Greenutra Resource Inc
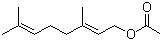
Product Name: Nicotiana Tabacum Extract, Solanesol Latin Name: Nicotiana tabacum L. CAS No.: 13190-97-1 Molecular Formula: C45H74O Molecular Weight: 631.079 Quality data Identific
Simagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:13190-97-1
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryHebei yanxi chemical co.,LTD.
Hebei Yanxi Chemical Co., Ltd. is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low car
Cas:13190-97-1
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:13190-97-1
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryXian Changyue Biological Technology Co., Ltd.
Solanol is a white powder or waxy solid, usually containing a small amount of impurity and is pale yellow. Solanol is soluble in organic solvents such as ether, acetone, and hydrocarbon, and is not soluble in water. It is non-selective absorption o
Chengdu Lemeitian Pharmaceutical Technology Co,. Ltd
ADVANTAGE: 1. More than 3,000 Chinese medicine reference substances / standard products available from stock, issued on the same day, multiple cities can be delivered the next day 2. The products are provided with COA, HPLC, NMR, quality assurance,
Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price in China. If you are looking for the product’s supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product specifica
Cas:13190-97-1
Min.Order:1 Kilogram
FOB Price: $2.0 / 3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hubei DiBo chemical co., LTD
Name:solanesol Alias:9 polyisoprene primary alcohol English Name:solanesol CAS No.13190-97-1 Molecular formula:C45H74O Molecular weight:631.0685 Packaging Specification: 25kg/drum high purity 99%min stock Hubei DiBo Chemical Co., Ltd. was
Cas:13190-97-1
Min.Order:25 Kilogram
FOB Price: $1.0 / 2.0
Type:Other
inquiryLeader Biochemical Group
About Product Details Solanesol Chemical Properties Melting point 33°C Boiling point 685.6±24.0 °C(Predi
Cas:13190-97-1
Min.Order:1 Kilogram
FOB Price: $3.0 / 10.0
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
Solanesol Basic information Market investigation The international market of the high-purity solanesol Output analysis Chemical PropertiesApplication Product Name: Solanesol Synony
Cas:13190-97-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our Advantage Rich Experience Our products are sold all over Europe,North&South America, Sino-East, Asia and pacific area as well as Africa,we establish long term. Quality service Company cooperates with research institutes. We strictly con
Cas:13190-97-1
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:13190-97-1
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Hangzhou Sartort Biopharma Co., Ltd
Appearance:White powder Storage:Room temperature Package:25kg/drum Application:Natural Extracts Transportation:Express/Sea/Air Port:Shanghai
Cas:13190-97-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:powder Storage:Store in sealed containers at cool & dry pla
Cas:13190-97-1
Min.Order:100 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:13190-97-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
Product Name: Solanesol CAS: 13190-97-1 MF: C45H74O MW: 631.07 EINECS: 603-532-7 Mol File: 13190-97-1.mol Solanesol Structure Solanesol Chemical Properties Melting point 33°C Boiling point 685.6±24.0 °C(Pred
Baoji Guokang Healthchem co.,ltd
About us Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad.
Qingdao Beluga Import and Export Co., LTD
Solanesol CAS:13190-97-1 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates,
Cas:13190-97-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Changchun Artel lmport and Export trade company
Minimum Order Qty. 10 Gram Supply Ability 500 Kilograms/Month Storage store in cool, dry, ventilated place 20℃ Delivery Time 3 business days after payment Payment Term TT,western union,Paypal,MoneyGram Package 10g,20g,50g,100g,500g,1KGS,
Cas:13190-97-1
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Triumph International Development Limilted
Appearance:off-white to white solid Storage:Store in a cool,dry place and keep away from direct strong light Package:As customer request Application:Used for Synthesis API and Research Transportation:As customer request Port:Qingda
Cas:13190-97-1
Min.Order:0 Metric Ton
FOB Price: $16.0 / 20.0
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Chengdu Biopurify Phytochemicals Ltd.
Chengdu Biopurify Phytochemicals Ltd. is a leading company in the research, development, manufacture and marketing of High Quality Phytochemicals and Extracts(especially Active Ingredients from Traditional Chinese Medicine,Traditional Chinese Medic
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Binbo Biological Co., Ltd
1.high purity 2.consistent quality 3.competitive price 4.fast shipping Binbo Biological Products Co. Ltd., is a professional high-tech enterprise which engaged in Biological products and raw materials. Binbo is engaged in R&D with perfect eq
Cas:13190-97-1
Min.Order:1 Kilogram
FOB Price: $5.0 / 10.0
Type:Lab/Research institutions
inquirySynthetic route

