-
Name
2,5-Dibromoterephthalic acid
- EINECS 237-300-8
- CAS No. 13731-82-3
- Article Data33
- CAS DataBase
- Density 2.205g/cm3
- Solubility
- Melting Point 315-318 ºC
- Formula C8H4 Br2 O4
- Boiling Point 445ºC at 760 mmHg
- Molecular Weight 323.925
- Flash Point 222.9°C
- Transport Information
- Appearance
- Safety S26;S37/39
- Risk Codes R36/37/38
-
Molecular Structure
-
Hazard Symbols

- Synonyms Terephthalicacid, 2,5-dibromo- (6CI,7CI,8CI); 1,4-Dibromo-2,5-benzenedicarboxylic acid;2,5-Dibromo-4-carboxybenzoic acid; 2,5-Dibromoterephthalic acid; NSC 115430
- PSA 74.60000
- LogP 2.60800
Synthetic route
-
-
1074-24-4
2,5-dibromo-p-xylene

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| With pyridine; potassium permanganate In water | 96% |
| With pyridine; potassium permanganate In water for 24h; Reflux; | 91% |
| With chromium(VI) oxide; sulfuric acid In acetic acid at 20℃; for 24h; | 90% |
-
-
20871-01-6
2,5-dibromo-4-methylbenzoic acid

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| With potassium permanganate; sodium hydrogencarbonate for 144h; Heating; | 94% |
| With potassium permanganate In water for 144h; Reflux; | 86% |
| With potassium permanganate; water for 144h; Reflux; | 86% |
| With alkaline permanganate | |
| With potassium permanganate; potassium carbonate |
-
-
18870-11-6
2,5-dibromobenzene-1,4-dinitrile

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide |

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid at 120 - 130℃; |

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| With nitric acid at 180℃; im geschlossenen Rohr; |
-
-
67-56-1
methanol

-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
18014-00-1
dimethyl 2,5-dibromoterephthalate

| Conditions | Yield |
|---|---|
| With thionyl chloride for 5h; Reflux; | 100% |
| With sulfuric acid Heating; | 95% |
| With sulfuric acid Heating; Reflux; | 95% |
-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
13815-90-2
3,6-dibromo-2,5-phenylenedi(carboxylic acid chloride)

| Conditions | Yield |
|---|---|
| With oxalyl dichloride; N,N-dimethyl-formamide In benzene at 80℃; | 100% |
| With oxalyl dichloride | 100% |
| With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane for 3h; Inert atmosphere; Reflux; | 100% |
-
-
743460-48-2
biphenyl-2-ylcarbamic acid 1-[2-(methylamino)ethyl]piperidin-4-yl ester

-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
864751-39-3
N-{2-[4-(Biphenyl-2-ylcarbamoyloxy)piperidin-1-yl]ethyl}-2,5-dibromo-N-methylterephthalamic Acid

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; HATU In DMF (N,N-dimethyl-formamide) at 20℃; for 3h; | 100% |
-
-
64-17-5
ethanol

-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
18013-97-3
2,5-dibromo-terephthalic acid diethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: 2,5-dibromoterephtalic acid With thionyl chloride for 4h; Reflux; Stage #2: ethanol In tetrahydrofuran for 10h; | 99% |
| sulfuric acid Reflux; | 96% |
| With sulfuric acid for 48h; Reflux; | 96% |
-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
1401211-90-2
N-(2-hexyldecyl)thiophene-3-amine

| Conditions | Yield |
|---|---|
| Stage #1: 2,5-dibromoterephtalic acid With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane at 20℃; for 20h; Schlenk technique; Inert atmosphere; Stage #2: N-(2-hexyldecyl)thiophene-3-amine With triethylamine In dichloromethane at 0 - 20℃; for 20h; | 99% |
-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
610-92-4
2,5-dihydroxy-1,4-benzenedicarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2,5-dibromoterephtalic acid With water; sodium carbonate; trans-N,N'-dimethylcyclohexane-1,2-diamine; copper(I) bromide In acetonitrile at 90℃; for 18h; Stage #2: With hydrogenchloride In water; acetonitrile at 25℃; Product distribution / selectivity; | 98% |
| Stage #1: 2,5-dibromoterephtalic acid With water; sodium carbonate for 1h; Heating / reflux; Stage #2: With water; trans-N,N'-dimethylcyclohexane-1,2-diamine; copper(I) bromide at 90℃; for 2h; Stage #3: With hydrogenchloride In water at 25℃; Product distribution / selectivity; | 97% |
| Stage #1: 2,5-dibromoterephtalic acid With water; sodium carbonate for 1h; Heating / reflux; Stage #2: 2,2,6,6-tetramethylheptane-3,5-dione; copper(I) bromide In water at 80℃; for 30h; Stage #3: With hydrogenchloride In water at 25℃; | 92% |

