-
Name
2-BROMOPROPENE
- EINECS 209-185-4
- CAS No. 557-93-7
- Article Data25
- CAS DataBase
- Density 1.405 g/cm3
- Solubility Immiscible with water.
- Melting Point -87 °C
- Formula C3H5Br
- Boiling Point 48.399 °C at 760 mmHg
- Molecular Weight 120.977
- Flash Point -7.691 °C
- Transport Information UN 1993 3/PG 2
- Appearance light yellow-green liquid
- Safety 23-26-36
- Risk Codes 11-36/37/38
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xi
Xi
- Synonyms Propene,2-bromo- (8CI);2-Bromo-1-propene;2-Bromopropylene;Isopropenyl bromide;a-Methylvinyl bromide;
- PSA 0.00000
- LogP 1.91490
Synthetic route
-
-
33673-74-4, 100304-89-0, 100304-94-7
2,3-dibromo-2-methylpropanoic acid

-
-
557-93-7
2-bromoprop-1-ene

| Conditions | Yield |
|---|---|
| With picoline at 80℃; | 77% |

| Conditions | Yield |
|---|---|
| Stage #1: acetone With magnesium sulfate; hydrazine hydrate In methanol at 20℃; Stage #2: acetone hydrazone With pyridine; N-Bromosuccinimide In dichloromethane at -10 - 0℃; | 74% |
-
-
110-86-1
pyridine

-
-
33673-74-4, 100304-89-0, 100304-94-7
2,3-dibromo-2-methylpropanoic acid

-
-
557-93-7
2-bromoprop-1-ene
-
-
64-17-5
ethanol

-
-
78-75-1
1,2-Dibromopropane

-
-
139-02-6
sodium phenoxide

-
A
-
590-13-6
(Z)-1-propenyl bromide

-
B
-
590-15-8
trans-2-propenyl bromide

-
C
-
557-93-7
2-bromoprop-1-ene

-
-
78-75-1
1,2-Dibromopropane

-
-
139-02-6
sodium phenoxide

-
A
-
590-14-7
1-bromo-1-propene

-
B
-
557-93-7
2-bromoprop-1-ene


| Conditions | Yield |
|---|---|
| With potassium carbonate | |
| With sodium phenoxide | |
| With sodium phenoxide | |
| With sodium ethanolate |

| Conditions | Yield |
|---|---|
| With sodium ethanolate at 100℃; | |
| With potassium carbonate |
-
-
110-52-1
1,4-dibromo-butane

-
-
463-49-0
1,2-propanediene

-
A
-
557-93-7
2-bromoprop-1-ene

-
B
-
106-95-6
allyl bromide

| Conditions | Yield |
|---|---|
| With water; hydrogen; oxygen at 37℃; under 150 Torr; Product distribution; Irradiation; variation of system composition and initial pressure; |

-
-
110-52-1
1,4-dibromo-butane

-
-
50457-57-3
2-propenyl cation

-
A
-
557-93-7
2-bromoprop-1-ene

-
B
-
22211-90-1
bromolanium

| Conditions | Yield |
|---|---|
| Mechanism; Irradiation; |

-
-
110-52-1
1,4-dibromo-butane

-
-
74-99-7
prop-1-yne

-
A
-
557-93-7
2-bromoprop-1-ene

-
B
-
106-95-6
allyl bromide

| Conditions | Yield |
|---|---|
| With water; hydrogen; oxygen at 37℃; under 150 Torr; Product distribution; Irradiation; variation of system composition and initial pressure; |

-
-
10035-10-6, 12258-64-9
hydrogen bromide

-
-
74-99-7
prop-1-yne

-
A
-
594-16-1
2,2-dibromopropane

-
B
-
557-93-7
2-bromoprop-1-ene


-
-
557-93-7
2-bromoprop-1-ene

| Conditions | Yield |
|---|---|
| With pyridine |
-
-
3017-95-6, 127054-44-8, 130232-86-9
2-bromo-1-chloropropane

