 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route
-
-
75-62-7
Bromotrichloromethane

-
-
110838-39-6
N,N,N',N'-Tetraisopropyl-P-methylphosphonous diamide

-
A
-
67-66-3
chloroform

-
B
-
124862-13-1
P-(bromomethyl)-N,N,N',N'-tetraisopropylphosphonous diamide

| Conditions | Yield |
|---|---|
| In diethyl ether for 0.25h; Ambient temperature; | A 100% B 45% |
| In diethyl ether for 0.25h; Ambient temperature; or P-ethyl-N,N,N',N'-tetraisopropylphosphonous diamide; | A n/a B 45% |

-
-
75-62-7
Bromotrichloromethane

-
-
122691-44-5
P-ethyl-N,N,N',N'-tetraisopropylphosphonous diamide

-
A
-
67-66-3
chloroform

-
B
-
124862-16-4
P-(1-bromoethyl)-N,N,N',N'-tetraisopropylphosphonous diamide

| Conditions | Yield |
|---|---|
| In diethyl ether for 0.25h; Ambient temperature; | A 100% B 45% |

-
-
116-16-5
1,1,1,3,3,3-hexachloro-propan-2-one

-
-
75-31-0
isopropylamine

-
A
-
67-66-3
chloroform

-
B
-
23144-67-4
N-Isopropyl-2,2,2-trichloroacetamide

| Conditions | Yield |
|---|---|
| In hexane | A n/a B 100% |

-
-
1768-31-6
pentachloroacetone

-
-
75-31-0
isopropylamine

-
A
-
67-66-3
chloroform

-
B
-
39063-24-6
N-Isopropyl-2,2-dichloroacetamide

| Conditions | Yield |
|---|---|
| In hexane for 0.5h; | A n/a B 100% |

-
-
116-16-5
1,1,1,3,3,3-hexachloro-propan-2-one

-
-
109-73-9
N-butylamine

-
A
-
67-66-3
chloroform

-
B
-
31464-96-7
trichloro-acetic acid butylamide

| Conditions | Yield |
|---|---|
| In hexane | A n/a B 100% |

-
-
1768-31-6
pentachloroacetone

-
-
74-89-5
methylamine

-
A
-
67-66-3
chloroform

-
B
-
5345-73-3
2,2-dichloro-N-methylacetamide

| Conditions | Yield |
|---|---|
| In hexane Heating; | A n/a B 100% |

-
-
64697-39-8
1,1,1,3,3-pentachlorobutanone

-
-
74-89-5
methylamine

-
A
-
67-66-3
chloroform

-
B
-
83703-95-1
2,2-Dichloro-N-methyl-propionamide

| Conditions | Yield |
|---|---|
| In hexane Heating; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| With tetrachloromethane In toluene under Ar; excess of CCl4 added to suspension of Cp3ZrH in PhMe; stirredat about 20°C for 5 h; stored overnight at 0-5°C; sepd. by pressure; dried in an Ar stream; elem. anal.; | A 99% B 89% |
-
-
116-16-5
1,1,1,3,3,3-hexachloro-propan-2-one

-
-
76-37-9
2,2,3,3-tetrafluoropropanol

-
A
-
67-66-3
chloroform

-
B
-
1422-70-4
bis(2,2,3,3-tetrafluoropropyl) carbonate

| Conditions | Yield |
|---|---|
| With potassium fluoride; zirconium(IV) oxide at 140℃; for 10h; Product distribution / selectivity; pressure tight reactor; | A 99% B 99% |

-
-
116-16-5
1,1,1,3,3,3-hexachloro-propan-2-one

-
-
75-89-8
2,2,2-trifluoroethanol

-
A
-
67-66-3
chloroform

-
B
-
1513-87-7
bis(2,2,2-trifluoroethyl) carbonate

| Conditions | Yield |
|---|---|
| With potassium fluoride; zirconium(IV) oxide at 140℃; for 10h; pressure tight reactor; | A 99% B 99% |

-
-
55596-11-7
N-(2,2,2-trichloroethylidene)benzenesulfonamide

-
-
21326-49-8
complex of sulfur dioxide with dimethylamine

-
A
-
127-18-4
1,1,2,2-tetrachloroethylene

-
B
-
67-66-3
chloroform

-
C
-
13707-43-2
N,N-dimethyl-N′-(phenylsulfonyl)formimidamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 24h; | A n/a B n/a C 98% |