| Conditions | Yield |
|---|---|
| (±)-N,N-dimethyl-trans-1,2-diaminocyclohexane; copper(ll) bromide In methanol for 8h; Heating / reflux; | 95% |
| 2,2,6,6-tetramethylheptane-3,5-dione; copper(ll) bromide In methanol for 8h; Heating / reflux; | |
| 1,2-bis[(2,4,6-trimethylphenyl)imino]-1,2-dimethylethane; copper(ll) bromide In methanol for 8h; Heating / reflux; |
-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
50880-39-2
2,5-dibromobenzene-1,4-carboxamide

| Conditions | Yield |
|---|---|
| Stage #1: 2,5-dibromoterephtalic acid With thionyl chloride; N,N-dimethyl-formamide for 3h; Inert atmosphere; Reflux; Stage #2: With ammonium hydroxide In 1,4-dioxane for 1h; | 94% |
| Multi-step reaction with 2 steps 1: PCl5 2: diethyl ether; ammonia View Scheme | |
| Multi-step reaction with 2 steps 1: thionyl chloride; N,N-dimethyl-formamide / 3 h / Reflux 2: ammonium hydroxide / 1,4-dioxane / 20 °C View Scheme |
-
-
13731-82-3
2,5-dibromoterephtalic acid

-
A
-
636-94-2
2-hydroxyterephthalic acid

-
B
-
610-92-4
2,5-dihydroxy-1,4-benzenedicarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2,5-dibromoterephtalic acid With sodium carbonate In water for 1h; Heating / reflux; Stage #2: With water; 2,2,6,6-tetramethylheptane-3,5-dione; copper(I) bromide at 80℃; for 30h; Stage #3: With hydrogenchloride In water at 25℃; | A n/a B 92% |
-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
71-36-3
butan-1-ol

-
-
58613-36-8
dibutyl 2,5-dibromoterephthalate

| Conditions | Yield |
|---|---|
| With sulfuric acid at 120℃; for 12h; | 89% |
| With sulfuric acid for 15h; | 81% |

-
-
83524-76-9
4,4'-methylenebis(3,5-dimethylpyrazole)

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| In water at 120℃; for 24h; Autoclave; | 82% |


-
-
83524-76-9
4,4'-methylenebis(3,5-dimethylpyrazole)

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| In water at 120℃; for 48h; Autoclave; | 79% |

-
-
64-18-6
formic acid

-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| at 150℃; for 24h; Autoclave; High pressure; | 78.2% |