-
A
-
7647-01-0
hydrogenchloride

-
B
-
557-93-7
2-bromoprop-1-ene

-
-
64-17-5
ethanol

-
-
78-75-1
1,2-Dibromopropane

-
-
139-02-6
sodium phenoxide

-
A
-
590-14-7
1-bromo-1-propene

-
B
-
557-93-7
2-bromoprop-1-ene

-
C
-
4696-23-5, 4696-24-6, 27318-96-3
(prop-1-enyloxy)benzene

| Conditions | Yield |
|---|---|
| Produkt 5: 2-Brom-1-phenoxy-propan; Produkt 6: 1.2-Diphenoxy-propan; Produkt 7: 1-Brom-2-phenoxy-propan; |



-
-
7462-74-0
2-bromo-2-methylpropanamide

-
-
7726-95-6
bromine

-
A
-
594-16-1
2,2-dibromopropane

-
B
-
557-93-7
2-bromoprop-1-ene

-
C
-
67-64-1
acetone


| Conditions | Yield |
|---|---|
| With hydrogen bromide |

| Conditions | Yield |
|---|---|
| at 209.9℃; Equilibrium constant; other temperature; |

| Conditions | Yield |
|---|---|
| at 209.9℃; Equilibrium constant; |
-
-
78-75-1
1,2-Dibromopropane

-
-
996-82-7
sodium diethylmalonate

-
A
-
590-14-7
1-bromo-1-propene

-
B
-
557-93-7
2-bromoprop-1-ene



| Conditions | Yield |
|---|---|
| With magnesium at 600℃; under 0.01 - 0.1 Torr; Title compound not separated from byproducts.; | A 77 % Spectr. B 12 % Spectr. |
-
-
78-75-1
1,2-Dibromopropane

-
A
-
57-55-6
propylene glycol

-
B
-
187737-37-7
propene

-
C
-
590-14-7
1-bromo-1-propene

-
D
-
557-93-7
2-bromoprop-1-ene

-
E
-
123-38-6
propionaldehyde

-
F
-
67-64-1
acetone

-
G
-
75-56-9, 16033-71-9
methyloxirane

| Conditions | Yield |
|---|---|
| With metal oxide; water at 150℃; under 5931.67 Torr; for 1h; |


| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With magnesium In tetrahydrofuran at 40℃; for 2h; Inert atmosphere; Stage #2: butanedial In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | 100% |
| (i) Mg, (ii) /BRN= 1735656/; Multistep reaction; |
-
-
557-93-7
2-bromoprop-1-ene

-
-
1257308-98-7
6-[[tris(1-methylethyl)silyl]oxy]-1,8-diphenyloctan-3-one

-
-
1257309-31-1
2-methyl-6-[[tris(1-methylethyl)silyl]oxy]-8-phenyl-3-(2-phenylethyl)-oct-1-en-3-ol

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With tert.-butyl lithium In diethyl ether; pentane for 0.75h; Stage #2: 6-[[tris(1-methylethyl)silyl]oxy]-1,8-diphenyloctan-3-one In diethyl ether; pentane for 0.25h; Stage #3: With water; ammonium chloride In diethyl ether; pentane | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With tert.-butyl lithium In diethyl ether at -78 - 20℃; for 0.183333h; Inert atmosphere; Stage #2: N-tosyl-4-piperidone In diethyl ether at -78℃; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With tert.-butyl lithium In tetrahydrofuran at -78 - 20℃; for 0.183333h; Inert atmosphere; Stage #2: 24-methyl-5β-cholan-24-one In tetrahydrofuran at -78℃; Inert atmosphere; | 100% |
-
-
557-93-7
2-bromoprop-1-ene

-
-
316791-67-0
(4Z,8E)-4-fluoro-8-methyl-13-phenyltrideca-4,8-dien-12-ynal

-
-
316791-68-1
(6Z,10E)-6-fluoro-2,10-dimethyl-15-phenylpentadeca-1,6,10-trien-14-yn-3-ol

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With magnesium In tetrahydrofuran Stage #2: (4Z,8E)-4-fluoro-8-methyl-13-phenyltrideca-4,8-dien-12-ynal In tetrahydrofuran at 0℃; for 0.5h; Further stages.; | 99% |
-
-
557-93-7
2-bromoprop-1-ene

