Antimex Chemical Limied
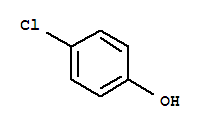
ProName: catechol 120-80-9 CasNo: 120-80-9 Molecular Formula: C6H6O2 Appearance: Light gray or light brown crystal Application: Pharmaceutical Intermediates DeliveryTime: prompt PackAge: 25kg/drum, 200kg/drum Port: Sh
Cas:120-80-9
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:120-80-9
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryDayang Chem (Hangzhou) Co.,Ltd.
Dayangchem’s R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantiti
Cas:120-80-9
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance White granules White granules Assay
Cas:120-80-9
Min.Order:1 Kilogram
FOB Price: $5.0 / 11.0
Type:Manufacturers
inquiryJinan Finer Chemical Co., Ltd
Product description: Product name Pyrocatechol CAS number 120-80-9 Assay ≥99% Appearance Off-white to light gray crystal Capacity 500mt/year Application Rubber hardener, elec
Cas:120-80-9
Min.Order:100 Gram
FOB Price: $12.0
Type:Lab/Research institutions
inquiryHefei TNJ chemical industry co.,ltd
1,ISO Certificate 2,Factory price 3,Free sample and Fast delivery Appearance:White flakes Storage:Storage: All products should be stored at a dry and ventilated location away from fire and other dangerous goods, and should avoid direct sunlight. Pa
Cas:120-80-9
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
The company has a professional R & D team, mature technology, very competitive prices and stable high quality products for customers to order high quality and low price product efforts! Appearance:White or off-white Solid Storage:Sealed, dr
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
shanghai Tauto Biotech Co., Ltd
17-year experience in Phytochemicals Each product has passed very strict tests (NMR\MS\HPLC) Agents in 13 countries Certified by ISO 9001:2008 quality management system Leader Supplier of Botanical Reference Materials in China
Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new ap
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hebei yanxi chemical co.,LTD.
Hebei Yanxi Chemical Co., Ltd. is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low car
Henan Tianfu Chemical Co., Ltd.
Product Name: Pyrocatechol Synonyms: 1,2-Benzenediol (pyrocatechol);1,2-benzenediol[qr];1,2-dihydroxybenzene[qr];2-Hydroxyphenol;2-hydroxyphenol[qr];ai3-03995[qr];Benzene, o-dihydroxy-;benzene,1,2-dihydroxy- CAS: 120-80-9 M
Cas:120-80-9
Min.Order:1 Kilogram
FOB Price: $8900.0
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Appearance: Light gray to light brown crystal . Molecular Formula : C6H6O2 . Molecular weight :110.11. Solubility: Soluble in water, soluble in ethanol, ethyl ether. Application: 1,anti-microbial raw material. 2, Used
Hubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:off-white to similar to white powder Storage:Store in sealed
Cas:120-80-9
Min.Order:100 Gram
Negotiable
Type:Other
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At prese
Triumph International Development Limilted
Appearance:white or yellowish white flaky crystal Storage:Keep the sun from direct sunlight ;prevention against high temperature Package:As customer request Application:Used for research and industrial manufacture. Transportatio
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Afine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:120-80-9
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Intermediates Tr
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:120-80-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China with ISO certificate, Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED
Cas:120-80-9
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
SAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in custom synthe
Beijing Greenchem Technology Co.,Ltd. ( Panjin Greenchem Technology Co.Ltd .)
Capability on chemical synthesis1. Beijing High-Tech Enterprises2. Strong R&D Team3. 8 years of experiences in R & D of high-tech Catalyst;4. 5000 production techniques, 69 items of national patents, and 360 kinds of products on sales;5. The producti
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,or Intermediates,fine chemicals Transportation:air,sea,courier
Synthetic route

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; MgAlZr0.1-HT In Petroleum ether at 79.9℃; for 8h; | 100% |
| With 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In methanol; chloroform at -25℃; for 0.333333h; | 97% |
| With tert.-butylhydroperoxide In water; acetonitrile at 70℃; for 6h; Catalytic behavior; Temperature; Solvent; | 89% |
-
-
2171-74-6
1,3-benzodioxol-2-one

