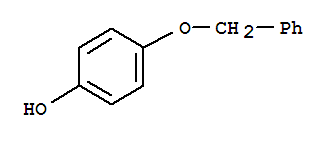
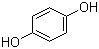
Zhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:53914-95-7
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:53914-95-7
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:53914-95-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:53914-95-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:53914-95-7
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:53914-95-7
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Cas:53914-95-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Cas:53914-95-7
Min.Order:0
Negotiable
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
1.Our services:A.Supply sampleB.The packing also can be according the customers` requirmentC.Any inquiries will be replied within 24 hoursD.we provide Commerical Invoice, Packing List, Bill of loading, COA , Health certificate and Origin certificate.
Cas:53914-95-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:53914-95-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryChangchun Artel lmport and Export trade company
Supply top quality products with a reasonable price Application:api
Henan Kanbei Chemical Co.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Cas:53914-95-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryChemlyte Solutions
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Shanghai Chinqesen Biotechnology Co., Ltd.
Good Quality Package:1kg/bag Application:Medical or chemical Transportation:Air/Train/Sea Port:Shenzhen
ZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Cas:53914-95-7
Min.Order:0
Negotiable
Type:Trading Company
inquiryJilin haofei import and export trade Co.,Ltd
Price, service, company and transport advantage: 1. Best service, place of origin China, high quality, and reasonable price. 2. It's customers' right to choose the package (EMS, DHL, FEDEX, UPS). 3. It's customers' right
Nanjing Raymon Biotech Co., Ltd.
BIS(1,4-PHENYLENE)-34-CROWN 10-ETHER Storage:keep in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/pharma intermediate Transportation:By Sea,by Air,By courier like DHL or Fedx. Port:Shanghai/Shenzh
Cas:53914-95-7
Min.Order:0
Negotiable
Type:Trading Company
inquiryChemical Co.Ltd
Bis(1,4-phenylene)-34-crown 10-EtherAppearance:Off white to slight yellow solid Storage:Stored in shaded, cool and dry places Package:1L 5L 10L 25L bottle Application:pharma intermediate Transportation:Handle with cares to avoid damaging the packages
Cas:53914-95-7
Min.Order:0
Negotiable
Type:Trading Company
inquirySynthetic route
-
-
37860-51-8
tetraethylene glycol di(p-toluenesulfonate)

-
-
14556-10-6
1,11-bis<4-hydroxyphenoxy>-3,6,9-trioxaundecane

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran for 120h; Heating; | 25% |
| With tetra-(n-butyl)ammonium iodide; caesium carbonate; cesium 4-methylbenzenesulfonate 1.) DMF, 80 deg C, 1.5 h, 2.) DMF, 100 deg C, 4 d; Yield given. Multistep reaction; |
-
-
134881-77-9
1,4-bis[2-(2-(2-(2-toluene-p-sulfonylethoxy)ethoxy)ethoxy)ethoxy]benzene

-
-
123-31-9
hydroquinone

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 48h; Heating; | 25% |
| With caesium carbonate for 72h; Cyclization; Heating; | 15% |
-
-
31255-26-2
1-bromo-2-{2-[2-(2-bromoethoxy)ethoxy]-ethoxy}ethane

-
-
14556-10-6
1,11-bis<4-hydroxyphenoxy>-3,6,9-trioxaundecane

-
A
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
B
-
134940-41-3
1,4,7,10,13,18,21,24,27,30,35,38,41,44,47,52,55,58,61,64-icosaoxa<13,13,13,13>tetraparacyclophane

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 80℃; for 48h; | A 18% B 0.23% |

-
-
37860-51-8
tetraethylene glycol di(p-toluenesulfonate)

-
-
123-31-9
hydroquinone

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| Stage #1: hydroquinone With sodium hydroxide In water; butan-1-ol for 0.5h; Reflux; Inert atmosphere; Stage #2: tetraethylene glycol di(p-toluenesulfonate) With caesium carbonate In 1,4-dioxane; butan-1-ol for 20h; Reflux; | 12.8% |
| With sodium hydroxide In isopropyl alcohol for 24h; Heating; | 150 mg |
-
-
37860-51-8
tetraethylene glycol di(p-toluenesulfonate)

-
-
113816-32-3
1,11-bis<4-(benzyloxy)phenoxy>-3,6,9-trioxaundecane

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen; sodium hydride 1.) MeOH, Et2O, 2.) Me2NCHO, 70 deg C, 24 h; Yield given. Multistep reaction; |
-
-
135974-61-7
1,4-bis[2-(2-hydroxyethoxy)ethoxy]benzene bis(4-methylbenzenesulfonate)