-
-
14618-80-5
(R)-benzyl glycidol

-
-
604775-07-7
(2R)-1-(benzyloxy)-4-methyl-4-penten-2-ol

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With magnesium In tetrahydrofuran for 2h; Stage #2: (R)-benzyl glycidol With copper(l) iodide In tetrahydrofuran at -35℃; for 1h; | 99% |
| Stage #1: 2-bromoprop-1-ene With iodine; magnesium In tetrahydrofuran Stage #2: (R)-benzyl glycidol With copper(l) iodide In tetrahydrofuran at -35℃; for 1h; Further stages.; | 99% |
-
-
557-93-7
2-bromoprop-1-ene

-
-
25662-28-6
1-(carboxymethoxy)cyclopentadiene

-
-
908125-67-7
2-isopropenyl-cyclopentanecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With magnesium; iodine In tetrahydrofuran at 20℃; Stage #2: 1-(carboxymethoxy)cyclopentadiene; copper(l) iodide In tetrahydrofuran at -15℃; for 1h; | 99% |
-
-
557-93-7
2-bromoprop-1-ene

-
-
1449427-53-5
1-ethynyl-2-({[(2E)-3-phenylprop-2-en-1-yl]oxy}methyl)benzene

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene; 1-ethynyl-2-({[(2E)-3-phenylprop-2-en-1-yl]oxy}methyl)benzene With bis-triphenylphosphine-palladium(II) chloride In triethylamine at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: With copper(l) iodide In triethylamine at 60℃; for 15h; Inert atmosphere; | 99% |
-
-
557-93-7
2-bromoprop-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With tert.-butyl lithium In diethyl ether; pentane at -78 - 20℃; for 1.5h; Inert atmosphere; Stage #2: With copper(l) iodide In diethyl ether; pentane at -78 - -45℃; for 1h; Inert atmosphere; Stage #3: dimethyl 2-(2-iodoethyl)-6-(prop-1-en-2-yl)dihydro-2H-pyran-3,3(4H)-dicarboxylate In diethyl ether; pentane at -78 - 0℃; for 0.5h; Inert atmosphere; | 99% |
-
-
557-93-7
2-bromoprop-1-ene

-
-
1571-08-0
methyl 4-formylbenzoate

-
-
71616-83-6
4-butyrylbenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With acetylacetonatodicarbonylrhodium(l); potassium formate; potassium carbonate; triphenylphosphine In 1,2-dimethoxyethane at 130℃; for 16h; Inert atmosphere; Sealed tube; | 99% |
-
-
557-93-7
2-bromoprop-1-ene

| Conditions | Yield |
|---|---|
| With lithium In tetrahydrofuran at 45 - 50℃; for 2.5h; ultrasonication; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With tert.-butyl lithium In diethyl ether; pentane at -78℃; for 0.25h; Stage #2: 2-allylcyclohexan-1-one In diethyl ether; pentane at -78℃; for 4h; | 98% |
-
-
5457-28-3
3-cyano-1H-indole

-
-
557-93-7
2-bromoprop-1-ene

-
-
1301751-04-1
1-(prop-1-en-2-yl)-1H-indole-3-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium phosphate; copper(l) iodide; ethylenediamine In 1,4-dioxane at 110℃; | 98% |
| With potassium phosphate; copper(l) iodide; ethylenediamine In 1,4-dioxane at 110℃; for 24h; | 98% |
-
-
557-93-7
2-bromoprop-1-ene

-
-
1394171-13-1
(R)-2-(2-(2-methoxy-4-methylphenyl)-5-methylhex-5-en-2-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

-
-
1394171-15-3
(R)-2-methoxy-4-methyl-1-(2,3,6-trimethylhepta-1,6-dien-3-yl)benzene

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With tert.-butyl lithium In tetrahydrofuran at -78℃; for 0.5h; UneV-irradiation; Stage #2: (R)-2-(2-(2-methoxy-4-methylphenyl)-5-methylhex-5-en-2-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane In tetrahydrofuran at -78 - -40℃; for 2h; Inert atmosphere; Stage #3: With iodine In tetrahydrofuran; methanol at -78 - 0℃; for 1h; | 98% |
-
-
557-93-7
2-bromoprop-1-ene