-
-
55596-11-7
N-(2,2,2-trichloroethylidene)benzenesulfonamide

-
-
109-89-7
diethylamine

-
A
-
127-18-4
1,1,2,2-tetrachloroethylene

-
B
-
67-66-3
chloroform

-
C
-
29665-24-5
N1,N1-diethyl-N2-phenylsulfonylformamidine

| Conditions | Yield |
|---|---|
| In dichloromethane at 0℃; for 24h; | A n/a B n/a C 98% |


| Conditions | Yield |
|---|---|
| With tetrachloromethane In toluene under Ar; elem. anal.; | A 98% B 62% |
-
-
42521-52-8
Trichlormethansulfinsaeureanilid

-
A
-
67-66-3
chloroform

-
B
-
222851-56-1
N-phenylsulfinylamine

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile Ambient temperature; | A n/a B 97.2% |
-
-
116-16-5
1,1,1,3,3,3-hexachloro-propan-2-one

-
-
107-11-9
1-amino-2-propene

-
A
-
67-66-3
chloroform

-
B
-
39089-56-0
2,2,2-trichloro-N-(2-propenyl)acetamide

| Conditions | Yield |
|---|---|
| In hexane for 0.5h; | A n/a B 97% |

-
-
116-16-5
1,1,1,3,3,3-hexachloro-propan-2-one

-
-
141-43-5
ethanolamine

-
A
-
67-66-3
chloroform

-
B
-
35234-31-2
2,2,2-trichloro-N-(2-hydroxyethyl)acetamide

| Conditions | Yield |
|---|---|
| In hexane for 0.5h; | A n/a B 96% |

-
-
56-23-5
tetrachloromethane

-
-
628-85-3
di-n-propylmercury

-
B
-
540-54-5
1-Chloropropane

-
C
-
67-66-3
chloroform

-
D
-
2440-40-6
n-propylmercury(II) chloride

| Conditions | Yield |
|---|---|
| In neat (no solvent) 150°C, 60 h; further products; | A <1 B 96% C >99 D 5% E 95% |

-
-
1768-31-6
pentachloroacetone

-
-
141-43-5
ethanolamine

-
A
-
67-66-3
chloroform

-
B
-
6419-44-9
2,2-dichloro-N-(2-hydroxyethyl)acetamide

| Conditions | Yield |
|---|---|
| In hexane for 0.5h; | A n/a B 95.4% |

-
-
116-16-5
1,1,1,3,3,3-hexachloro-propan-2-one

-
-
74-89-5
methylamine

-
A
-
67-66-3
chloroform

-
B
-
23170-77-6
N-methyltrichloroacetamide

| Conditions | Yield |
|---|---|
| In hexane Heating; | A n/a B 95.2% |

-
-
56-23-5
tetrachloromethane

-
-
80937-33-3
oxygen

-
-
1071-39-2
diisopropylmercury

-
A
-
74-98-6
propane

-
B
-
75-29-6
isopropyl chloride

-
C
-
67-66-3
chloroform

-
D
-
30615-19-1
isopropylmercury(II) chloride

| Conditions | Yield |
|---|---|
| In neat (no solvent) 20°C, 96 h; further products; | A 4% B 48% C 30% D 95% E 5% |

-
-
1768-31-6
pentachloroacetone

-
-
109-73-9
N-butylamine

-
A
-
67-66-3
chloroform

-
B
-
5345-74-4
2,2-dichloro-N-butylacetamide

| Conditions | Yield |
|---|---|
| In hexane | A n/a B 93.9% |

-
-
64697-39-8
1,1,1,3,3-pentachlorobutanone

-
-
141-43-5
ethanolamine

-
A
-
67-66-3
chloroform

-
B
-
83704-00-1
2,2-Dichloro-N-(2-hydroxy-ethyl)-propionamide

| Conditions | Yield |
|---|---|
| In hexane for 0.5h; | A n/a B 93.5% |

-
-
64697-39-8
1,1,1,3,3-pentachlorobutanone

-
-
107-11-9
1-amino-2-propene

-
A
-
67-66-3
chloroform

-
B
-
83703-99-5
N-Allyl-2,2-dichloro-propionamide

| Conditions | Yield |
|---|---|
| In hexane for 0.5h; | A n/a B 93.5% |

-
-
116-16-5
1,1,1,3,3,3-hexachloro-propan-2-one

-
-
64-17-5
ethanol

-
-
76-37-9
2,2,3,3-tetrafluoropropanol

-
A
-
67-66-3
chloroform

-
B
-
2062-06-8
2,2,3,3-Tetrafluor-propyl-propionat

| Conditions | Yield |
|---|---|
| Stage #1: 1,1,1,3,3,3-hexachloro-propan-2-one; ethanol With potassium fluoride at 30℃; for 1h; pressure tight reactor; Stage #2: 2,2,3,3-tetrafluoropropanol at 100℃; | A 93% B 74% |