-
-
35648-87-4
1,4-bis[2-(2-hydroxyethoxy)ethoxy]benzene

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| With sodium hydride 1.) THF, 1 h, 2.) THF, reflux 4 d; Yield given. Multistep reaction; |

-
A
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In dimethylsulfoxide-d6 at 100℃; Rate constant; |
-
-
113816-15-2
C28H40O10*C12H14N2(2+)*2F6P(1-)

-
A
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
B
-
67994-95-0
N,N'-dimethyl-4,4'-bipyridinium dihexafluorophosphate

| Conditions | Yield |
|---|---|
| In acetone at 25℃; Equilibrium constant; slipping-off; |

-
A
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In [D3]acetonitrile at 50℃; Activation energy; slipping-off; |
-
-
37860-51-8
tetraethylene glycol di(p-toluenesulfonate)

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 80 percent / K2CO3 / acetone / 48 h / Heating 2: 25 percent / K2CO3 / acetone / 48 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: 68 percent / NaH / dimethylformamide / 24 h / 80 °C 2: 1.) H2, Pd/C, 2.) NaH / 1.) MeOH, Et2O, 2.) Me2NCHO, 70 deg C, 24 h View Scheme | |
| Multi-step reaction with 3 steps 1: 1.) NaH / 1.) DMF, 15 min, 2.) DMF, 80 deg C, 24 h 2: 99 percent / H2 / 10percent Pd/C / methanol; CHCl3 3: 25 percent / NaH / tetrahydrofuran / 120 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 80 percent / K2CO3 / acetone / 48 h / Heating 2: 25 percent / K2CO3 / acetone / 48 h / Heating View Scheme |
-
-
103-16-2
4-Benzyloxyphenol

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 68 percent / NaH / dimethylformamide / 24 h / 80 °C 2: 1.) H2, Pd/C, 2.) NaH / 1.) MeOH, Et2O, 2.) Me2NCHO, 70 deg C, 24 h View Scheme | |
| Multi-step reaction with 3 steps 1: 1.) NaH / 1.) DMF, 15 min, 2.) DMF, 80 deg C, 24 h 2: 99 percent / H2 / 10percent Pd/C / methanol; CHCl3 3: 25 percent / NaH / tetrahydrofuran / 120 h / Heating View Scheme |
-
-
123-31-9
hydroquinone

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 63 percent / K2CO3 / dimethylformamide / 18 h / Ambient temperature 2: 68 percent / NaH / dimethylformamide / 24 h / 80 °C 3: 1.) H2, Pd/C, 2.) NaH / 1.) MeOH, Et2O, 2.) Me2NCHO, 70 deg C, 24 h View Scheme | |
| Multi-step reaction with 2 steps 1: 1.) K2CO3 / 1.) DMF, 30 min, 2.) DMF, 75 deg C, 7 d 2: 1.) NaH / 1.) THF, 1 h, 2.) THF, reflux 4 d View Scheme | |
| Multi-step reaction with 3 steps 1: 1.) K2CO3 / 1.) DMF, 30 min, 2.) DMF, 75 deg C, 7 d 2: 88 percent / triethylamine / CH2Cl2 / 15 h / Ambient temperature 3: 1.) NaH / 1.) THF, 1 h, 2.) THF, reflux 4 d View Scheme |
-
-
113816-32-3
1,11-bis<4-(benzyloxy)phenoxy>-3,6,9-trioxaundecane

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 99 percent / H2 / 10percent Pd/C / methanol; CHCl3 2: 25 percent / NaH / tetrahydrofuran / 120 h / Heating View Scheme |
-
-
35648-87-4
1,4-bis[2-(2-hydroxyethoxy)ethoxy]benzene

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 88 percent / triethylamine / CH2Cl2 / 15 h / Ambient temperature 2: 1.) NaH / 1.) THF, 1 h, 2.) THF, reflux 4 d View Scheme |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine |
-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| In dichloromethane | 90% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In methanol; acetone at 20℃; | 90% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In water equimolar soln. of bipyridyl-compound, Pd-complex and macrocycle in H2O was heated at 100°C for 12 d, NH4PF6 was added; | 85% |


-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| Stage #1: 4-[2-(2-{4-[bis-(4-tert-butyl-phenyl)-phenyl-methyl]-phenoxy}-ethoxy)-ethoxy]-pyridine; C62H65BrN2O3(2+)*2F6P(1-); bis(p-phenylene)[34]crown-10 at 20℃; for 4.08333h; Air atmosphere; Neat (no solvent); Stage #2: With ammonium hexafluorophosphate In chloroform; water; acetone | A 8% B 85% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
134815-81-9
C48H40N4(4+)*2C28H40O10*4F6P(1-)