-
-
67-63-0
isopropyl alcohol

-
A
-
6482-34-4
bis-2-propyl carbonate

-
B
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With sodium methylate In neat (no solvent) at 100℃; under 1500.15 Torr; for 5h; Inert atmosphere; Autoclave; | A 67% B 100% |
| With sodium methylate at 100℃; for 5h; Temperature; Autoclave; Inert atmosphere; | A 90 %Chromat. B n/a |


| Conditions | Yield |
|---|---|
| With 1,3-dimethyl-2-imidazolidinone; sodium hexamethyldisilazane In tetrahydrofuran at 185℃; for 12h; further reagent: LDA; | 99% |
| With sodium di(ethyl)amine In N,N,N,N,N,N-hexamethylphosphoric triamide; benzene for 12h; Heating; | 85% |
| With aluminium(III) iodide In carbon disulfide for 7h; Heating; Var. solvent, time and substrate reagent ratio; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-methoxy-phenol With pyridine; iodine; aluminium In acetonitrile for 18h; Reflux; Stage #2: With hydrogenchloride In water; acetonitrile at 20℃; | 99% |
| With hydrogenchloride In water at 250℃; under 37503.8 Torr; for 3h; Reagent/catalyst; Autoclave; Inert atmosphere; Green chemistry; | 97% |
| With aluminium(III) iodide; calcium oxide In acetonitrile at 80℃; for 18h; Reagent/catalyst; | 94% |
-
-
14005-14-2
2,2-dimethyl-1,3-benzodioxole

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In dichloromethane at 20℃; for 18h; Catalytic behavior; Reagent/catalyst; Solvent; | 99% |
| With indium(III) triflate In water; acetonitrile at 120℃; for 0.5h; Microwave irradiation; | 91% |
| With sodium di(ethyl)amine In N,N,N,N,N,N-hexamethylphosphoric triamide; benzene for 12h; Heating; | 88% |
-
-
116915-91-4
6H,11H-dibenzo<14>dioxocin

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With hydrogen; palladium hydroxide - carbon In ethanol under 760 Torr; | 99% |

| Conditions | Yield |
|---|---|
| With glycolic Acid; copper hydroxide; sodium hydroxide In water; dimethyl sulfoxide at 120℃; for 6h; Inert atmosphere; Schlenk technique; | 99% |
| With β-D-glucose; copper(II) acetate monohydrate; potassium hydroxide In water; dimethyl sulfoxide at 20 - 120℃; for 16h; | 83% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide at 20℃; for 0.666667h; Time; Dakin Phenol Oxidation; Green chemistry; | 98% |
| With dihydrogen peroxide In water at 20℃; for 2h; Dakin Phenol Oxidation; Green chemistry; | 98% |
| With 7,8-difluoro-1,3-dimethyl-5-ethyl-4a-hydroperoxyalloxazine; dihydrogen peroxide; sodium hydrogencarbonate In methanol; water at 20℃; for 1h; Dakin oxidation; | 92% |

| Conditions | Yield |
|---|---|
| With methanol; 1,3-disulfonic acid imidazolium hydrogen sulfate at 20℃; for 0.0666667h; Green chemistry; | 98% |
-
-
17600-72-5
2-ethoxyanisole

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 250℃; under 37503.8 Torr; for 3h; Autoclave; Inert atmosphere; Green chemistry; | 98% |
| With water; hydrogen bromide; Aliquat 336 at 105℃; for 9h; Catalytic behavior; | 72% |

| Conditions | Yield |
|---|---|
| With 1,3-dimethyl-2-imidazolidinone; lithium diisopropyl amide In tetrahydrofuran; n-heptane; ethylbenzene at 185℃; for 12h; | 97% |
| With hydrogen iodide at 25℃; for 24h; Inert atmosphere; | 96% |
| With aluminium(III) iodide In acetonitrile for 0.5h; Heating; Var. solvent, time and substrate reagent ratio; | 93% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 250℃; under 37503.8 Torr; for 3h; Sealed tube; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 8-quinolinol; potassium hydroxide In water; dimethyl sulfoxide; tert-butyl alcohol at 100℃; for 48h; Inert atmosphere; | 95% |
| Stage #1: 2-Iodophenol With copper(l) iodide; 1,10-Phenanthroline; potassium hydroxide In water; dimethyl sulfoxide at 20 - 100℃; Inert atmosphere; Stage #2: With hydrogenchloride In water; dimethyl sulfoxide at 20℃; Inert atmosphere; | 86% |
| With basolite C300; potassium hydroxide In water; dimethyl sulfoxide at 125℃; for 12h; | 84% |

