-
Name
Aprepitant
- EINECS 677-636-6
- CAS No. 170729-80-3
- Article Data21
- CAS DataBase
- Density 1.51 g/cm3
- Solubility
- Melting Point 75-76°C(lit.)
- Formula C23H21F7N4O3
- Boiling Point
- Molecular Weight 534.434
- Flash Point
- Transport Information
- Appearance off-white to light yellow cyrstalline solid
- Safety
- Risk Codes R20/21/22; R36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms 3H-1,2,4-Triazol-3-one,5-[[2-[1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)-4-morpholinyl]methyl]-1,2-dihydro-,[2R-[2a(R*),3a]]-;Emend;L 754030;MK 0869;MK 869;ONO7436;
- PSA 83.24000
- LogP 4.89000
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium carbonate In water; N,N-dimethyl-formamide at 0℃; for 3h; | 99% |
-
-
219821-37-1
2-[2-[(2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)-4-morpholinyl]-1-iminoethyl]hydrazinecarboxylic acid methyl ester

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| In 5,5-dimethyl-1,3-cyclohexadiene for 3h; Reflux; | 98.4% |
| In 5,5-dimethyl-1,3-cyclohexadiene at 130 - 139℃; for 4h; Temperature; | 97.7% |
| In acetonitrile at 120 - 125℃; | 60.4% |


-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| With diethyl sulfate; triethylamine; lithium diisopropyl amide In dimethylsulfoxide-d6; 5,5-dimethyl-1,3-cyclohexadiene at 33℃; for 14h; Temperature; Inert atmosphere; | 97.9% |

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| With triethylsilane; boron trifluoride diethyl etherate In acetonitrile at 0 - 35℃; for 5h; | 94% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 0 - 10℃; for 4h; | 92% |
-
-
252742-72-6
3-(chloromethyl)-1H-1,2,4-triazol-5(4H)-one

-
-
171338-27-5
[2R-[2α(R*),3α]]-2-[1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)morpholine

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 0.5h; | 68% |

-
-
171338-27-5
[2R-[2α(R*),3α]]-2-[1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)morpholine

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Stage #1: N'-(1-amino-2-chloroethylidene)hydrazinecarboxylic acid methyl ester; [2R-[2α(R*),3α]]-2-[1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)morpholine With N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 3h; Stage #2: In acetonitrile at 110℃; under 1125.11 Torr; for 55h; Product distribution / selectivity; | 53% |
-
-
219821-37-1
2-(R)-(1-(R)-(3,5-bis(trifluoromethyl)phenyl)ethoxy)-3-(S)-4-fluorophenyl-4-2-(N-methylcarboxyactamidrazono)morpholine

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| With pyrographite In methanol at 60℃; for 1h; | 0.96 kg |
| In toluene at 140℃; for 3h; | |
| at 135 - 137℃; for 2h; |

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide at 20 - 23℃; for 1.5h; Product distribution / selectivity; |
-
-
30071-93-3
3,5-bis(trifluoromethyl)phenyl methyl ketone

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 97 percent / borane-methyl sulfide complex; (S)-Me-CBS / various solvent(s); toluene / 1 h / -5 °C 2: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 3: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme | |
| Multi-step reaction with 5 steps 1: 97 percent / borane-methyl sulfide complex; (S)-Me-CBS / various solvent(s); toluene / 1 h / -5 °C 2: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 3: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr 4: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 5: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme | |
| Multi-step reaction with 4 steps 1: 97 percent / borane-methyl sulfide complex; (S)-Me-CBS / various solvent(s); toluene / 1 h / -5 °C 2: boron trifluoride etherate / toluene; heptane / 1 h / 20 °C 3: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme |
-
-
368-63-8, 68120-60-5, 127852-28-2
(R)-[3,5-bis(trifluoromethyl)phenyl]ethanol