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| With lithium; ethylamine In diethyl ether at -78℃; | 50% |
-
-
68690-45-9, 80370-68-9, 53254-63-0
(2E,6E)-3,7,11-trimethyl-1-(p-tolylsulfonyl)dodeca-2,6,10-triene

-
-
71135-48-3
(2E,6E,10E)-1-Benzyloxy-3,7,11-trimethyldodeca-2,6,10-trien-12-ol

-
-
70238-40-3
(2E,6E,10E)-2,6,10-trimethyl-12-(toluene-4-sulfonyl)-dodeca-2,6,10-trien-1-ol

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multistep reaction; |

-
-
105-87-3
3,7-dimethyl-2E,6-octadien-1-yl acetate

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 14 steps 1: 44 percent / SeO2 / aq. ethanol / 9 h / Heating 2: 72 percent / NaBH4 / methanol / 5 h / -10 °C 3: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 4: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 5: 5percent sodium amalgam, disodium hydrogen phosphate / methanol / 2 h / 0 °C 6: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 7: 78 percent / dimethylformamide / 24 h / Ambient temperature 8: 1.) n-BuLi, 3.) H2O, KOH 9: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 10: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 11: 1.) n-BuLi, 3.) H2O, KOH 12: 70 percent / Li, ethylamine / diethyl ether 13: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 14: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 13 steps 1: 44 percent / SeO2 / aq. ethanol / 9 h / Heating 2: 72 percent / NaBH4 / methanol / 5 h / -10 °C 3: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 4: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 5: NaOMe / methanol / 2 h / Ambient temperature 7: 1.) n-BuLi, 3.) H2O, KOH 8: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 9: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 10: 1.) n-BuLi, 3.) H2O, KOH 11: 70 percent / Li, ethylamine / diethyl ether 12: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 13: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 15 steps 1: 44 percent / SeO2 / aq. ethanol / 9 h / Heating 2: 72 percent / NaBH4 / methanol / 5 h / -10 °C 3: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 4: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 5: NaOMe / methanol / 2 h / Ambient temperature 6: 264 mg / Li, ethylamine / diethyl ether / 1 h / -78 °C 7: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 8: 78 percent / dimethylformamide / 24 h / Ambient temperature 9: 1.) n-BuLi, 3.) H2O, KOH 10: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 11: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 12: 1.) n-BuLi, 3.) H2O, KOH 13: 70 percent / Li, ethylamine / diethyl ether 14: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 15: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
50848-64-1
2E,6E,10E-geranylgeranyl bromide

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: 78 percent / dimethylformamide / 24 h / Ambient temperature 2: 1.) n-BuLi, 3.) H2O, KOH 3: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 4: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 5: 1.) n-BuLi, 3.) H2O, KOH 6: 70 percent / Li, ethylamine / diethyl ether 7: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 8: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
37905-04-7
(2E,6E)-8-bromo-3,7-dimethylocta-2,6-dienyl acetate