| Conditions | Yield |
|---|---|
| With aluminium(III) iodide In dimethyl sulfoxide; acetonitrile at 80℃; for 18h; | 95% |
| With aluminium(III) iodide; dimethyl sulfoxide In acetonitrile at 80℃; for 18h; | 95% |
| With aluminium(III) iodide; N,N-dimethyl-formamide dimethyl acetal In acetonitrile at 80℃; for 18h; | 72% |
| With aluminium(III) iodide; N,N-dimethyl-formamide dimethyl acetal In acetonitrile at 80℃; for 18h; | 72% |
| With oxygen; copper(II) perchlorate; ascorbic acid In water; acetone at 60℃; for 20h; | 25% |

| Conditions | Yield |
|---|---|
| With boron dimethyl-trifluoro sulphide In dichloromethane at 0℃; for 0.0833333h; | 95% |

| Conditions | Yield |
|---|---|
| With ammonium bicarbonate In water at 20℃; for 2h; Schlenk technique; | 95% |
-
-
33130-24-4
2-(2-phenylethoxy)phenol

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With aluminium(III) iodide In dimethyl sulfoxide; acetonitrile at 80℃; for 18h; | 95% |
| With aluminium(III) iodide; dimethyl sulfoxide In acetonitrile at 80℃; for 18h; | 95% |
| With aluminium(III) iodide In dimethyl sulfoxide; acetonitrile at 80℃; for 18h; | 93% |

| Conditions | Yield |
|---|---|
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; malonic acid In methanol at 25℃; for 12h; Catalytic behavior; Reagent/catalyst; Temperature; Solvent; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With aluminium(III) iodide; diisopropyl-carbodiimide In acetonitrile at 80℃; for 18h; | 94% |
| With aluminium(III) iodide; diisopropyl-carbodiimide In acetonitrile at 80℃; for 18h; | 94% |
| With aluminium(III) iodide; calcium oxide In acetonitrile at 80℃; for 18h; | 94% |

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene | 93% |

| Conditions | Yield |
|---|---|
| With aluminium(III) iodide; dimethyl sulfoxide In acetonitrile at 80℃; for 18h; | 93% |
| With aluminium(III) iodide; N,N-dimethyl-formamide dimethyl acetal In acetonitrile at 80℃; for 18h; | 63% |
| With aluminium(III) iodide; N,N-dimethyl-formamide dimethyl acetal In acetonitrile at 80℃; for 18h; | 63% |

| Conditions | Yield |
|---|---|
| With Na-X zeolite In water at 200℃; for 1h; Autoclave; | 91% |
| With Cocos nucifera juice at 20℃; for 48h; Inert atmosphere; | 88% |
| With cucumber juice at 30 - 35℃; for 48h; Inert atmosphere; Green chemistry; | 85% |

| Conditions | Yield |
|---|---|
| With Nafion-H; toluene for 2h; Heating; | A 97 % Chromat. B 91% |
-
-
98-29-3
4-tert-Butylcatechol

-
-
108-88-3
toluene

-
A
-
98-51-1
4-tert-butyltoluene

-
B
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With Nafion-H for 2h; Heating; | A 97 % Chromat. B 91% |

-
-
165105-99-7
4-oxo-cyclohexyl 2,2-dimethylpropanoate

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With iodine; oxygen; dimethyl sulfoxide at 80℃; for 12h; | 91% |

| Conditions | Yield |
|---|---|
| With aluminium(III) iodide In acetonitrile for 1h; Heating; | 90% |
| With sulphurous acid | |
| Rate constant; pH 7.00; reaction with substrate reduced glucose oxidase; |

| Conditions | Yield |
|---|---|
| With sodium percarbonate In tetrahydrofuran; water; N,N-dimethyl-formamide for 8h; ultrasonication; | 90% |
| With sulfuric acid; dihydrogen peroxide; boric acid In tetrahydrofuran; water at 20℃; for 36h; Oxidation; | 90% |
-
-
75724-54-8
1-acetoxy-6-oxocyclohexa-2,4-dienyl propanoate

