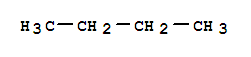Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:463-82-1
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:463-82-1
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:463-82-1
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:Colorless gas or the highly volatile liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request.
DB BIOTECH CO., LTD
Q1: Are you a manufacturer Answer: Yes, we are factory founded on 2012.Q2: How to contact with us Click "contact supplier" And then send us message the product you interest in, you will get reply within 12 hours.Q3:Which kind of payment terms do you
Henan Tianfu Chemical Co., Ltd.
1.Our services:A.Supply sampleB.The packing also can be according the customers` requirmentC.Any inquiries will be replied within 24 hoursD.we provide Commerical Invoice, Packing List, Bill of loading, COA , Health certificate and Origin certificate.
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:463-82-1
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryWuhan ZeShanCheng Biomedical Technology Co., Ltd.
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highl
Hunan Longxianng Runhui Trading Co.,Ltd
2,2-dimethylpropaneAppearance:ask Storage:Keep in dry and cool condition Package:foil aluminium bag/vacuum packing Application:intermediates Transportation:By express (Door to door) such as FEDEX, DHL, EMS for small amount. By air(airport to airport)
Xi'an Harmonious Natural Bio-Technology Co., Ltd.
Before-Sale Service:---Any inquiries will be replied within 12 hours.---Golden Manufactory under?ISO/GMP?with?large stock.---Dedication to quality,We?have?strict?quality?control?system.?---OEM/ODM Available.---Sample is available for your evaluation
Cas:463-82-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHuBei Ipure Biotech CO.,ltd
HuBei ipure is a diversified product production and operation enterprise, with API, pharma intermediates, and other fine chemicals as well as R&D and pigments development and sales as one of the large enterprises, with more than 130 acres of pl
Cas:463-82-1
Min.Order:1 Kilogram
FOB Price: $1.0 / 2.0
Type:Trading Company
inquiryShanghai united Scientific Co.,Ltd.
United Scientific Company Located in Shanghai of China , is a competitive player in the global specialty and fine chemical market. Fenghua has both the expertise and flexibility to produce a wide range of chemicals. Focusing on developing the innovat
Shanghai AngewChem Co., Ltd.
Shanghai AngewChemCo., Ltd. is an innovative enterprise on fine chemicals and pharmaceuticals. Based on Shanghai R&D center and Hunan chemical manufacturing plant, we offer chemical research, process development, and large-scale production. Complete
Hebei mingxing biotech co.,ltd
Known for its best quality and competitve price, this chemicals we offered is widely appreciated by our customers. Prompt reaction, good quality and best service make us reliable and outstanding in this industry.Appearance:yellow to white solid or po
Nanjing Raymon Biotech Co., Ltd.
2,2-dimethylpropaneAppearance:Pls see the Details Storage:keep in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/pharma intermediate Transportation:By Sea,by Air,By courier like DHL or Fedx. Port:Sh
Hebei Minshang Biotechnology Co., Ltd
2,2-dimethylpropane Application:intermediates
Wuxi Morality Chemical Co., Ltd
Do best quality products, erect the morality model Application:Please email us, thanks
Shanghai Yanchu chemicals Co., Ltd
low price and good quality Application:It is an important raw material and intermediate used in Organic Synthesis, Pharmaceuticals, Agrochemicals and D
Hebei Ruishun Trade Co.,Ltd
Supply top quality products with a reasonable price Application:api
Chemical Co.Ltd
Manufacturer,strong R&D,professional team Storage:Store in a cool, dry place. Store in a tightly closed container. Package:according to your requirement Application:ZhiShang Chemical is owned by ZhiShang Group, is a professional new-type chemicals en
NovaChemistry
high purity Application:Drug intermediates Materials intermediates and active molecules
Synthetic route

-
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In benzene-d6 Kinetics; thermolysis at 167 +/- 1°C; | 100% |
-
-
116563-64-5
trans-neopentyl(trifluoromethanesulfonato)bis(trimethylphosphine)platinum(II)

-
-
108-88-3
toluene

-
C
-
463-82-1
2,2-dimethylpropane

-
D
-
1630-94-0
1,1-dimethylcyclopropane

| Conditions | Yield |
|---|---|
| In toluene Kinetics; thermolysis in toluene at 133°C for 80 min (Ar or N2); | A n/a B n/a C 99% D 1% |

-
-
34557-54-5
methane

-
-
586367-34-2
[(η5-pentamethylcyclopentadienide)Sc(CH2C(CH3)3]

