxy)heptan-1-ol

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With acide phosphorique at 180 - 200℃; | 98.6% |

| Conditions | Yield |
|---|---|
| With sodium bis(2-methoxyethoxy)aluminium dihydride In 2-methyltetrahydrofuran at 45℃; for 5h; Inert atmosphere; | 98% |
| With samarium diiodide; water; triethylamine In tetrahydrofuran at 20℃; for 20h; Inert atmosphere; | 87% |
| With ethanol; sodium |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 5 - 20℃; for 2.5h; Large scale; | 95.3% |
| With ethanol; sodium | |
| With sodium; butan-1-ol | |
| With lithium aluminium tetrahydride; diethyl ether |

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With carbon tetrabromide In methanol for 3h; Deacetylation; ether cleavage; Heating; | 74% |
-
-
1192601-85-6
7-hydroxyheptyl phenyl telluride

-
A
-
629-30-1
1,7-heptandiol

-
B
-
4117-10-6
6-hepten-1-ol

-
C
-
22054-13-3
7-hydroxyheptanal

| Conditions | Yield |
|---|---|
| With oxygen In hexane for 0.0333333h; UV-irradiation; | A 16 %Chromat. B 6 %Chromat. C 72% |

-
-
6485-79-6
chlorotriisopropylsilane

-
-
81036-11-5
7-(2-tetrahydro-2H-pyranyloxy)heptan-1-ol

-
A
-
629-30-1
1,7-heptandiol

-
B
-
859509-85-6
7-((triisopropylsilyl)oxy)heptan-1-ol

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In toluene at 0℃; chemoselective reaction; | A n/a B 58% |


| Conditions | Yield |
|---|---|
| Stage #1: 1,5-dibromo-pentane With magnesium In tetrahydrofuran at 200℃; for 2h; Inert atmosphere; Stage #2: formaldehyd In tetrahydrofuran for 12h; | 50% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; samarium diiodide In tetrahydrofuran; water for 0.01h; Ambient temperature; | 25% |
| With lithium aluminium tetrahydride; diethyl ether | |
| With copper-aluminium-zinc catalyst at 280℃; under 147102 Torr; Hydrogenation; | |
| Multi-step reaction with 2 steps 1: concentrated sulfuric acid 2: sodium; alcohol View Scheme | |
| Multi-step reaction with 2 steps 1: toluene-4-sulfonic acid / 7 h / Reflux; Large scale 2: lithium aluminium tetrahydride / tetrahydrofuran / 2.5 h / 5 - 20 °C / Large scale View Scheme |

| Conditions | Yield |
|---|---|
| With hydrogen In butan-1-ol at 150℃; under 7500.75 Torr; for 4h; Autoclave; | 9% |

| Conditions | Yield |
|---|---|
| With hydrogen In butan-1-ol at 150℃; under 7500.75 Torr; for 8h; Autoclave; | A 7% B 6% |
-
-
137333-33-6
1,7-dimethoxyheptane

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With hydrogen iodide |

| Conditions | Yield |
|---|---|
| With silver(I) acetate Destillierung das Diacetat ueber Natronkalk; |
-
-
84143-36-2
3-heptene-1,7-diol

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| Hydrogenation; |
-
-
102547-94-4
hept-3-yne-1,7-diol

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With methanol; nickel under 22065.2 - 36775.4 Torr; Hydrogenation; |
-
-
503-30-0
trimethylene oxide

-
-
58766-02-2
4-<(tetrahydropyran-2-yl)oxy>butylmagnesium chloride

-
-
629-30-1
1,7-heptandiol
-
-
66997-43-1
2,8-dihydroxy-octanoic acid

-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With chromium(VI) oxide |

| Conditions | Yield |
|---|---|
| With lithium triethylborohydride In various solvent(s) for 0.416667h; Thermodynamic data; ΔH; |
-
-
3070-53-9
1,6-heptadiene

-
A
-
629-30-1
1,7-heptandiol

-
B
-
60096-09-5
heptane-1,5-diol

-
C
-
23418-82-8
cis-1,5-cyclooctanediol

| Conditions | Yield |
|---|---|
| With 9-borabicyclo[3.3.1]nonane dimer; dimethylsulfide borane complex Product distribution; |


-
-
629-30-1
1,7-heptandiol

| Conditions | Yield |
|---|---|
| With copper chromite; ethanol at 200℃; under 73550.8 - 147102 Torr; Hydrogenation; |
1,7-Heptanediol Specification
The 1,7-Heptanediol, also known as 1,7-Dihydroxyheptane, is an organic compound with the formula C7H16O2. It belongs to the product categories of alpha,omega-Alkanediols; alpha,omega-Bifunctional Alkanes; Monofunctional & alpha,omega-Bifunctional Alkanes; Linear Hydrocarbon Series; Intermediates. Its EINECS registry number is 211-085-0. With the CAS registry number 629-30-1, its IUPAC name is heptane-1,7-diol. When you are using this chemical, please be cautious about it. You must avoid contact with skin and eyes. What's more, it should be protected from the oxidant, reducing agent, acidic chloride, anhydride and chlorine formic acid ester.
Physical properties of 1,7-Heptanediol: (1)ACD/LogP: 0.46; (2)ACD/LogD (pH 5.5): 0.46; (3)ACD/LogD (pH 7.4): 0.46; (4)ACD/BCF (pH 5.5): 1.33; (5)ACD/BCF (pH 7.4): 1.33; (6)ACD/KOC (pH 5.5): 42.58; (7)ACD/KOC (pH 7.4): 42.58; (8)#H bond acceptors: 2; (9)#H bond donors: 2; (10)#Freely Rotating Bonds: 8; (11)Index of Refraction: 1.452; (12)Molar Refractivity: 37.54 cm3; (13)Molar Volume: 139 cm3; (14)Surface Tension: 37.4 dyne/cm; (15)Density: 0.95 g/cm3; (16)Flash Point: 120.9 °C; (17)Enthalpy of Vaporization: 58.04 kJ/mol; (18)Boiling Point: 262 °C at 760 mmHg; (19)Vapour Pressure: 0.00159 mmHg at 25°C.
Preparation of 1,7-Heptanediol: this chemical can be prepared by 7-tetrahydropyranyloxy-heptan-1-ol. This reaction will need reagent acide phosphorique. The reaction occurs at 180 - 200 °C. The yield is about 98.6%.
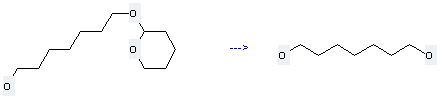
Uses of 1,7-Heptanediol: it can be used to produce 7-chloro-heptan-1-ol at 95 °C. This reaction will need reagent concentrated HCl.

You can still convert the following datas into molecular structure:
(1)Canonical SMILES: C(CCCO)CCCO
(2)InChI: InChI=1S/C7H16O2/c8-6-4-2-1-3-5-7-9/h8-9H,1-7H2
(3)InChIKey: SXCBDZAEHILGLM-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD | intraperitoneal | > 500mg/kg (500mg/kg) | "Summary Tables of Biological Tests," National Research Council Chemical-Biological Coordination Center. Vol. 6, Pg. 147, 1954. |
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 62932-92-7
- 62932-93-8
- 62933-29-3
- 629-33-4
- 62934-94-5
- 6293-56-7
- 62935-72-2
- 6293-61-4
- 62936-23-6
- 62936-31-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View