-
-
76-37-9
2,2,3,3-tetrafluoropropanol

-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
1268628-20-1
2,5-bis(2,2,3,3-tetrafluoropropoxy)terephthalic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2,2,3,3-tetrafluoropropanol With sodium hydride In tetrahydrofuran Stage #2: 2,5-dibromoterephtalic acid; cis-N,N'-dimethyl-1,2-diaminocyclohexane; copper(ll) bromide In tetrahydrofuran; 2,2,3,3-tetrafluoropropanol at 60℃; for 48h; Stage #3: With hydrogenchloride In tetrahydrofuran; water | 72% |
| Stage #1: 2,2,3,3-tetrafluoropropanol With sodium hydride In tetrahydrofuran Stage #2: 2,5-dibromoterephtalic acid; cis-N,N'-dimethyl-1,2-diaminocyclohexane; copper(ll) bromide In tetrahydrofuran; 2,2,3,3-tetrafluoropropanol at 60℃; for 48h; Stage #3: With hydrogenchloride In tetrahydrofuran; water | 72% |
| Stage #1: 2,2,3,3-tetrafluoropropanol With sodium hydride In tetrahydrofuran Stage #2: 2,5-dibromoterephtalic acid; N-Methylanthranilic acid; copper(ll) bromide In tetrahydrofuran; 2,2,3,3-tetrafluoropropanol at 60℃; for 48h; Stage #3: With hydrogenchloride In tetrahydrofuran; water | |
| Stage #1: 2,2,3,3-tetrafluoropropanol With sodium hydride In tetrahydrofuran Stage #2: 2,5-dibromoterephtalic acid; 2,3-bis[N,N-(2-trifluoromethylphenyl)imino]butane; copper(ll) bromide In tetrahydrofuran; 2,2,3,3-tetrafluoropropanol at 60℃; for 48h; Stage #3: With hydrogenchloride In tetrahydrofuran; water | |
| Stage #1: 2,2,3,3-tetrafluoropropanol With sodium hydride In tetrahydrofuran Stage #2: 2,5-dibromoterephtalic acid; acetylacetone; copper(ll) bromide In tetrahydrofuran; 2,2,3,3-tetrafluoropropanol at 60℃; for 48h; Stage #3: With hydrogenchloride In tetrahydrofuran; water |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 120℃; for 72h; Sealed tube; High pressure; | 72% |


| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 120℃; for 72h; Sealed tube; High pressure; | 72% |


-
-
197370-00-6
(2,5-dibromo-1,4-phenylene)bis((4-butylphenyl)methanone)

| Conditions | Yield |
|---|---|
| Stage #1: 2,5-dibromoterephtalic acid With thionyl chloride for 10h; Reflux; Stage #2: 1-butylbenzene With aluminium trichloride In dichloromethane at 25℃; for 2h; | 72% |
-
-
75-89-8
2,2,2-trifluoroethanol

-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
1268628-19-8
2,5-bis(2,2,2-trifluoroethoxy)terephthalic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2,2,2-trifluoroethanol With sodium hydride In tetrahydrofuran Stage #2: 2,5-dibromoterephtalic acid; cis-N,N'-dimethyl-1,2-diaminocyclohexane; copper(ll) bromide In tetrahydrofuran; 2,2,2-trifluoroethanol at 60℃; for 96h; Stage #3: With hydrogenchloride In tetrahydrofuran; water | 71% |
| Stage #1: 2,2,2-trifluoroethanol With sodium hydride In tetrahydrofuran Stage #2: 2,5-dibromoterephtalic acid; cis-N,N'-dimethyl-1,2-diaminocyclohexane; copper(ll) bromide In tetrahydrofuran; 2,2,2-trifluoroethanol at 60℃; for 96h; Stage #3: With hydrogenchloride In tetrahydrofuran; water | 71% |
| Stage #1: 2,2,2-trifluoroethanol With sodium hydride In tetrahydrofuran Stage #2: 2,5-dibromoterephtalic acid; D,L-valine; copper(ll) bromide In tetrahydrofuran; 2,2,2-trifluoroethanol at 60℃; for 96h; Stage #3: With hydrogenchloride In tetrahydrofuran; water | |
| Stage #1: 2,2,2-trifluoroethanol With sodium hydride In tetrahydrofuran Stage #2: 2,5-dibromoterephtalic acid; 1,2-bis[(2,4,6-trimethylphenyl)imino]-1,2-dimethylethane; copper(ll) bromide In tetrahydrofuran; 2,2,2-trifluoroethanol at 60℃; for 96h; Stage #3: With hydrogenchloride In tetrahydrofuran; water | |
| Stage #1: 2,2,2-trifluoroethanol With sodium hydride In tetrahydrofuran Stage #2: 2,5-dibromoterephtalic acid; copper(ll) bromide In tetrahydrofuran; 2,2,2-trifluoroethanol at 60℃; for 96h; Stage #3: With hydrogenchloride In tetrahydrofuran; water |
-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
108-67-8
1,3,5-trimethyl-benzene

| Conditions | Yield |
|---|---|
| Stage #1: 2,5-dibromoterephtalic acid With thionyl chloride In dichloromethane for 24h; Reflux; Stage #2: 1,3,5-trimethyl-benzene With aluminum (III) chloride In dichloromethane at 0 - 20℃; for 12h; | 70% |
| Stage #1: 2,5-dibromoterephtalic acid With thionyl chloride In dichloromethane for 12h; Reflux; Stage #2: 1,3,5-trimethyl-benzene With aluminum (III) chloride In dichloromethane Cooling with ice; Reflux; | 65% |
-
-
2425-77-6
2-hexyldecan-1-ol