-
A
-
99707-15-0
(eta.5-C5Me5)2ScCH3

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In Cyclohexane-d12 Kinetics; under Ar, in the dark; in NMR tube; CH4 added to soln. of Sc complex in cyclohexane-d12 at 77 K; warmed to 11-50°C; not isolated; monitored by (1)H NMR spectra; | A 99% B 99% |
| In benzene-d6 Kinetics; under Ar, in the dark; in NMR tube; CH4 added to soln. of Sc complex in benzene-d6 at 77 K; warmed to 11-50°C; not isolated; monitored by (1)H NMR spectra; | A 99% B 99% |


| Conditions | Yield |
|---|---|
| In diethyl ether under Ar; soln. of t-BuOH in Et2O added to In compd. (molar ratio 1:1) in Et2O at room temp.; stirred for 1 h; volatiles removed by vac. distn.; recrystd. from Et2O at -40°C; crystals washed with pentane at 0°C; elem. anal.; | A 0% B 98.3% |


-
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| With hydrogenchloride In toluene protonolysis with gaseous HCl; | 98% |

| Conditions | Yield |
|---|---|
| In Cyclopentane; cyclohexane (Ar); a soln. of Pt-complex heated at 150°C for 24 min; quenched thermally at -196°C; evapd.; dissolved in acetonitrile;separated; evapd.; | A 98% B n/a |
-
-
110638-22-7
trineopentylaluminum*H2NCH3

-
A
-
110638-25-0
dineopentylaluminum(CH3NH)

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In neat (no solvent) Freshly sublimed ((CH3)3CCH2)3Al*CH3NH2 is heated under protective gas for 10 h at 96°C.; After cooling distn. of neopentane, vac. sublimation of residue at 80°C, elem. anal., mol wt.; | A 97% B n/a |

-
-
111823-35-9
3-methyl-2-isopropyl-1-butene

-
A
-
111848-52-3
{3-methyl-2-(2-propyl)-1-butene}{bis(dicyclohexylphosphino)ethane}platinum(0)

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In cyclohexane Kinetics; thermolysis of cis-hydridoneopentyl{bis(dicyclohexylphosphino)ethane}platinum(II) in 3-methyl-2-(2-propyl)-1-butene/cyclohexane via supposed {bis(dicyclohexylphosphino)ethane}platinum(0) at 69°C to completion; | A 97% B n/a |

-
-
106136-98-5
trineopentylindium

-
-
829-85-6
diphenylphosphane

-
A
-
110138-91-5
((CH3)3CCH2)2In(P(C6H5)2)

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In benzene The mixt. of reagents in benzene was kept at 60°C for 3 d;; evapd., washed with benzene, pentane at -78°C;; | A 69.3% B 96.6% |


| Conditions | Yield |
|---|---|
| With hydrogen; silica gel; palladium at 330℃; | 96% |

-
-
558-37-2
tert-butylethylene

-
A
-
105373-18-0
(3,3-dimethyl-1-butene){bis(dicyclohexylphosphino)ethane}platinum(0)

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In cyclohexane Kinetics; thermolysis of cis-hydridoneopentyl{bis(dicyclohexylphosphino)ethane}platinum(II) in 3,3-dimethyl-1-butene/cyclohexane via supposed {bis(dicyclohexylphosphino)ethane}platinum(0) at 69°C for 2.2 h; | A >99 B 96% |


-
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| With hydrogenchloride In toluene protonolysis with gaseous HCl; | 96% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 0℃; for 1h; | 95% |
-
-
563-79-1
2,3-Dimethyl-2-butene

-
A
-
111848-53-4
(2,3-dimethyl-2-butene){bis(dicyclohexylphosphino)ethane}platinum(0)

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In cyclohexane Kinetics; thermolysis of cis-hydridoneopentyl{bis(dicyclohexylphosphino)ethane}platinum(II) in 2,3-dimethyl-2-butene/cyclohexane via supposed {bis(dicyclohexylphosphino)ethane}platinum(0) at 69°C to completion; | A 94% B >99 |


-
-
2622-14-2
tricyclohexylphosphine

-
A
-
111848-54-5
(tricyclohexylphosphine){bis(dicyclohexylphosphino)ethane}platinum(0)

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In cyclohexane Kinetics; thermolysis of cis-hydridoneopentyl{bis(dicyclohexylphosphino)ethane}platinum(II) in tricyclohexylphosphine/cyclohexane via supposed {bis(dicyclohexylphosphino)ethane}platinum(0) at 68.9°C to completion; | A 94% B >99 |


-
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| With sulfuric acid -10°C; GLC; | 93% |
-
-
15501-33-4
neopentyl iodide

-
A
-
1071-81-4
2,2,5,5-tetramethylhexane

-
B
-
463-82-1
2,2-dimethylpropane

-
C
-
1630-94-0
1,1-dimethylcyclopropane

| Conditions | Yield |
|---|---|
| With tetrabutylammonium perchlorate; nickel(II) perchlorate; triphenylphosphine In acetonitrile at 25℃; | A 92% B 2.5% C 2.5% |


| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at 0℃; for 3h; | 90% |
-
-
87640-51-5
neopentyl(η5-pentamethylcyclopentadienyl)bis(trimethylphosphine)ruthenium