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 2: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme | |
| Multi-step reaction with 4 steps 1: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 2: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr 3: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 4: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme | |
| Multi-step reaction with 3 steps 1: boron trifluoride etherate / toluene; heptane / 1 h / 20 °C 2: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme |
-
-
110843-90-8
4-benzyl-morpholine-2,3-dione

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 80 percent / lithium tri(sec-butyl)borohydride; sodium hydroxide; hydrogen peroxide / tetrahydrofuran; H2O / 1 h / 10 °C 2: acetonitrile / 5 - 34 °C 3: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 4: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme | |
| Multi-step reaction with 6 steps 1: 80 percent / lithium tri(sec-butyl)borohydride; sodium hydroxide; hydrogen peroxide / tetrahydrofuran; H2O / 1 h / 10 °C 2: acetonitrile / 5 - 34 °C 3: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 4: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr 5: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 6: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme | |
| Multi-step reaction with 5 steps 1: 80 percent / lithium tri(sec-butyl)borohydride; sodium hydroxide; hydrogen peroxide / tetrahydrofuran; H2O / 1 h / 10 °C 2: 91 percent / DBU / toluene; heptane / 18 h / 20 °C 3: boron trifluoride etherate / toluene; heptane / 1 h / 20 °C 4: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme |
-
-
104-63-2
N-Benzylethanolamine

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 76 percent / tetrahydrofuran; H2O / 21 h / Heating 2: acetonitrile / 5 - 34 °C 3: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 4: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme | |
| Multi-step reaction with 6 steps 1: 76 percent / tetrahydrofuran; H2O / 21 h / Heating 2: acetonitrile / 5 - 34 °C 3: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 4: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr 5: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 6: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme | |
| Multi-step reaction with 5 steps 1: 76 percent / tetrahydrofuran; H2O / 21 h / Heating 2: 91 percent / DBU / toluene; heptane / 18 h / 20 °C 3: boron trifluoride etherate / toluene; heptane / 1 h / 20 °C 4: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme |

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 2: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / N,N-dimethyl-formamide / 10 - 20 °C 2: potassium hydroxide / ethanol; water / 0.2 h / 90 - 100 °C View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / N,N-dimethyl-formamide / 10 - 20 °C 2: potassium hydroxide / ethanol; water / 0.2 h / 90 - 100 °C View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate; N-benzyl-N,N,N-triethylammonium chloride / 5,5-dimethyl-1,3-cyclohexadiene; dimethyl sulfoxide / 20 - 25 °C / Inert atmosphere 2: 5,5-dimethyl-1,3-cyclohexadiene / 2 h / 135 - 141 °C / Inert atmosphere View Scheme |
-
-
287930-73-8
4-benzyl-2-hydroxy-morpholin-3-one

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: acetonitrile / 5 - 34 °C 2: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 3: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme | |
| Multi-step reaction with 5 steps 1: acetonitrile / 5 - 34 °C 2: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 3: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr 4: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 5: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme | |
| Multi-step reaction with 4 steps 1: 91 percent / DBU / toluene; heptane / 18 h / 20 °C 2: boron trifluoride etherate / toluene; heptane / 1 h / 20 °C 3: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme |
-
-
502537-07-7
trifluoro-acetic acid 4-benzyl-3-oxo-morpholin-2-yl ester

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 2: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme | |
| Multi-step reaction with 4 steps 1: 3.51 kg / BF3*Et2O / acetonitrile / 2 h / 20 °C 2: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr 3: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 4: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme |
-
-
502536-97-2
2,2,2-trichloro-acetimidic acid 4-benzyl-3-oxo-morpholin-2-yl ester