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 2: 5percent sodium amalgam, disodium hydrogen phosphate / methanol / 2 h / 0 °C 3: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 4: 78 percent / dimethylformamide / 24 h / Ambient temperature 5: 1.) n-BuLi, 3.) H2O, KOH 6: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 7: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 8: 1.) n-BuLi, 3.) H2O, KOH 9: 70 percent / Li, ethylamine / diethyl ether 10: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 11: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 10 steps 1: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 2: NaOMe / methanol / 2 h / Ambient temperature 4: 1.) n-BuLi, 3.) H2O, KOH 5: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 6: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 7: 1.) n-BuLi, 3.) H2O, KOH 8: 70 percent / Li, ethylamine / diethyl ether 9: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 10: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 12 steps 1: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 2: NaOMe / methanol / 2 h / Ambient temperature 3: 264 mg / Li, ethylamine / diethyl ether / 1 h / -78 °C 4: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 5: 78 percent / dimethylformamide / 24 h / Ambient temperature 6: 1.) n-BuLi, 3.) H2O, KOH 7: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 8: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 9: 1.) n-BuLi, 3.) H2O, KOH 10: 70 percent / Li, ethylamine / diethyl ether 11: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 12: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 7 steps 1: 1.) n-BuLi, 3.) H2O, KOH 2: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 3: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 4: 1.) n-BuLi, 3.) H2O, KOH 5: 70 percent / Li, ethylamine / diethyl ether 6: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 7: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) n-BuLi, 3.) H2O, KOH 2: 70 percent / Li, ethylamine / diethyl ether 3: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 4: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
37905-03-6
8-hydroxygeranyl acetate

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 12 steps 1: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 2: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 3: 5percent sodium amalgam, disodium hydrogen phosphate / methanol / 2 h / 0 °C 4: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 5: 78 percent / dimethylformamide / 24 h / Ambient temperature 6: 1.) n-BuLi, 3.) H2O, KOH 7: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 8: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 9: 1.) n-BuLi, 3.) H2O, KOH 10: 70 percent / Li, ethylamine / diethyl ether 11: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 12: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 11 steps 1: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 2: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 3: NaOMe / methanol / 2 h / Ambient temperature 5: 1.) n-BuLi, 3.) H2O, KOH 6: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 7: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 8: 1.) n-BuLi, 3.) H2O, KOH 9: 70 percent / Li, ethylamine / diethyl ether 10: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 11: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 13 steps 1: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 2: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 3: NaOMe / methanol / 2 h / Ambient temperature 4: 264 mg / Li, ethylamine / diethyl ether / 1 h / -78 °C 5: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 6: 78 percent / dimethylformamide / 24 h / Ambient temperature 7: 1.) n-BuLi, 3.) H2O, KOH 8: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 9: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 10: 1.) n-BuLi, 3.) H2O, KOH 11: 70 percent / Li, ethylamine / diethyl ether 12: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 13: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 8 steps 1: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 2: 1.) n-BuLi, 3.) H2O, KOH 3: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 4: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 5: 1.) n-BuLi, 3.) H2O, KOH 6: 70 percent / Li, ethylamine / diethyl ether 7: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 8: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 5 steps 1: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 2: 1.) n-BuLi, 3.) H2O, KOH 3: 70 percent / Li, ethylamine / diethyl ether 4: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 5: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
37905-02-5
(2E,6E)-3,7-dimethyl-8-oxoocta-2,6-dien-1-yl acetate

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 13 steps 1: 72 percent / NaBH4 / methanol / 5 h / -10 °C 2: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 3: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 4: 5percent sodium amalgam, disodium hydrogen phosphate / methanol / 2 h / 0 °C 5: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 6: 78 percent / dimethylformamide / 24 h / Ambient temperature 7: 1.) n-BuLi, 3.) H2O, KOH 8: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 9: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 10: 1.) n-BuLi, 3.) H2O, KOH 11: 70 percent / Li, ethylamine / diethyl ether 12: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 13: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 12 steps 1: 72 percent / NaBH4 / methanol / 5 h / -10 °C 2: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 3: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 4: NaOMe / methanol / 2 h / Ambient temperature 6: 1.) n-BuLi, 3.) H2O, KOH 7: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 8: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 9: 1.) n-BuLi, 3.) H2O, KOH 10: 70 percent / Li, ethylamine / diethyl ether 11: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 12: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 14 steps 1: 72 percent / NaBH4 / methanol / 5 h / -10 °C 2: 94 percent / PBr3, pyridine / diethyl ether / 6 h / 0 °C 3: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 4: NaOMe / methanol / 2 h / Ambient temperature 5: 264 mg / Li, ethylamine / diethyl ether / 1 h / -78 °C 6: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 7: 78 percent / dimethylformamide / 24 h / Ambient temperature 8: 1.) n-BuLi, 3.) H2O, KOH 9: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 10: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 11: 1.) n-BuLi, 3.) H2O, KOH 12: 70 percent / Li, ethylamine / diethyl ether 13: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 14: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
24034-73-9
Geranylgeraniol

