-
Name
(2S,3S)(-)-Dihydroxybutane-1,4-dioic acid diethyl ester
- EINECS 237-458-8
- CAS No. 13811-71-7
- Article Data16
- CAS DataBase
- Density 1.262 g/cm3
- Solubility insoluble in water
- Melting Point 17 °C
- Formula C8H14O6
- Boiling Point 280 °C at 760 mmHg
- Molecular Weight 206.196
- Flash Point 93.3 °C
- Transport Information
- Appearance Colorless to light yellow liquid
- Safety 24/25-36-26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi Xi
Xi
- Synonyms Butanedioicacid, 2,3-dihydroxy-, diethyl ester, [S-(R*,R*)]- (9CI);Tartaric acid, diethylester, (-)- (8CI);(2S,3S)-Diethyl tartrate;(S,S)-Diethyl tartrate;D-(-)-Diethyl tartrate;D-Tartaric acid diethyl ester;Diethyl (-)-D-tartrate;Diethyl (2S,3S)-(-)-tartrate;Diethyl(S,S)-(-)-tartrate;
- PSA 93.06000
- LogP -1.16560
Synthetic route

| Conditions | Yield |
|---|---|
| With thionyl chloride at 20℃; | 100% |
| With thionyl chloride at 20℃; | 100% |
| With hydrogenchloride In chloroform; water for 60h; Catalytic behavior; Reflux; enantioselective reaction; | 99.6% |

-
-
13811-71-7
diethyl (2S,3S)-tartrate

| Conditions | Yield |
|---|---|
| With indium; water; ammonium chloride In methanol for 1h; Heating; | 89% |

-
-
13811-71-7
diethyl (2S,3S)-tartrate

| Conditions | Yield |
|---|---|
| With indium; water; ammonium chloride In methanol for 2h; Heating; | 85% |
-
-
136087-36-0
diethyl (4R,5R)-1,3,2-dioxathiolane-4,5-dicarboxylate 2-oxide

-
-
13811-71-7
diethyl (2S,3S)-tartrate

| Conditions | Yield |
|---|---|
| With sodium azide In ethanol; water at 20℃; for 14h; |
-
-
136087-36-0
diethyl (4R,5R)-1,3,2-dioxathiolane-4,5-dicarboxylate 2-oxide

-
A
-
13811-71-7
diethyl (2S,3S)-tartrate

-
B
-
101924-53-2
diethyl (2S,3R)-2-azido-3-hydroxysuccinate

| Conditions | Yield |
|---|---|
| With sodium azide In water; N,N-dimethyl-formamide at 20℃; for 14h; |

-
-
57968-71-5
(+/-)-diethyl tartrate

-
A
-
13811-71-7
diethyl (2S,3S)-tartrate

-
B
-
87-91-2
diethyl (2R,3R)-tartrate

| Conditions | Yield |
|---|---|
| With (S)-aminopropyl alcohol(at)silica 1 In hexane; isopropyl alcohol at 20℃; for 1h; Purification / work up; Resolution of racemate; Inert atmosphere; | A n/a B n/a |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
35506-91-3, 133522-25-5, 136087-36-0
diethyl (4S,5S)-1,3,2-dioxathiolane-4,5-dicarboxylate 2-oxide

| Conditions | Yield |
|---|---|
| With thionyl chloride In tetrachloromethane Heating; | 100% |
| With thionyl chloride; triethylamine In dichloromethane for 3h; | 100% |
| With thionyl chloride; N,N-dimethyl-formamide In dichloromethane at 0 - 50℃; for 2.5h; | 95% |
-
-
109-87-5
Dimethoxymethane