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: boron trifluoride etherate / toluene; heptane / 1 h / 20 °C 2: KO-t-Bu / 2-methyl-propan-2-ol; hexane / 16 h / 22 °C 3: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme | |
| Multi-step reaction with 3 steps 1: boron trifluoride etherate / toluene; heptane / 1 h / 20 °C 2: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme | |
| Multi-step reaction with 4 steps 1: boron trifluoride etherate / toluene; heptane / 1 h / 20 °C 2: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr 3: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 4: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme | |
| Multi-step reaction with 5 steps 1: boron trifluoride etherate / toluene; heptane / 1 h / 20 °C 2: KO-t-Bu / 2-methyl-propan-2-ol; hexane / 16 h / 22 °C 3: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr 4: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 5: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme |
-
-
327623-36-9
(2S,2αR)-4-benzyl-2-[1-[3,5-bis(trifluoromethyl)phenyl]]ethoxy-morpholin-3-one

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: KO-t-Bu / 2-methyl-propan-2-ol; hexane / 16 h / 22 °C 2: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme | |
| Multi-step reaction with 4 steps 1: KO-t-Bu / 2-methyl-propan-2-ol; hexane / 16 h / 22 °C 2: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr 3: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 4: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme |
-
-
287930-75-0
(2R,2αR)-4-benzyl-2-[1-[3,5-bis(trifluoromethyl)phenyl]]ethoxy-morpholin-3-one

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr View Scheme | |
| Multi-step reaction with 3 steps 1: 47.6 g / 4-toluenesulfonic acid; H2; HCl / Pd/C / tetrahydrofuran; methanol / 3 h / 20 - 25 °C / 1034.32 Torr 2: potassium carbonate / toluene; dimethylsulfoxide / 2 h / 15 °C 3: 0.96 kg / Darco / methanol / 1 h / 60 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: 5%-palladium/activated carbon; hydrogen / methanol; water / 4.5 h / 50 - 60 °C / 26252.6 Torr 2.1: sodium hydroxide / acetonitrile / 2.5 h / 30 - 40 °C 3.1: tetrahydrofuran / 1 h / 15 - 25 °C 3.2: 1 h / 20 - 65 °C 4.1: triethylsilane; boron trifluoride diethyl etherate / acetonitrile / 5 h / 0 - 35 °C View Scheme |
-
-
472968-68-6
(2S,3R)-3-(4-fluorophenyl)-4-[(1R)-1-phenylethyl]-2-morpholinol

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: K2CO3 / toluene / 5 h / 20 °C 2: 14.3 g / BF3*Et2O / tetrahydrofuran / 1 h / -20 °C 3: TsOH*H2O; hydrogen / 5percent Pd/C / ethanol; toluene / 3 h / 30 °C / 2068.65 Torr 4: NCS; K2CO3 / dimethylformamide / 0.5 h / 0 °C 5: DBU / dimethylformamide / 3 h / 0 - 20 °C 6: hydrogen / 5percent Pd/C / dimethylformamide; ethanol / 2 h / 20 °C / 2068.65 Torr View Scheme |

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: DIBALH / toluene; tetrahydrofuran / 0.5 h / -20 °C 2: K2CO3 / toluene / 5 h / 20 °C 3: 14.3 g / BF3*Et2O / tetrahydrofuran / 1 h / -20 °C 4: TsOH*H2O; hydrogen / 5percent Pd/C / ethanol; toluene / 3 h / 30 °C / 2068.65 Torr 5: NCS; K2CO3 / dimethylformamide / 0.5 h / 0 °C 6: DBU / dimethylformamide / 3 h / 0 - 20 °C 7: hydrogen / 5percent Pd/C / dimethylformamide; ethanol / 2 h / 20 °C / 2068.65 Torr View Scheme |
-
-
472968-69-7
2,2,2-Trichloro-acetimidic acid (2R,3R)-3-(4-fluoro-phenyl)-4-((R)-1-phenyl-ethyl)-morpholin-2-yl ester

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 14.3 g / BF3*Et2O / tetrahydrofuran / 1 h / -20 °C 2: TsOH*H2O; hydrogen / 5percent Pd/C / ethanol; toluene / 3 h / 30 °C / 2068.65 Torr 3: NCS; K2CO3 / dimethylformamide / 0.5 h / 0 °C 4: DBU / dimethylformamide / 3 h / 0 - 20 °C 5: hydrogen / 5percent Pd/C / dimethylformamide; ethanol / 2 h / 20 °C / 2068.65 Torr View Scheme |
-
-
380499-07-0
(2R)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)-5,6-dihydro-2H-1,4-oxazine