-
-
1768-31-6
pentachloroacetone

-
-
110-58-7
1-pentanamine

-
A
-
67-66-3
chloroform

-
B
-
83703-97-3
N-Pentyl-2,2-dichloroacetamide

| Conditions | Yield |
|---|---|
| In hexane for 0.5h; | A n/a B 92% |

-
-
64697-39-8
1,1,1,3,3-pentachlorobutanone

-
-
110-58-7
1-pentanamine

-
A
-
67-66-3
chloroform

-
B
-
83703-98-4
2,2-Dichloro-N-pentyl-propionamide

| Conditions | Yield |
|---|---|
| In hexane for 0.5h; | A n/a B 92% |

-
-
55596-11-7
N-(2,2,2-trichloroethylidene)benzenesulfonamide

-
-
124-40-3
dimethyl amine

-
A
-
127-18-4
1,1,2,2-tetrachloroethylene

-
B
-
67-66-3
chloroform

-
C
-
13707-43-2
N,N-dimethyl-N′-(phenylsulfonyl)formimidamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 24h; | A n/a B n/a C 92% |

-
-
112-30-1
1-Decanol

-
-
1768-31-6
pentachloroacetone

-
A
-
67-66-3
chloroform

-
B
-
83005-00-9
decyl 2,2-dichloroacetate

| Conditions | Yield |
|---|---|
| With triethylamine Heating; | A n/a B 91.4% |

-
-
56-23-5
tetrachloromethane

-
-
1071-39-2
diisopropylmercury

-
B
-
75-29-6
isopropyl chloride

-
C
-
67-66-3
chloroform

-
D
-
30615-19-1
isopropylmercury(II) chloride

| Conditions | Yield |
|---|---|
| In neat (no solvent) 130°C, 10 h; further products; | A 3% B 51% C 50% D 91% E 3% |


| Conditions | Yield |
|---|---|
| With potassium hydroxide; tetrabutylammomium bromide; 1,3,5-trimethyl-benzene In dichloromethane at 40℃; for 6h; | 100% |
| With PEG-supported tetrakis ammonium salt In dichloromethane at 25℃; for 0.5h; | 98% |
| With tetrabutylammomium bromide; sodium hydroxide In dichloromethane at 20℃; for 0.0833333h; | 96% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; Sucrose-ethyleneoxide adducts In chloroform at 20℃; for 2h; Product distribution; further catalysts: PEG, DB18K6; further objects of study: phase-transfer catalysis;; | 100% |
| With sodium hydroxide; Sucrose-ethyleneoxide adducts In chloroform at 20℃; for 2h; Product distribution; further catalysts: PEG, DB18K6; | 100% |
| With potassium hydroxide; 18-crown-6 ether In dichloromethane at 40℃; for 6h; | 98% |

| Conditions | Yield |
|---|---|
| With Grubbs catalyst first generation at 65℃; for 2h; | 100% |
| Grubbs catalyst first generation at 65 - 80℃; Kharasch addition; | 99% |
| With N2[(RuCl2)2(1,3,5-iPr3C6H3)(tricyclohexylphosphine]2 In toluene at 40℃; for 48h; | 93% |
-
-
67-66-3
chloroform

-
-
83466-54-0, 93602-74-5, 79073-99-7
Bis(2,4,6-tri-tert-butylphenyl)diphosphen

-
-
111888-01-8, 126976-48-5
1,2-bis(2,4,6-tri-tert-butylphenyl)3,3-dichloro diphosphirane

| Conditions | Yield |
|---|---|
| With potassium hydroxide In hexane at 15℃; for 2h; sonication; | 100% |
| With potassium tert-butylate |
-
-
67-66-3
chloroform

-
-
132067-80-2
3,3-Diethyl-6,7-dimethoxy-1-phenyl-3,4-dihydro-isoquinoline

-
-
132067-87-9
6,7-Dimethoxy-8bphenyl-1,1-dichloro-3,3-diethyl-1,3,4,8b-tetrahydroazirino<2,1-a>isoquinoline

| Conditions | Yield |
|---|---|
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride In hexane 1) 3 h, 12 deg C 2) 1 h, 20 deg C; | 100% |