-
-
108-88-3
toluene

-
A
-
114674-67-8
(η5-pentamethylcyclopentadienyl)-m-tolylbis(trimethylphosphine)ruthenium

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In toluene Ar atmosphere, 60°C, 24 h; volatiles removal (vacuum); | A 90% B n/a |


-
-
75-76-3
tetramethylsilane

-
-
105373-20-4
cis-hydrido(trimethylsilylmethyl){bis(dicyclohexylphosphino)ethane}platinum(II)

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In cyclohexane Kinetics; thermolysis of cis-hydridoneopentyl{bis(dicyclohexylphosphino)ethane}platinum(II) in tetramethylsilane/cyclohexane via supposed {bis(dicyclohexylphosphino)ethane}platinum(0) at 69.8°C for 3 h; | A 85% B 83% |

-
-
116563-64-5
trans-neopentyl(trifluoromethanesulfonato)bis(trimethylphosphine)platinum(II)

-
-
1076-43-3
benzene-d6

-
B
-
463-82-1
2,2-dimethylpropane

-
C
-
4741-94-0
neopentane-d1

-
D
-
1630-94-0
1,1-dimethylcyclopropane

| Conditions | Yield |
|---|---|
| In benzene-d6 Kinetics; react. in a sealed 5-mm NMR tube, 133°C, 80 min (2 half-lives) (Ar or N2); | A n/a B 14% C 83% D 3% |


| Conditions | Yield |
|---|---|
| In toluene inert atmosphere, -35°C, 70 h; pptn. of complex (hexane addn.), recrystn. (toluene/hexane); elem. anal.; | A 12% B 83% C 7.3% |


-
A
-
1552-12-1, 111-78-4
1,5-cis,cis-cyclooctadiene

-
B
-
1071-81-4
2,2,5,5-tetramethylhexane

-
C
-
463-82-1
2,2-dimethylpropane

-
D
-
1871-52-9
(1Z,3Z,5Z)-Cycloocta-1,3,5-triene

| Conditions | Yield |
|---|---|
| With tris(1-methylethyl)phosphine In Cyclopentane; cyclohexane 50 deg C,15 h; 126 deg C, 2 h; Further byproducts given; | A 80% B 36 % Chromat. C 106 % Chromat. D 14 % Chromat. |

-
-
110638-21-6
trineopentylaluminum*P(Ph2)H

-
A
-
110638-24-9
dineopentylaluminum{Ph2phosphide}

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| Trineopentylaluminum*P(Ph)2H is heated under protective gas at 180°C for 24 h.; Resulting solid Al-compd. is washed with pentane.; | A 78% B n/a |
-
-
116301-53-2
cis-dioxo ReO2(CH2CMe3)3

-
A
-
123154-26-7
ReO2(CH2tBu)(neopentylidene)

-
B
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In pyridine Irradiation (UV/VIS); photolysis for 1 h, using Pyrex glass-filtered light from a medium pressure mercury lamp;; Re-complex and neopentane were the only identifiable products, isolation of Re-complex by sublimation (40 ° C/ 10 E-4 torr); | A 76% B n/a |
| In acetonitrile Irradiation (UV/VIS); photolysis for 1 h, using Pyrex glass-filtered light from a medium pressure mercury lamp;; isolation of Re-complex by sublimation (40 ° C/ 10 E-4 torr); |

-
A
-
463-82-1
2,2-dimethylpropane

-
B
-
74-85-1
ethene

-
C
-
1630-94-0
1,1-dimethylcyclopropane

-
D
-
563-45-1
3-Methyl-1-butene

-
E
-
115-11-7
isobutene

| Conditions | Yield |
|---|---|
| In toluene inert atmosphere, thermal decompn. (85°C); GLC; | A 5% B 5% C 74% D 4% E 12% |


-
A
-
34557-54-5
methane

-
B
-
463-82-1
2,2-dimethylpropane

-
C
-
78-78-4
methylbutane

-
D
-
1630-94-0
1,1-dimethylcyclopropane

-
E
-
115-11-7
isobutene

| Conditions | Yield |
|---|---|
| With hydrogen In toluene High Pressure; 5 atm H2, room temp., complete reaction at 52°C for 2 h; GLC; | A 4% B 69% C 6% D 14% E 6% |


-
A
-
34557-54-5
methane

-
B
-
74-84-0
ethane

-
C
-
463-82-1
2,2-dimethylpropane

-
D
-
1630-94-0
1,1-dimethylcyclopropane

-
E
-
115-11-7
isobutene

| Conditions | Yield |
|---|---|
| With hydrogen In toluene High Pressure; 5 atm H2, room temp., complete reaction at 52°C for 2 h; i-C4H8 and i-C5H12 also formed; GLC; | A 53% B 7% C 10% D 32% E 62% |