-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
159593-22-3
diethyl (2S,3S)-2,3-di-O-methoxymethyl-tartrate

| Conditions | Yield |
|---|---|
| With phosphorus pentoxide In dichloromethane | 100% |
| With phosphorus pentoxide In dichloromethane for 3h; | 100% |
| With phosphorus pentoxide In chloroform for 2h; Alkylation; | 84% |
| With phosphorus pentoxide In dichloromethane | |
| With phosphorus pentoxide In dichloromethane at 0℃; for 10h; Inert atmosphere; |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
1034-39-5
dibromotriphenylphosphorane

| Conditions | Yield |
|---|---|
| With triethylamine In toluene Cyclization; | 100% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
77-78-1
dimethyl sulfate

-
-
21066-69-3
(-)-(2S,3S)-O,O-Dimethylweinsaeure-diethylester

| Conditions | Yield |
|---|---|
| With sodium hydride In diethyl ether at 0 - 20℃; Inert atmosphere; | 100% |
| With sodium hydride In mineral oil at 0 - 20℃; for 12h; | 82% |

| Conditions | Yield |
|---|---|
| Stage #1: titanium(IV) isopropylate; omeprazole sodium; diethyl (2S,3S)-tartrate With triethylamine In propan-1-ol at 40℃; for 0.166667h; Stage #2: (S)-Mandelic acid In propan-1-ol at 40℃; for 3h; Product distribution / selectivity; | 99.8% |

-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
80640-15-9
diethyl (2S,3R)-(+)-erythro-2-hydroxy-3-bromosuccinate

| Conditions | Yield |
|---|---|
| With hydrogen bromide; acetic acid at 0 - 25℃; for 13h; Inert atmosphere; | 99.1% |
| With hydrogen bromide; acetic acid at 0 - 20℃; for 12h; | 99.1% |
| With hydrogen bromide; acetic acid In benzene Heating; | 90% |
| Stage #1: diethyl (2S,3S)-tartrate With hydrogen bromide; acetic acid at 20℃; for 16h; Stage #2: With ethanol; acetyl chloride for 5h; Heating; | |
| Multi-step reaction with 2 steps 1: hydrogen bromide / 6.25 h / 0 - 20 °C 2: hydrogen bromide / acetic acid; ethanol / 10 h / Reflux View Scheme |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
2186-92-7
p-Anisaldehyde dimethyl acetal

-
-
135972-13-3
(2S,3S)-2,3-O-(4-methoxybenzylidene)-diethyl tartrate

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In N,N-dimethyl-formamide at 50℃; under 10 Torr; for 5h; | 99% |
| With toluene-4-sulfonic acid In N,N-dimethyl-formamide at 50℃; under 18 Torr; for 5h; | 99% |
| With toluene-4-sulfonic acid In N,N-dimethyl-formamide | 96% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
67-64-1
acetone

-
-
73346-73-3
(4S,5S)-diethyl 2,2-dimethyl-1,3-dioxolane-4,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With orthoformic acid triethyl ester | 99% |
| With iodine at 20℃; for 12h; | 77% |
| In orthoformic acid triethyl ester at 20℃; Reflux; | 72.6 g |

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate at 0 - 20℃; solid phase reaction; | 99% |

-
-
107-25-5
methoxyethene

-
-
13811-71-7
diethyl (2S,3S)-tartrate

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane for 48h; 0 deg C then room temp.; | 98% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
77-76-9
2,2-dimethoxy-propane

-
-
59779-75-8
diethyl (4R,5R)-2,2-dimethyl-1,3-dioxolane-4,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid; orthoformic acid triethyl ester at 45℃; for 9h; Temperature; Reagent/catalyst; Reflux; | 98% |
| With camphor-10-sulfonic acid | 68.5% |
| With toluene-4-sulfonic acid; triethylamine In chloroform for 17h; Reflux; | 48.8% |
| With toluene-4-sulfonic acid In chloroform for 18h; Reflux; | 42.8% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
100-39-0
benzyl bromide