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 20℃; for 0.5h; | 100% |
-
-
67-66-3
chloroform

-
-
156-43-4
4-Ethoxyaniline

-
-
67-64-1
acetone

-
-
74262-33-2
α-p-phenetidino-isobutyric acid p-phenetidide

| Conditions | Yield |
|---|---|
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride In dichloromethane at 5℃; | 100% |

-
-
67-66-3
chloroform

| Conditions | Yield |
|---|---|
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride 0 deg C, 30 min; RT, 2.5 h; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide for 24h; | 100% |
| With ammonium hydroxide at 20℃; for 24h; | 74.9% |
-
-
67-66-3
chloroform

| Conditions | Yield |
|---|---|
| With potassium hydroxide; N-benzyl-N,N,N-triethylammonium chloride at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetrabutylammomium bromide In water for 16h; | 100% |

-
-
67-66-3
chloroform

| Conditions | Yield |
|---|---|
| palladium In methanol; N,N-dimethyl-formamide | 100% |
| palladium In methanol; N,N-dimethyl-formamide | 100% |

-
-
67-66-3
chloroform

| Conditions | Yield |
|---|---|
| palladium In methanol; N,N-dimethyl-formamide | 100% |

-
-
67-66-3
chloroform

| Conditions | Yield |
|---|---|
| In ethanol; chloroform byproducts: CH2Cl2, CH3CHO; (Ar); using Schlenk techniques; dissolving of ((Ph4C4CO)2H)Ru2(CO)4(H) in degassed CHCl3 and C2H5OH; closing of flask, stirring at 90°C for 7 h; removal of solvent under high vac., elem .anal.; | 100% |
-
-
67-66-3
chloroform

| Conditions | Yield |
|---|---|
| In chloroform Zr((CH3)2C6H2(O)CH2)3NH) dissolved in dry CHCl3 in air; recrystd.; elem. anal.; detd. by XRD; | 100% |
-
-
67-66-3
chloroform

-
-
866488-88-2
bis(η2-ethene)[η5-(8-quinolyl)cyclopentadienyl]iridium(I)

-
-
1184999-49-2
[η5-(8-quinolyl)cyclopentadienyl]dichloridoiridium(III)

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); under Ar atm.soln. Ir complex in CHCl3 was irradiated for 3 days with light 150-W Hg high-pressure lamp; solvent was evapd.; | 100% |
-
-
67-66-3
chloroform


| Conditions | Yield |
|---|---|
| In chloroform 80°C, 48 h; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: AgCl; under N2 or Ar using Schlenk app.; CH2Cl2 added to solid mixt. of (Pd(C6H4NNC6H5)Cl)2 (0.20 mol) and 1,4,7-trithiacyclononane (0.40 mol); stirred for 18 h; filtered; evapd. to dryness; extd. (MeOH); ext. filtered and evapd. to dryness; crystd. (CHCl3 and hexane) at room temp. for 3 d; | 100% |

-
-
39929-21-0
(tetrahydrothiophene)gold(I) chloride

-
-
67-66-3
chloroform

| Conditions | Yield |
|---|---|
| In dichloromethane (Schlenk, N2) to stirred soln. of phosphane in CH2Cl2 was added to a soln. of Au-complex in CH2Cl2, 30 min; evapd. to dryness, column chromy. (silica gel, CH2Cl2-CH3OH, 96:4, v/v);elem. anal.; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: 2,4,6-trimethylaniline With 15-crown-5; sodium hydride In benzene at 40 - 50℃; for 0.25h; Stage #2: chloroform In benzene at 40℃; for 1h; | 100% |
| With benzyltriethylammonium chloride; sodium hydroxide In dichloromethane; water for 22h; | 53% |

| Conditions | Yield |
|---|---|
| With N-benzyl-N,N,N-triethylammonium chloride; sodium hydroxide In chloroform; water at 40 - 45℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With N-benzyl-N,N,N-triethylammonium chloride; sodium hydroxide In water at 40 - 45℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With N-benzyl-N,N,N-triethylammonium chloride; sodium hydroxide In water at 40 - 45℃; | 100% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View



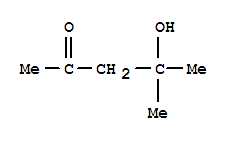


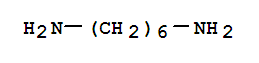








 Xn,
Xn,  F,
F,  T
T