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: hydrogen / 5percent Pd/C / dimethylformamide; ethanol / 2 h / 20 °C / 2068.65 Torr View Scheme | |
| Multi-step reaction with 2 steps 1.1: potassium formate / palladium on carbon / ethanol; water / 2 h / 20 °C 2.1: N-ethyl-N,N-diisopropylamine / acetonitrile / 3 h / 20 °C 2.2: 55 h / 110 °C / 1125.11 Torr View Scheme |
-
-
380499-06-9
(2R,3R)-2-[(1R)-1-(3,5-bis-trifluoro-methylphenyl)ethoxy]-3-(4-fluorophenyl)morpholine

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: NCS; K2CO3 / dimethylformamide / 0.5 h / 0 °C 2: DBU / dimethylformamide / 3 h / 0 - 20 °C 3: hydrogen / 5percent Pd/C / dimethylformamide; ethanol / 2 h / 20 °C / 2068.65 Torr View Scheme | |
| Multi-step reaction with 3 steps 1.1: sodium hypochlorite; potassium carbonate / acetonitrile / 0.25 h 1.2: 14 h / 20 °C 2.1: potassium formate / palladium on carbon / ethanol; water / 2 h / 20 °C 3.1: potassium carbonate / N,N-dimethyl-formamide / 0.5 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: sodium hypochlorite; potassium carbonate / acetonitrile / 0.25 h 1.2: 14 h / 20 °C 2.1: potassium formate / palladium on carbon / ethanol; water / 2 h / 20 °C 3.1: N-ethyl-N,N-diisopropylamine / acetonitrile / 3 h / 20 °C 3.2: 55 h / 110 °C / 1125.11 Torr View Scheme |

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: DBU / dimethylformamide / 3 h / 0 - 20 °C 2: hydrogen / 5percent Pd/C / dimethylformamide; ethanol / 2 h / 20 °C / 2068.65 Torr View Scheme |
-
-
472968-70-0
(2R,3R)-2-[(R)-1-(3,5-Bis-trifluoromethyl-phenyl)-ethoxy]-3-(4-fluoro-phenyl)-4-((R)-1-phenyl-ethyl)-morpholine

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: TsOH*H2O; hydrogen / 5percent Pd/C / ethanol; toluene / 3 h / 30 °C / 2068.65 Torr 2: NCS; K2CO3 / dimethylformamide / 0.5 h / 0 °C 3: DBU / dimethylformamide / 3 h / 0 - 20 °C 4: hydrogen / 5percent Pd/C / dimethylformamide; ethanol / 2 h / 20 °C / 2068.65 Torr View Scheme |
-
-
1349902-81-3
C27H31F7N4O4

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 90 - 100℃; for 1h; Reagent/catalyst; Solvent; Large scale; |

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 90 - 100℃; for 0.2h; |
-
-
1185503-10-9
(2S,3R)-4-benzyl-3-(4-fluorophenyl)morpholin-2-ol

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: boron trifluoride diethyl etherate / toluene / 1 h / -10 °C 2.1: hydrogen; toluene-4-sulfonic acid / palladium 10% on activated carbon / methanol / 16 h / 20 °C 3.1: sodium hypochlorite; potassium carbonate / acetonitrile / 0.25 h 3.2: 14 h / 20 °C 4.1: potassium formate / palladium on carbon / ethanol; water / 2 h / 20 °C 5.1: potassium carbonate / N,N-dimethyl-formamide / 0.5 h / 20 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: boron trifluoride diethyl etherate / toluene / 1 h / -10 °C 2.1: hydrogen; toluene-4-sulfonic acid / palladium 10% on activated carbon / methanol / 16 h / 20 °C 3.1: sodium hypochlorite; potassium carbonate / acetonitrile / 0.25 h 3.2: 14 h / 20 °C 4.1: potassium formate / palladium on carbon / ethanol; water / 2 h / 20 °C 5.1: N-ethyl-N,N-diisopropylamine / acetonitrile / 3 h / 20 °C 5.2: 55 h / 110 °C / 1125.11 Torr View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: aprepitant With dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate; sodium hexamethyldisilazane In tetrahydrofuran at -3℃; for 2h; Stage #2: 1-deoxy-1-(methylamino)-D-glucitol With palladium 10% on activated carbon; hydrogen In methanol; water for 4h; | 98% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
170729-80-3
aprepitant