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide In [D3]acetonitrile at 80℃; for 72h; | 84% |

-
-
623-24-5
1,4-bis(bromomethyl)benzene

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide | 80% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
544708-59-0
4-[2-[2-[4-[4-ethylphenyl-bis{4-t-butylphenyl}methyl]phenoxy]ethoxy]ethoxy]benzylbromide


| Conditions | Yield |
|---|---|
| In benzonitrile at 20℃; for 144h; | 75% |

-
-
623-24-5
1,4-bis(bromomethyl)benzene

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
108861-20-7
1,1-[1,4-phenylenebis(methylene)]bis-4,4'-pyridylpiridinium bis(hexafluorophosphate)

-
-
122801-13-2
<2>---catenane tetrakis(hexafluorophosphate)

| Conditions | Yield |
|---|---|
| With ammonium hexafluorophosphate In N,N-dimethyl-formamide under 7500600 Torr; Ambient temperature; | 74% |
| With ammonium hexafluorophosphate at 20℃; catenation; | 70% |
| In N,N-dimethyl-formamide at 80℃; for 1h; | 34% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In acetonitrile heating soln. of platinum compd. and cyclophane deriv. in acetonitrile at 50°C for 7 d; cooling to room temp, filtration, addn. of water and amberlite CG-400, sirring for 24 h at room temp., filtration, addn. of KPF6, filtration, elem. anal.; | 70% |

-
-
553-26-4
4,4'-bipyridine

-
-
31608-22-7
4-bromo-1-(2-tetrahydropyranyloxy)butane

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| Multistep reaction; | 66% |


-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| Stage #1: C44H49BrO3; C54H57N2O3(1+)*F6P(1-); bis(p-phenylene)[34]crown-10 at 20℃; for 2.08333h; Air atmosphere; Neat (no solvent); Stage #2: With ammonium hexafluorophosphate In chloroform; water; acetone | A 20% B 64% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
13362-78-2
trans-1,2-bis(pyridin-4-yl)ethene

-
-
120-80-9
benzene-1,2-diol

-
-
108-88-3
toluene

-
-
73852-19-4
3,5-bis-trifluromethylphenylboronic acid

| Conditions | Yield |
|---|---|
| In toluene DPE, catechol, B compd., and crown (1:2:2:1) heated; cooled to room temp., septd., NMR; | 63% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
120-80-9
benzene-1,2-diol

-
-
73852-19-4
3,5-bis-trifluromethylphenylboronic acid

-
-
1135-32-6
trans-1,2-bis(4-pyridyl)ethylene

-
-
1061570-77-1
C28H40O10*C40H24B2F12N2O4

| Conditions | Yield |
|---|---|
| In benzene for 1h; Reflux; Inert atmosphere; | 63% |


-
-
623-24-5
1,4-bis(bromomethyl)benzene

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide | 61% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
108861-18-3
1,1''-(1,3-phenylene-bis(methylene))bis-4,4'-bipyridinium-bis(hexafluorophosphate)

-
-
626-15-3
1,3-bis-(bromomethyl)benzene

| Conditions | Yield |
|---|---|
| With ammonium hexafluorophosphate In N,N-dimethyl-formamide under 7500600 Torr; for 7h; Ambient temperature; | 59% |

-
-
1020666-50-5
1-{4-[tris(4-t-butylphenyl)-methyl]-phenyl}-4,4'-bipyridinium hexafluorophosphate

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
544708-54-5
4-(2-(2-(4-[tris-{4-t-butylphenyl}-methyl]-phenoxy)-ethoxy)-ethoxy)-benzylbromide


| Conditions | Yield |
|---|---|
| In benzonitrile at 20℃; for 120h; | A 37% B 56% |

-
-
623-24-5
1,4-bis(bromomethyl)benzene

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| With ammonium chloride In N,N-dimethyl-formamide at 25℃; for 336h; Substitution; | 53% |


| Conditions | Yield |
|---|---|
| In acetonitrile at 55℃; for 240h; | 52% |
| In acetonitrile at 24.9℃; Kinetics; var. temp.; |
-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 55℃; for 240h; rotaxane formation; | A 52% B 14% |

-
-
17084-13-8
potassium hexafluorophosphate

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
960068-70-6
(ethylenediamine)Pt(OTf)2