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 2: 78 percent / dimethylformamide / 24 h / Ambient temperature 3: 1.) n-BuLi, 3.) H2O, KOH 4: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 5: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 6: 1.) n-BuLi, 3.) H2O, KOH 7: 70 percent / Li, ethylamine / diethyl ether 8: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 9: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
53254-60-7
3,7-dimethyl-1-(p-toluenesulfonyl)-2(E),6-octadiene

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 2: 5percent sodium amalgam, disodium hydrogen phosphate / methanol / 2 h / 0 °C 3: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 4: 78 percent / dimethylformamide / 24 h / Ambient temperature 5: 1.) n-BuLi, 3.) H2O, KOH 6: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 7: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 8: 1.) n-BuLi, 3.) H2O, KOH 9: 70 percent / Li, ethylamine / diethyl ether 10: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 11: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 10 steps 1: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 2: NaOMe / methanol / 2 h / Ambient temperature 4: 1.) n-BuLi, 3.) H2O, KOH 5: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 6: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 7: 1.) n-BuLi, 3.) H2O, KOH 8: 70 percent / Li, ethylamine / diethyl ether 9: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 10: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 12 steps 1: 1.) n-BuLi / 1.) THF, HMPA, hexane, -78 deg C, 1.5 h, 2.) 6 h 2: NaOMe / methanol / 2 h / Ambient temperature 3: 264 mg / Li, ethylamine / diethyl ether / 1 h / -78 °C 4: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 5: 78 percent / dimethylformamide / 24 h / Ambient temperature 6: 1.) n-BuLi, 3.) H2O, KOH 7: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 8: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 9: 1.) n-BuLi, 3.) H2O, KOH 10: 70 percent / Li, ethylamine / diethyl ether 11: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 12: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
24529-80-4
(E)-1-O-acetyl-4-chloro-3-methyl-2-buten-1-ol

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 2: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
33569-79-8
(E,E,E,E,E,E,E)-3,7,11,15,19,23,27,31-octamethyldotriaconta-2,6,10,14,18,22,26,30-octaen-1-ol

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 2: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
68778-93-8
(2E,6E,10E,14E,18E)-3,7,11,15,19,23-hexamethyltetracosa-2,6,10,14,18,22-hexaen-1-ol

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 2: 1.) n-BuLi, 3.) H2O, KOH 3: 70 percent / Li, ethylamine / diethyl ether 4: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 5: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
68690-47-1
1-methyl-4-{[(2E,6E,10E)-3,7,11,15-tetramethylhexadeca-2,6,10,14-tetraen-1-yl]sulfonyl}benzene

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 1.) n-BuLi, 3.) H2O, KOH 2: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 3: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 4: 1.) n-BuLi, 3.) H2O, KOH 5: 70 percent / Li, ethylamine / diethyl ether 6: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 7: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
71816-56-3
(2E,6E,10E)-3,7,11,15-tetramethyl-9-p-tolylsulphonylhexadeca-2,6,10,14-tetraen-1-ol

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 2: 1.) n-BuLi, 3.) H2O, KOH 3: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 4: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 5: 1.) n-BuLi, 3.) H2O, KOH 6: 70 percent / Li, ethylamine / diethyl ether 7: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 8: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 10 steps 1: 264 mg / Li, ethylamine / diethyl ether / 1 h / -78 °C 2: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 3: 78 percent / dimethylformamide / 24 h / Ambient temperature 4: 1.) n-BuLi, 3.) H2O, KOH 5: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 6: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 7: 1.) n-BuLi, 3.) H2O, KOH 8: 70 percent / Li, ethylamine / diethyl ether 9: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 10: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
73127-51-2
(2E,6E,10E)-3,7,11,15-tetramethyl-9-[(4-methylphenyl)sulfonyl]hexadeca-2,6,10,14-tetraen-1-yl acetate