-
-
265121-01-5
[3-[[(2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)-4-morpholinyl]methyl]-2,5-dihydro-5-oxo-1H-1,2,4-triazol-1-yl]phosphonic acid bis(phenylmethyl)ester

| Conditions | Yield |
|---|---|
| With sodium hexamethyldisilazane In tetrahydrofuran at 0℃; for 1h; Temperature; Inert atmosphere; | 93.6% |
| With sodium hexamethyldisilazane In tetrahydrofuran at 0℃; for 1h; Temperature; Inert atmosphere; | 93.6% |
| With sodium hexamethyldisilazane In tetrahydrofuran at 0℃; for 0.25h; phosphorylation; | 90% |
-
-
17176-77-1
Dibenzyl phosphite

-
-
170729-80-3
aprepitant

-
-
265121-01-5
[3-[[(2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)-4-morpholinyl]methyl]-2,5-dihydro-5-oxo-1H-1,2,4-triazol-1-yl]phosphonic acid bis(phenylmethyl)ester

| Conditions | Yield |
|---|---|
| With triethylamine In tetrachloromethane; dichloromethane at 0 - 10℃; Reagent/catalyst; Solvent; | 92.4% |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tert-butyl methyl ether at 0 - 30℃; | 90.6% |

| Conditions | Yield |
|---|---|
| With dmap In chloroform at 0 - 20℃; | 90.4% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| Stage #1: aprepitant With lithium hydroxide monohydrate In 1-methyl-pyrrolidin-2-one at 20℃; for 0.5h; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In 1-methyl-pyrrolidin-2-one at 25 - 30℃; for 0.5h; Quantum yield; | 87.4% |
-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| With dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate; sodium hexamethyldisilazane In tetrahydrofuran at -3℃; for 2h; | 86.7% |
| Multi-step reaction with 3 steps 1: sodium hexamethyldisilazane / tetrahydrofuran / 3.5 h / -20 °C 2: hydrogen / palladium 10% on activated carbon / methanol / 1 h / 5171.62 Torr 3: hydrogenchloride / water; methanol / 0.08 h / 0 - 5 °C / pH 1 View Scheme | |
| Multi-step reaction with 2 steps 1: sodium hexamethyldisilazane / tetrahydrofuran / 1 h / 0 °C / Inert atmosphere 2: borane pyridine; methanol / 2 h / 20 °C / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: aprepitant With sodium hexamethyldisilazane In tetrahydrofuran at -5 - 5℃; for 0.5h; Inert atmosphere; Cooling with ice; Stage #2: C26H22ClO3P In tetrahydrofuran at 5 - 20℃; for 1h; Solvent; | 82% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrachloromethane; dichloromethane at 0 - 10℃; Reagent/catalyst; Solvent; | 81.1% |
-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| With deuteroacetic acid; magnesium In dimethylsulfoxide-d6; d(4)-methanol; water-d2 at 20℃; for 2h; regioselective reaction; | 43% |
-
-
170729-80-3
aprepitant

| Conditions | Yield |
|---|---|
| With magnesium; acetic acid In water; dimethyl sulfoxide at 20℃; for 2h; regioselective reaction; | 41% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
170729-80-3
aprepitant