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether for 1h; Heating; | 90% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In benzene for 4h; Solvent; Time; Temperature; Reflux; | 90% |

| Conditions | Yield |
|---|---|
| In water at 210℃; for 0.25h; Microwave irradiation; | 100% |
| at 200℃; | |
| at 200 - 210℃; in geschlossenen Rohr; | |
| With potassium dichromate; acetic acid for 24h; Reflux; |

| Conditions | Yield |
|---|---|
| at 80℃; for 1h; | 100% |
| for 0.166667h; Schlenk technique; | 96% |
| In dichloromethane; ethyl acetate at 20℃; | 81% |
-
-
120-80-9
benzene-1,2-diol

-
-
17345-77-6
1-bromo-3,4-dihydroxybenzene

| Conditions | Yield |
|---|---|
| With benzyltriphenylphosphonium peroxodisulfate; potassium bromide In acetonitrile for 3.5h; Heating; | 100% |
| With tetrafluoroboric acid diethyl ether; N-Bromosuccinimide In acetonitrile at -30 - 20℃; | 100% |
| With N-Bromosuccinimide; tetrafluoroboric acid In diethyl ether; acetonitrile regioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With 2,2'-bipyridylchromium peroxide In benzene for 0.5h; Product distribution; Heating; effect of various chromium(VI) based oxidants; | 100% |
| With barium ferrate(VI) In benzene for 0.7h; Product distribution; Heating; | 100% |
| With 2,2'-bipyridylchromium peroxide In benzene for 0.5h; Heating; | 100% |
-
-
33842-02-3, 529510-96-1
dichloromethylenedimethyliminium chloride

-
-
120-80-9
benzene-1,2-diol

-
-
36156-21-5
Benzo[1,3]dioxol-2-ylidene-dimethyl-ammonium; chloride

| Conditions | Yield |
|---|---|
| at -20℃; for 1h; ball mill; | 100% |
| In dichloromethane for 1h; Heating; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 48h; Williamson ether synthesis; Reflux; | 100% |
| With potassium carbonate In acetone for 48h; Heating; | 95% |
| With potassium carbonate; potassium iodide In acetone for 48h; Reflux; | 95% |
-
-
120-80-9
benzene-1,2-diol

-
-
105-36-2
ethyl bromoacetate

-
-
52376-09-7
ethyl 2-<2-<(ethoxycarbonyl)methoxy>phenoxy>acetate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 120h; | 100% |
| With N-benzyl-N,N,N-triethylammonium chloride; potassium carbonate In acetonitrile for 8h; Heating; | 95% |
| With potassium carbonate In N,N-dimethyl-formamide at 75 - 80℃; for 1.5h; | 90% |
-
-
120-80-9
benzene-1,2-diol

-
-
142841-88-1
1-(benzenesulfonyl)-3-methylimidazolium triflate

-
-
3905-43-9
1,2-bis<(benzenesulfonyl)oxy>benzene

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole In tetrahydrofuran 1) 0 deg C, 30 min, 2) RT, 6 h; | 100% |
-
-
72142-16-6
(3-methyl-2-butenyl)trimethoxysilane

-
-
120-80-9
benzene-1,2-diol

-
-
121-44-8
triethylamine

-
-
114571-77-6
triethylammonium bis(catecholato)(3-methyl-2-butenyl)siliconate

| Conditions | Yield |
|---|---|
| at 45℃; for 5h; | 100% |

-
-
2551-83-9
allyltrimethoxysilane

-
-
120-80-9
benzene-1,2-diol

-
-
121-44-8
triethylamine

-
-
114612-18-9
triethylammonium bis(pyrocatecholato)allylsilicate

| Conditions | Yield |
|---|---|
| for 5h; room temperature to 40 deg C; | 100% |
| at 40℃; for 4h; Inert atmosphere; Sealed tube; | 55% |

-
-
125715-25-5
(2-methyl-2-propenyl)trimethoxysilane

-
-
120-80-9
benzene-1,2-diol

-
-
121-44-8
triethylamine

-
-
125715-30-2
triethylammonium bis(catecholato)(2-methyl-2-propenyl)siliconate

| Conditions | Yield |
|---|---|
| at 45℃; for 5h; | 100% |


| Conditions | Yield |
|---|---|
| at 45℃; for 5h; | 100% |

-
-
120-80-9
benzene-1,2-diol

-
-
161868-74-2
1,1-dichloro-5-aza-2,8-dioxa-1-phosphaV-dibenzo<9,9',11,11'-tetra-tert-butyl>-bicyclo<3.3.0>octadiene

| Conditions | Yield |
|---|---|
| In benzene for 24h; Ambient temperature; | 100% |
-
-
64248-62-0
3,4-Difluorobenzonitrile