-
-
1141448-00-1
1-(4-(pyridin-4-yl)benzyl)-4,4'-bipyridin-1-ium nitrate

| Conditions | Yield |
|---|---|
| In acetonitrile soln. Pt complex, ligand and polyether in MeCN was heated at 55°Cfor 8 days, cooled to room temp., Et2O was added, ppt. was filtered, su spnd. in water, Amberlite IRA-402 was added, stirred for 24 h; soln. was filtered and evapd. in vacuo, column chromy. on silica (acetone - 1.5M NH4Cl - MeOH 5:4:1), solvent was removed in vacuo, residue was dissolved in water, KPF6 was added, ppt. was filtered and washed with water; elem. anal.; | 52% |


| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 20℃; for 240h; Substitution; | 51% |
| In acetonitrile for 168h; Ambient temperature; | 35% |


-
-
623-24-5
1,4-bis(bromomethyl)benzene

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide | 51% |

-
-
1020666-50-5
1-{4-[tris(4-t-butylphenyl)-methyl]-phenyl}-4,4'-bipyridinium hexafluorophosphate

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
544708-54-5
4-(2-(2-(4-[tris-{4-t-butylphenyl}-methyl]-phenoxy)-ethoxy)-ethoxy)-benzylbromide

-
A
-
1030850-41-9
1-{4-[tris(4-t-butylphenyl)-methyl]phenyl}-1'-(4-(2-(2-(4-[tris-{4-t-butylphenyl}-methyl]-phenoxy)-ethoxy)-ethoxy)-benzyl)-[4,4']-bipyridinium bis-p-phenylene-34-crown-10-12,152-disulphonate

| Conditions | Yield |
|---|---|
| Stage #1: bis(p-phenylene)[34]crown-10 With chlorosulfonic acid at 0℃; for 1h; Stage #2: With tetramethyl ammoniumhydroxide In methanol for 2h; Stage #3: 1-{4-[tris(4-t-butylphenyl)-methyl]-phenyl}-4,4'-bipyridinium hexafluorophosphate; 4-(2-(2-(4-[tris-{4-t-butylphenyl}-methyl]-phenoxy)-ethoxy)-ethoxy)-benzylbromide In acetonitrile at 20℃; for 144h; Further stages.; | A 50% B 50% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
138926-07-5
(N,N’)-diethyl-4,4’-bipyridinium dihexafluorophosphate

-
-
1030850-36-2
1,1'-diethyl-[4,4']-bipyridinium bis-p-phenylene-34-crown-10-12,152-disulphonate sesquiethanolate

| Conditions | Yield |
|---|---|
| Stage #1: bis(p-phenylene)[34]crown-10 With chlorosulfonic acid at 0℃; for 1h; Stage #2: With tetramethyl ammoniumhydroxide In methanol for 2h; Stage #3: (N,N’)-diethyl-4,4’-bipyridinium dihexafluorophosphate In methanol; ethanol Further stages.; | 50% |
-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
108861-18-3
1,1''-(1,3-phenylene-bis(methylene))bis-4,4'-bipyridinium-bis(hexafluorophosphate)

-
-
93824-64-7
3,3'-bis(α-bromomethyl)azobenzene

| Conditions | Yield |
|---|---|
| With ammonium hexafluorophosphate; nitromethane; tetraethylammonium chloride; water In acetonitrile for 192h; Ambient temperature; | 49% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
544708-54-5
4-(2-(2-(4-[tris-{4-t-butylphenyl}-methyl]-phenoxy)-ethoxy)-ethoxy)-benzylbromide



| Conditions | Yield |
|---|---|
| In benzonitrile at 20℃; for 120h; | A 36% B 48% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10


| Conditions | Yield |
|---|---|
| In acetonitrile at 55℃; for 240h; | 47% |
| In acetonitrile at 29.9℃; Kinetics; var. temp.; |
-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In acetonitrile at 50℃; for 240h; | A 26% B 46% C 6% |
| In acetonitrile at 50℃; for 240h; | A 41% B 19% C 22% |

-
-
53914-95-7
bis(p-phenylene)[34]crown-10

| Conditions | Yield |
|---|---|
| In acetonitrile at 55℃; for 240h; | 45% |
| In acetonitrile at 24.9℃; Kinetics; var. temp.; |
-
-
53914-95-7
bis(p-phenylene)[34]crown-10

-
-
13362-78-2
trans-1,2-bis(pyridin-4-yl)ethene

| Conditions | Yield |
|---|---|
| With ammonium hexafluorophosphate In N,N-dimethyl-formamide for 360h; Ambient temperature; | 43% |
| With ammonium hexafluorophosphate In acetonitrile for 120h; Ambient temperature; | 23% |

Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View