-
A
-
265121-01-5
[3-[[(2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)-4-morpholinyl]methyl]-2,5-dihydro-5-oxo-1H-1,2,4-triazol-1-yl]phosphonic acid bis(phenylmethyl)ester

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at 20℃; for 0.75h; phosphorylation; | A 39% B 5 % Chromat. |

-
-
170729-80-3
aprepitant


| Conditions | Yield |
|---|---|
| With magnesium; acetic acid In water; dimethyl sulfoxide at 20℃; for 5h; regioselective reaction; | A 16% B 22% |
Aprepitant Chemical Properties
Product Name: Aprepitant (CAS NO.170729-80-3)
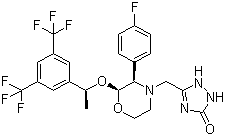
Molecular Formula: C23H21F7N4O3
Molecular Weight: 534.43g/mol
Mol File: 170729-80-3.mol
Appearance: Off-White to Light Yellow Cyrstalline Solid
Density: 1.51 g/cm3
Index of Refraction: 1.564
Molar Refractivity: 115.07 cm3
Molar Volume: 353.5 cm3
Surface Tension: 39 dyne/cm
XLogP3-AA: 4.2
H-Bond Donor: 2
H-Bond Acceptor: 12
Structure Descriptors of Aprepitant (CAS NO.170729-80-3):
IUPAC Name: 5-[[(2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)morpholin-4-yl]methyl]-1,2-dihydro-1,2,4-triazol-3-one
Canonical SMILES: CC(C1=CC(=CC(=C1)C(F)(F)F)C(F)(F)F)OC2C(N(CCO2)CC3=NC(=O)NN3)C4=CC=C(C=
C4)F
Isomeric SMILES: C[C@H](C1=CC(=CC(=C1)C(F)(F)F)C(F)(F)F)O[C@@H]2[C@@H](N(CCO2)CC3=NC(=O)
NN3)C4=CC=C(C=C4)F
InChI: InChI=1S/C23H21F7N4O3/c1-12(14-8-15(22(25,26)27)10-16(9-14)23(28,29)30)37-20-19(13-2-4-17(24)5-3-13)34(6-7-36-20)11-18-31-21(35)33-32-18/h2-5,8-10,12,19-20H,6-7,11H2,1H3,(H2,31,32,33,35)/t12-,19+,20-/m1/s1
InChIKey: ATALOFNDEOCMKK-OITMNORJSA-N
Product Categories: Chiral Reagents; Heterocycles; Intermediates & Fine Chemicals; Pharmaceuticals
Aprepitant Uses
Aprepitant (CAS NO.170729-80-3) is classified as an NK1 antagonist. Aprepitant(170729-80-3) also has been shown to inhibit both the acute and delayed emesis induced by cytotoxic chemotherapeutic.
Aprepitant Toxicity Data With Reference
Aprepitant (CAS NO.170729-80-3) should not be used concurrently with pimozide, terfenadine, astemizole, or cisapride. Dose-dependent inhibition of CYP3A4 by Aprepitant could result in elevated plasma concentrations of these drugs, potentially causing serious or life-threatening reactions.
Aprepitant Specification
Aprepitant , its CAS NO. is 170729-80-3, the synonyms are 3-(((2R,3S)-3-(p-Fluorophenyl)-2-(((alphaR)-alpha-methyl-
3,5-bis(trifluoromethyl)benzyl)oxy)morpholino)methyl)-delta(sup 2)-1,2,4-triazolin-5-one ; 3H-1,2,4-Triazol-3-one, 5-(((2R,3S)-2-((1R)-1-(3,5-bis(trifluoromethyl)phenyl)ethoxy)-3-(4-fluorophenyl)-4-morpholinyl)methyl)-1,2-dihydro- ; Emend .
Related Products
- Aprepitant
- 170730-06-0
- 170735-25-8
- 170737-46-9
- 170737-53-8
- 170737-93-6
- 170744-18-0
- 170749-59-4
- 17075-03-5
- 17075-14-8
- 17075-39-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View