-
-
4127-47-3
2,2,3,3-tetramethylcyclopropane

-
-
105373-22-6
cis-hydrido(2,2,3,3-tetramethylcyclopropyl){bis(dicyclohexylphosphino)ethane}platinum(II)

-
-
111848-55-6
cis-hydrido{(1,2,2-trimethylcyclopropyl)methyl}{bis(dicyclohexylphosphino)ethane}platinum(II)

-
C
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In further solvent(s) Kinetics; thermolysis of cis-hydridoneopentyl{bis(dicyclohexylphosphino)ethane}platinum(II) in 1,1,2,2-tetramethylcyclopropane via supposed {bis(dicyclohexylphosphino)ethane}platinum(0) at 35.0°C for 154 h; | A 56% B 12% C n/a |
| In further solvent(s) Kinetics; thermolysis of cis-hydridoneopentyl{bis(dicyclohexylphosphino)ethane}platinum(II) in 1,1,2,2-tetramethylcyclopropane via supposed {bis(dicyclohexylphosphino)ethane}platinum(0) at 44.9°C for 28.5 h; | A 54% B 12% C n/a |


-
A
-
463-82-1
2,2-dimethylpropane

-
B
-
74-85-1
ethene

-
C
-
1630-94-0
1,1-dimethylcyclopropane

-
D
-
563-45-1
3-Methyl-1-butene

-
E
-
115-11-7
isobutene

| Conditions | Yield |
|---|---|
| With triphenylphosphine In toluene inert atmosphere, thermal decompn. (24°C, 5-fold molar excess PPh3); GLC; | A 2% B 28% C 11% D 7% E 52% |
| In toluene inert atmosphere, thermal decompn. (24°C); GLC; | A 6% B 15% C 47% D 6% E 26% |

-
-
463-82-1
2,2-dimethylpropane

-
-
77-95-2
D-(-)-quinic acid

-
-
32384-42-2
(1S,3R,4S,5R)-3,4-O-isopropylidene-1,5-quinic lactone

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In ethyl acetate at 0 - 78℃; for 12h; | 92% |
-
-
463-82-1
2,2-dimethylpropane


| Conditions | Yield |
|---|---|
| With sulfuric acid In acetone at 20 - 45℃; for 1h; | 90% |
-
-
463-82-1
2,2-dimethylpropane


| Conditions | Yield |
|---|---|
| With sulfuric acid In acetone at 20 - 45℃; for 1h; | 90% |

| Conditions | Yield |
|---|---|
| With dichloromethane; antimony pentafluoride 1.) -78 deg C, 2 h, 2.) RT, 24 h; | 88% |
-
-
463-82-1
2,2-dimethylpropane

-
A
-
34557-54-5
methane

-
B
-
74-84-0
ethane

-
C
-
74-98-6
propane

-
D
-
75-28-5
Isobutane

-
E
-
78-78-4
methylbutane

| Conditions | Yield |
|---|---|
| With hydrogen; platinum at 243℃; Product distribution; further reaction temperatures, catalysts; | A 2.7% B 1.5% C 1.5% D 6.7% E 87.8% |
| at 304℃; Product distribution; also from n-butane, other products,other temperatures, other catalysts; | A 7% B 1.7% C 2.4% D 15.6% E 73.3% |
| With hydrogen; NaY-500; palladium at 216℃; Product distribution; Kinetics; Thermodynamic data; other catalysts, other temperatures; activation energies; | |
| With hydrogen In neat (no solvent) at 275℃; under 1225.5 Torr; Reagent/catalyst; Inert atmosphere; |

-
-
80146-01-6
pentamethylcyclopentadienyliridium(III) PMe3 dihydride

-
-
463-82-1
2,2-dimethylpropane

| Conditions | Yield |
|---|---|
| In neat (no solvent) Irradiation (UV/VIS); dihydride Ir complex and C(CH3)4 irradiating for 5.3 h, cooling to -40°C, filtering, filtrate collecting, solvent removing in vacuo; identified by elem. anal., IR and NMR spectra; | 83% |
| In further solvent(s) Irradiation (UV/VIS); irradn. in neopentane for 5.3 h; | 80% |
| In further solvent(s) Irradiation (UV/VIS); soln. of complex in neopentane was irradiated for 36 h, then heated at 115°C for 21 h (inert atm.); cold chromy.(-80°C) on alumina III column, Et2O-hexane; elem. anal.; | 40% |
| In neat (no solvent) Irradiation (UV/VIS); (1)H-, (13)C-NMR and IR spectroscopy; |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View