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 5percent sodium amalgam, disodium hydrogen phosphate / methanol / 2 h / 0 °C 2: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 3: 78 percent / dimethylformamide / 24 h / Ambient temperature 4: 1.) n-BuLi, 3.) H2O, KOH 5: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 6: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 7: 1.) n-BuLi, 3.) H2O, KOH 8: 70 percent / Li, ethylamine / diethyl ether 9: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 10: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 9 steps 1: NaOMe / methanol / 2 h / Ambient temperature 3: 1.) n-BuLi, 3.) H2O, KOH 4: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 5: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 6: 1.) n-BuLi, 3.) H2O, KOH 7: 70 percent / Li, ethylamine / diethyl ether 8: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 9: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme | |
| Multi-step reaction with 11 steps 1: NaOMe / methanol / 2 h / Ambient temperature 2: 264 mg / Li, ethylamine / diethyl ether / 1 h / -78 °C 3: 91 percent / PBr3, pyridine / diethyl ether / 6 h / -5 °C 4: 78 percent / dimethylformamide / 24 h / Ambient temperature 5: 1.) n-BuLi, 3.) H2O, KOH 6: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 7: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 8: 1.) n-BuLi, 3.) H2O, KOH 9: 70 percent / Li, ethylamine / diethyl ether 10: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 11: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
68690-49-3
(2E,6E,10E,14E,18E)-3,7,11,15,19,23-hexamethyl-1-p-tolylsulphonyltetracosa-2,6,10,14,18,22-hexaene

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 1.) n-BuLi, 3.) H2O, KOH 2: 70 percent / Li, ethylamine / diethyl ether 3: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 4: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
78284-77-2
(2E,6E,10E,14E,18E)-3,7,11,15,19,23-hexamethyl-9-p-tolylsulphonyltetracosa-2,6,10,14,18,22-hexaen-1-ol

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 63 percent / Li, ethylamine / diethyl ether / 1 h / -78 °C 2: 1.) PBr3, pyridine / 1.) ether, 2.) DMF, ambient temperature 3: 1.) n-BuLi, 3.) H2O, KOH 4: 70 percent / Li, ethylamine / diethyl ether 5: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 6: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 70 percent / Li, ethylamine / diethyl ether 2: 1.) n-BuLi / 1.) THF, HMPA, 2.) -78 deg C 3: 50 percent / Li, ethylamine / diethyl ether / -78 °C View Scheme |
-
-
13190-97-1
solanesol

-
-
52610-77-2
solanesyl bromide

| Conditions | Yield |
|---|---|
| With pyridine; phosphorus tribromide In diethyl ether | 99% |
| With phosphorus tribromide; triethylamine In di-isopropyl ether at -10℃; for 2h; Product distribution / selectivity; | 94% |
| With phosphorus tribromide In tetrahydrofuran at -10℃; for 2h; Product distribution / selectivity; | 88% |
-
-
13190-97-1
solanesol

-
-
138647-72-0
solanesyl chloride

| Conditions | Yield |
|---|---|
| With phosphorus trichloride In N,N-dimethyl-formamide at 20℃; for 1h; | 96.1% |
| With N,N-dimethyl-formamide; phosphorus trichloride In tetrahydrofuran at 20℃; for 2h; | 95.1% |
| With N,N-dimethyl-formamide; phosphorus trichloride In tetrahydrofuran at 20℃; for 2h; Product distribution / selectivity; | 95.1% |

| Conditions | Yield |
|---|---|
| With pyridine; trichloroacetonitrile at 80℃; for 1h; Microwave irradiation; | 84% |

| Conditions | Yield |
|---|---|
| With pyridine; trichloroacetonitrile at 80℃; for 1h; Microwave irradiation; | 82% |
-
-
13190-97-1
solanesol