-
-
120-80-9
benzene-1,2-diol

-
-
234113-44-1
2-cyanodibenzo[1,4]dioxine

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide; toluene at 125 - 130℃; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide; toluene at 130 - 135℃; Etherification; cyclization; | 98% |
-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| With ozone In water at 20℃; for 0.1h; pH=2; Oxidation; Formation of xenobiotics; | 100% |
-
-
120-80-9
benzene-1,2-diol

-
-
791-28-6
Triphenylphosphine oxide

-
-
62785-50-6
triphenyl(1,2-phenylenedioxy)phosphorane

| Conditions | Yield |
|---|---|
| at 70 - 80℃; for 2h; | 100% |
-
-
866252-03-1
(E)-(2-(phenyldiazenyl)phenyl)boronic acid

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| In toluene for 4h; Heating; | 100% |
-
-
120-80-9
benzene-1,2-diol

-
-
19812-60-3
tetraethylene glycol monoacrylate

| Conditions | Yield |
|---|---|
| Stage #1: benzene-1,2-diol With Trimethyl borate In acetonitrile at 60℃; for 1h; Stage #2: tetraethylene glycol monoacrylate In acetonitrile for 2h; Further stages.; | 100% |

| Conditions | Yield |
|---|---|
| react. of the educts in a molar ratio of 1:1; | A 100% B n/a |

-
-
11113-50-1
boric acid

-
-
120-80-9
benzene-1,2-diol

-
-
791-31-1
triphenylhydroxysilane

-
-
82172-55-2
2-triphenylsiloxy-1,3,2-benzodioxaborole

| Conditions | Yield |
|---|---|
| In benzene byproducts: H2O; azeotropic removal of water; elem. anal.; | 100% |


| Conditions | Yield |
|---|---|
| In acetonitrile stirring of catechol and B(OMe)3 at 60 °C for 1h, addn of tetraethylene glycol monoacrylate, 2 h stirring; cooling, vac. evapn. at 40 °C for 24 h; | 100% |

-
-
1048382-97-3
(E)-dihydroxy(2-{[4-(trifluoromethyl)phenyl]azo}phenyl)borane

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| In toluene for 16h; Heating; | 100% |
-
-
120-80-9
benzene-1,2-diol

-
-
101-83-7
N-cyclohexyl-cyclohexanamine

-
-
76128-65-9
N-(trimethoxysilylmethyl)hexahydroazepin-2-one

| Conditions | Yield |
|---|---|
| In o-xylene at 130 - 140℃; | 100% |

-
-
112-35-6
triethylene glucol monomethyl ether

-
-
6867-35-2
lithium tetramethanolatoborate

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| Stage #1: triethylene glucol monomethyl ether; lithium tetramethanolatoborate for 24h; Reflux; Inert atmosphere; Stage #2: benzene-1,2-diol for 24h; Reflux; Inert atmosphere; | 100% |

-
-
480424-86-0
9,9-didodecylfluorene-2,7-diboronic acid

-
-
120-80-9
benzene-1,2-diol

-
-
1446012-94-7
C49H64B2O4

| Conditions | Yield |
|---|---|
| In toluene Reflux; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| In toluene Dean-Stark; Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With sodium sulfate In toluene at 50℃; Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dimethylsulfoxide-d6 at 20℃; for 1h; | 100% |
-
-
5419-55-6
Triisopropyl borate

-
-
22348-32-9, 63401-04-7, 112022-88-5, 112068-01-6
(S)-diphenylprolinol

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| In toluene at 80℃; for 1h; Inert atmosphere; | 100% |
| Stage #1: Triisopropyl borate; benzene-1,2-diol In toluene Inert atmosphere; Reflux; Stage #2: (S)-diphenylprolinol In toluene at 80℃; for 1h; Inert atmosphere; Reflux; | 100% |

Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View














 Xn
Xn