-
-
199525-29-6
tert-Butyl-{2-[(1R,2R,5S,8aS)-5-(3-iodo-propyl)-1,2,5-trimethyl-1,2,3,5,6,7,8,8a-octahydro-naphthalen-1-yl]-ethoxy}-diphenyl-silane

-
-
199525-15-0
(1S,4aS,5R,6R)-(-)-1,2,3,4,4a,5,6,7-octahydro-5-[2-(t-butyldiphenylsiloxy)ethyl]-1-(4-methyl-4-pentenyl)-1,5,6-trimethylnaphthalene

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With tert.-butyl lithium In diethyl ether; pentane at -78 - 23℃; for 1.5h; Metallation; Stage #2: With copper(l) iodide In diethyl ether; pentane at -78 - -40℃; for 1h; transmetallation; Stage #3: tert-Butyl-{2-[(1R,2R,5S,8aS)-5-(3-iodo-propyl)-1,2,5-trimethyl-1,2,3,5,6,7,8,8a-octahydro-naphthalen-1-yl]-ethoxy}-diphenyl-silane In diethyl ether; pentane at -78 - 0℃; for 0.5h; Substitution; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With magnesium In tetrahydrofuran for 8h; Heating; Stage #2: 10-iodo-1-decene With copper(l) iodide In tetrahydrofuran at -78 - 20℃; | 97% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; N,N`-dimethylethylenediamine; copper(l) iodide In toluene at 110℃; for 24h; | 97% |
| With palladium diacetate; 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate In toluene for 24h; Heating; |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With potassium phosphate; N,N-diisopropyl 2-dicyclohexylphosphino-4-(2',6'-dimethylphenyl)benzamide; palladium diacetate In tetrahydrofuran; water at 20℃; for 0.0833333h; Suzuki-Miyaura Coupling; Inert atmosphere; Stage #2: C27H39BO2Si In tetrahydrofuran; water at 20℃; for 1.5h; Reagent/catalyst; Solvent; Time; Suzuki-Miyaura Coupling; Inert atmosphere; | 97% |
-
-
557-93-7
2-bromoprop-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With tert.-butyl lithium In diethyl ether; pentane at -78 - 0℃; for 1h; Inert atmosphere; Stage #2: 2,2-dimethyl-6-trimethylsilanyl-hex-5-ynal In diethyl ether at -78℃; for 0.5h; Inert atmosphere; | 97% |
-
-
557-93-7
2-bromoprop-1-ene

-
-
100-50-5
3-Cyclohexene-1-carboxaldehyde

-
-
139021-69-5
4-(1'-hydroxy-2'-methylprop-2-enyl)cyclohexene

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With lithium In diethyl ether at -30 - 20℃; for 1.75h; Inert atmosphere; Stage #2: 3-Cyclohexene-1-carboxaldehyde In diethyl ether at -30 - 20℃; for 1.75h; Inert atmosphere; Stage #3: With water; ammonium chloride In diethyl ether Cooling with ice; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With tert.-butyl lithium In tetrahydrofuran; pentane at -78 - 0℃; for 1.28333h; Inert atmosphere; Stage #2: Decyl-oxiran With 2,2,6,6-tetramethylpiperidinyl-lithium In tetrahydrofuran; pentane at -78 - 20℃; for 2h; Inert atmosphere; | 96% |
-
-
51589-70-9
benzo[d][1,3]dioxole-5-carbaldehyde-formyl-d1

-
-
557-93-7
2-bromoprop-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With iodine; magnesium In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; Stage #2: benzo[d][1,3]dioxole-5-carbaldehyde-formyl-d1 In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 96% |
-
-
120-57-0
piperonal

-
-
557-93-7
2-bromoprop-1-ene

-
-
1398038-85-1
(±)-1-(benzo[d][1,3]dioxol-5-yl)-2-methylprop-2-en-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With iodine; magnesium In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; Stage #2: piperonal In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); sodium hexamethyldisilazane; (2S,3S)-2,3-bis(diphenylphosphino)butane In tetrahydrofuran at 20℃; for 12h; enantioselective reaction; | 96% |

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); sodium hexamethyldisilazane; (2S,3S)-2,3-bis(diphenylphosphino)butane In tetrahydrofuran at 20℃; for 12h; enantioselective reaction; | 96% |
-
-
557-93-7
2-bromoprop-1-ene