-
-
79-04-9
chloroacetyl chloride

-
-
1190409-13-2
chloro-acetic acid 3,7,11,15,19,23,27,31,35-nonamethyl-hexatriaconta-2,6,10,14,18,22,26,30,34-nonaen-1-yl ester

| Conditions | Yield |
|---|---|
| With triethylamine In benzene at 5℃; for 4h; | 80.3% |
-
-
108-30-5
succinic acid anhydride

-
-
13190-97-1
solanesol

-
-
630393-89-4
succinic acid mono-(3,7,11,15,19,23,27,31,35-nonamethyl-hexatriaconta-2,6,10,14,18,22,26,30,34-nonaen-1-yl) ester

| Conditions | Yield |
|---|---|
| With pyridine; dmap at 20℃; for 6h; | 80% |
| With dmap; triethylamine In dichloromethane at 20℃; for 48h; | 70% |
| With pyridine; dmap at 20℃; for 6h; | |
| With dmap; triethylamine In dichloromethane; N,N-dimethyl-formamide at 20℃; for 48h; Inert atmosphere; |
-
-
13190-97-1
solanesol

-
-
6099-04-3
3-methoxycinnamic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 6h; | 80% |
-
-
13190-97-1
solanesol

-
-
637-44-5
phenylpropyolic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 25℃; for 6h; | 69% |
-
-
13190-97-1
solanesol

-
-
541-47-9
3-Methylbutenoic acid

-
-
1190409-09-6
3-methyl-but-2-enoic acid 3,7,11,15,19,23,27,31,35-nonamethyl-hexatriaconta-2,6,10,14,18,22,26,30,34-nonaen-1-yl ester

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 6h; | 66.3% |
-
-
13190-97-1
solanesol

-
-
830-09-1
3-(4'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 6h; | 64% |

| Conditions | Yield |
|---|---|
| Stage #1: solanesol; 2,3-dimethoxy-5-methylbenzene-1,4-diol With phosphotungstic acid In nitromethane; n-heptane; water at 40℃; for 3h; Stage #2: With acetic acid; silver(l) oxide In n-heptane at 20℃; for 1h; Product distribution / selectivity; | 63% |
| With acetic acid; zinc(II) chloride Behandeln des Reaktionsprodukts mit Ag2O in Aether; |
-
-
13190-97-1
solanesol

-
-
35896-58-3
2,3,4,5-tetramethoxytoluene

| Conditions | Yield |
|---|---|
| With bismuth(lll) trifluoromethanesulfonate In nitromethane; hexane at 50℃; for 12h; Product distribution / selectivity; | 62.9% |
| With scandium tris(trifluoromethanesulfonate) In nitromethane; hexane at 50℃; for 16h; Product distribution / selectivity; | 32% |
| With bismuth(lll) trifluoromethanesulfonate In nitromethane; n-heptane at 50℃; for 12h; Product distribution / selectivity; |
-
-
13190-97-1
solanesol

-
-
6099-03-2
3-(2'-methoxyphenyl)propenoic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 6h; | 61% |
-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 6h; | 60% |
-
-
90-50-6
3,4,5-trimethoxycinnamic acid

-
-
13190-97-1
solanesol

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 6h; | 60% |
-
-
13190-97-1
solanesol

-
-
39068-88-7
2,3,4-trimethoxy-6-methylphenol

-
-
110582-10-0
2,3,4-trimethoxy-6-methyl-5-solanesylphenol

| Conditions | Yield |
|---|---|
| With scandium tris(trifluoromethanesulfonate) In nitromethane; hexane at 50℃; for 16h; Product distribution / selectivity; | 51% |
| With bismuth(lll) trifluoromethanesulfonate In nitromethane; n-heptane at 50℃; for 12h; Product distribution / selectivity; |
-
-
13190-97-1
solanesol