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2,5-dibromoterephtalic acid With thionyl chloride Reflux; Stage #2: 2-hexyldecan-1-ol With pyridine at 80℃; for 18h; | 70% |
-
-
1134-35-6
4,4'-dimethyl-2,2'-bipyridines

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| In water; N,N-dimethyl-formamide at 120℃; for 72h; Sealed tube; | 70% |

-
-
13731-82-3
2,5-dibromoterephtalic acid

-
-
114460-21-8
calcium acetate hydrate

-
-
25372-07-0
1,4-bis-(1H-imidazol-1-yl)benzene

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 120℃; for 72h; Sealed tube; High pressure; | 69% |

-
-
553-26-4
4,4'-bipyridine

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| In ethanol; water at 160℃; for 24h; Sealed tube; High pressure; | 65% |

-
-
366-18-7
[2,2]bipyridinyl

-
-
13731-82-3
2,5-dibromoterephtalic acid

| Conditions | Yield |
|---|---|
| In water; N,N-dimethyl-formamide at 120℃; for 72h; Sealed tube; | 65% |


| Conditions | Yield |
|---|---|
| In ethanol; water for 0.5h; | 63% |
2,5-Dibromoterephthalic acid Chemical Properties
IUPAC Name: 2,5-dibromoterephthalic acid
Empirical Formula: C8H4Br2O4
Molecular Weight: 323.923g/mol
EINECS: 237-300-8
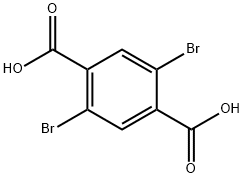
Structure of 1,4-Benzenedicarboxylicacid, 2,5-dibromo- (CAS NO.13731-82-3):
Index of Refraction: 1.679
Molar Refractivity: 55.49 cm3
Molar Volume: 146.8 cm3
Polarizability: 21.99×10-24cm3
Surface Tension: 75.2 dyne/cm
Density: 2.205 g/cm3
Flash Point: 222.9 °C
Enthalpy of Vaporization: 74.08 kJ/mol
Melting Point: 320-325°C
Boiling Point: 445 °C at 760 mmHg
Vapour Pressure: 1.06E-08 mmHg at 25°C
Physical Appearance: white crystalline powder
Product Categories: Phthalic Acids, Esters and Derivatives;Carboxylic Acids;Phenyls & Phenyl-Het;Organic acids;Carboxylic Acids;Phenyls & Phenyl-Het;Derivatives of phthalic acid
Canonical SMILES: C1=C(C(=CC(=C1Br)C(=O)O)Br)C(=O)O
InChI: InChI=1S/C8H4Br2O4/c9-5-1-3(7(11)12)6(10)2-4(5)8(13)14/h1-2H,(H,11,12)(H,13,14)
InChIKey: VUTICWRXMKBOSF-UHFFFAOYSA-N
2,5-Dibromoterephthalic acid Safety Profile
Hazard Codes:  C,
C, Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-37/39-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S37/39:Wear suitable gloves and eye/face protection.
S36:Wear suitable protective clothing.
2,5-Dibromoterephthalic acid Specification
1,4-Benzenedicarboxylicacid, 2,5-dibromo- , its cas register number is 13731-82-3. It also can be called 2,5-Dibromoterephthalic acid ; NSC 115430 . 1,4-Benzenedicarboxylicacid, 2,5-dibromo- (CAS NO.13731-82-3) is irritating to eyes, respiratory system and skin.
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 13732-00-8
- 137320-56-0
- 13732-32-6
- 137-32-6
- 13732-62-2
- 137330-13-3
- 137335-79-6
- 13733-91-0
- 137339-65-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View