-
-
220018-45-1
(E)-(2S,7R)-7-(tert-Butyl-dimethyl-silanyloxy)-2,6-dimethyl-4-methylene-non-5-enal

-
-
220018-46-2
(E)-(4S,9R)-9-(tert-Butyl-dimethyl-silanyloxy)-2,4,8-trimethyl-6-methylene-undeca-1,7-dien-3-ol

| Conditions | Yield |
|---|---|
| With chromium dichloride; nickel dibromide In N,N-dimethyl-formamide for 48h; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-methyl-1-tetralone With lithium hexamethyldisilazane; [Pd(P-tBu3)Br]2 In toluene at 20℃; for 0.0833333h; Stage #2: 2-bromoprop-1-ene In toluene at 80℃; for 24h; Further stages.; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With tert.-butyl lithium In tetrahydrofuran; pentane at -78℃; for 0.333333h; Stage #2: C23H34O5 In tetrahydrofuran; pentane at -78℃; for 1h; | 95% |
-
-
557-93-7
2-bromoprop-1-ene

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); (R)-N-((S)-(2-(di((3R,5R,7R)-adamantan-1-yl)phosphaneyl)-4,5-dimethoxyphenyl)(phenyl)methyl)-N,2-dimethylpropane-2-sulfinamide; sodium t-butanolate In toluene at 65℃; for 12h; Inert atmosphere; Schlenk technique; enantioselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); sodium hexamethyldisilazane; (2S,3S)-2,3-bis(diphenylphosphino)butane In tetrahydrofuran at 20℃; for 12h; enantioselective reaction; | 95% |
2-Bromopropene Specification
The 2-Bromopropene, with the CAS registry number 557-93-7, is also known as 2-Bromo-1-propene. It belongs to the classification code of Mutation Data. Its EINECS registry number is 209-185-4. This chemical's molecular formula is C3H5Br and molecular weight is 120.98. What's more, its IUPAC name is called 2-Bromoprop-1-ene. It should be stored in a cool, dry and well-ventilated place.
Physical properties about 2-Bromopropene are: (1)ACD/LogP: 1.989; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.99; (4)ACD/LogD (pH 7.4): 1.99; (5)ACD/BCF (pH 5.5): 19.11; (6)ACD/BCF (pH 7.4): 19.11; (7)ACD/KOC (pH 5.5): 287.60; (8)ACD/KOC (pH 7.4): 287.60; (9)#H bond acceptors: 0; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 0 Å2; (13)Index of Refraction: 1.452; (14)Molar Refractivity: 23.247 cm3; (15)Molar Volume: 86.132 cm3; (16)Polarizability: 9.216×10-24cm3; (17)Surface Tension: 23.103 dyne/cm; (18)Density: 1.405 g/cm3; (19)Flash Point: -7.691 °C; (20)Enthalpy of Vaporization: 27.992 kJ/mol; (21)Boiling Point: 48.399 °C at 760 mmHg; (22)Vapour Pressure: 324.193 mmHg at 25 °C.
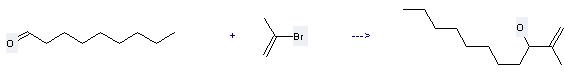
Uses of 2-Bromopropene: (1) it is used as refrigerant and can be used in organic synthesis; (2) it is used to produce other chemicals. For example, it can react with nonanal to get 2-methyl-undec-1-en-3-ol. This reaction needs reagent chromium(II) chloride and solvent dimethylformamide at temperature of 25 °C. The reaction time is 3 hours. The yield is 62 %.
When you are dealing with this chemical, you should be very careful. This chemical may cause inflammation to the skin or other mucous membranes. And it is irritating to eyes, respiratory system and skin. It may catch fire in contact with air, only need brief contact with an ignition source and have a very low flash point or evolve highly flammable gases in contact with water. Therefore, you should wear suitable protective clothing and can not breathe the gas/fumes/vapour/spray. In case of contacting with eyes, you should rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1) SMILES: CC(=C)Br
(2) InChI: InChI=1S/C3H5Br/c1-3(2)4/h1H2,2H3
(3) InChIKey: PHMRPWPDDRGGGF-UHFFFAOYSA-N
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View