-
-
539-47-9
3-(2-furyl)acrylic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 6h; | 50% |

| Conditions | Yield |
|---|---|
| Stage #1: solanesol With pyridine; dmap In dichloromethane at 5℃; for 0.166667h; Sealed tube; Inert atmosphere; Stage #2: methyl chloroformate In dichloromethane at 5 - 20℃; for 1.16667h; Sealed tube; Inert atmosphere; | 46% |
-
-
13190-97-1
solanesol

-
-
3066-90-8
2,3-dimethoxy-5-methylbenzene-1,4-diol

-
A
-
303-97-9
ubiquinone-45

-
B
-
5677-54-3
ubiquinone-9

| Conditions | Yield |
|---|---|
| With scandium tris(trifluoromethanesulfonate) In nitromethane; hexane at 50℃; for 16h; | A 11.6% B 40.4% |


| Conditions | Yield |
|---|---|
| Stage #1: solanesol; tetrabutylammonium dihydrogen phosphate With trichloroacetonitrile In dichloromethane at 20℃; for 1h; Inert atmosphere; Darkness; Stage #2: With ammonium hydroxide In tetrahydrofuran; water at 20℃; for 14h; Inert atmosphere; | 33% |

| Conditions | Yield |
|---|---|
| Stage #1: solanesol With tetrabutylammonium dihydrogen phosphate; trichloroacetonitrile In dichloromethane at 20℃; for 14h; Darkness; Stage #2: ammonium hydroxide In tetrahydrofuran; dichloromethane at 20℃; for 0.5h; | 32% |

| Conditions | Yield |
|---|---|
| Stage #1: solanesol; tetrabutylammonium dihydrogen phosphate With trichloroacetonitrile In dichloromethane at 20℃; for 14h; Inert atmosphere; Darkness; Stage #2: With ammonium hydroxide In tetrahydrofuran; methanol; water; toluene at 20℃; for 1h; Inert atmosphere; | 27% |
-
-
13190-97-1
solanesol

-
-
22205-68-1
3,4,5-triiodobenzoyl chloride

-
-
124159-62-2
3,4,5-triiodo-benzoic acid-(3,7,11,15,19,23,27,31,35-nonamethyl-hexatriaconta-2t,6t,10t,14t,18t,22t,26t,30t,34-nonaenyl ester)
-
-
13190-97-1
solanesol

-
-
28973-76-4, 55528-85-3
solanesylaldehyd

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide |
-
-
13190-97-1
solanesol

-
-
15575-04-9
decaprenol

| Conditions | Yield |
|---|---|
| ueber mehrere Zwischenstufen; | |
| Multi-step reaction with 5 steps 1: PBr3 / pyridine / diethyl ether; petroleum ether / 2 h / 0 °C 2: Na / ethanol / 1,) 0 deg C, 3 h, 2.) 60 deg C, 4 h, 3.) r.t., overnight 3: 10percent aq. NaOH / ethanol / 1.) r.t., 2 h, 2.) 60 deg C, 3 h 4: 89 percent / NaH / tetrahydrofuran / 5 h / Heating 5: i-Bu2AlH / petroleum ether; cyclopentane / 3 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: 1.1 g / PBr3 / tetrahydrofuran / 0.42 h / -20 °C 2: 70 percent / 1.) n-BuLi / tetrahydrofuran; hexane / 1.) -70 deg C, 40 min, 2.) 40 min 3: Na / ethanol; tetrahydrofuran / 0.25 h / -40 °C View Scheme |
-
-
13190-97-1
solanesol

-
-
77621-63-7
2,6,10,14,18,22,26,30,34-nonamethyl-hexatriacontane

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; ethanol Hydrogenation; |
-
-
13190-97-1
solanesol

-
-
70495-48-6
3,7,11,15,19,23,27,31,35-nonamethyl-hexatriacontan-1-ol

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal Hydrogenation; |
-
-
13190-97-1
solanesol

-
-
108193-67-5
3,5-dinitro-benzoic acid-(3,7,11,15,19,23,27,31,35-nonamethyl-hexatriaconta-2t,6t,10t,14t,18t,22t,26t,30t,34-nonaenyl ester)
-
-
13190-97-1
solanesol

-
-
641-70-3
3-nitrophthalic acid anhydride
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View