-
-
113321-58-7
(2S,3S)-diethyl 2,3-bis(phenylmethoxy)succinate

| Conditions | Yield |
|---|---|
| With potassium iodide; silver(l) oxide In dichloromethane for 1h; Heating; | 97% |
| With potassium iodide; silver(l) oxide In dichloromethane for 1h; Reflux; Inert atmosphere; | 97% |
| With thallium (I) ethoxide In acetonitrile 1.) 55 deg C, 48 h, 2.) room temp., 12 h; | 88% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
77-76-9
2,2-dimethoxy-propane

-
-
73346-73-3
(4S,5S)-diethyl 2,2-dimethyl-1,3-dioxolane-4,5-dicarboxylate

| Conditions | Yield |
|---|---|
| Acidic conditions; | 97% |
| With toluene-4-sulfonic acid In dichloromethane at 40℃; for 17h; | 90% |
| With toluene-4-sulfonic acid In benzene for 8h; Reflux; | 79.2% |
-
-
25636-49-1
3,3-dimethoxypentane

-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
1226768-08-6
diethyl (4S,5S)-2,2-diethyl-1,3-dioxolane-4,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene for 3h; Inert atmosphere; Reflux; | 96% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
122-51-0
orthoformic acid triethyl ester

-
-
95588-28-6
D-(S,S)-2-ethoxy-1,3-dioxolane-4,5-dicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| With camphor-10-sulfonic acid In toluene at 95℃; | 95% |
| With acetic acid In toluene Heating; | 94% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
100-52-7
benzaldehyde

-
-
141042-56-0
diethyl(4S,5S)-2-phenyl-1,3-dioxolan-4,5-dicarboxylate

| Conditions | Yield |
|---|---|
| Stage #1: benzaldehyde With camphor-10-sulfonic acid; toluene-4-sulfonic acid; orthoformic acid triethyl ester In toluene for 0.75h; Heating / reflux; Stage #2: diethyl (2S,3S)-tartrate In toluene Heating / reflux; | 94.1% |
| With toluene-4-sulfonic acid; orthoformic acid triethyl ester | 84% |
| With toluene-4-sulfonic acid In cyclohexane for 4h; Reflux; |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
100-46-9
benzylamine

-
-
108321-43-3
N,N'-dibenzyl-(S,S)-tartaric acid diamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol for 8h; Heating; | 94% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
74213-59-5
diethyl (2S,3R)-erythro-2-acetoxy-3-bromosuccinate

| Conditions | Yield |
|---|---|
| With hydrogen bromide In acetic acid for 4h; Ambient temperature; | 93% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
100-39-0
benzyl bromide

-
-
119770-87-5
diethyl (2S,3S)-2-(benzyloxy)-3-hydroxybutanedioate

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; di(n-butyl)tin oxide; N-ethyl-N,N-diisopropylamine In neat (no solvent) at 70℃; for 1h; regioselective reaction; | 93% |
| With silver(l) oxide In dichloromethane at 20℃; for 2h; | 17.9 g |

-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
165184-98-5
(E)-α-hexylcinnamaldehyde

-
-
122-51-0
orthoformic acid triethyl ester

-
-
144407-12-5
(E)-(4R,5R)-2-(1-Hexyl-2-phenyl-1-ethenyl)-4,5-dicarboethoxy-1,3-dioxolane

| Conditions | Yield |
|---|---|
| In toluene-4-sulfonic acid; toluene | 91% |

-
-
123-75-1
pyrrolidine

-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
102197-56-8
1,1'-[(2R,3R)-2,3-dihydroxy-1,4-dioxo-1,4-butanediyl]bis-pyrrolidine

| Conditions | Yield |
|---|---|
| In ethanol for 12h; Reflux; | 91% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
503-38-8
trichloromethyl chloroformate

| Conditions | Yield |
|---|---|
| With pyrographite In tetrahydrofuran at 50℃; for 120h; | 90% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
136087-36-0
diethyl (4R,5R)-1,3,2-dioxathiolane-4,5-dicarboxylate 2-oxide

| Conditions | Yield |
|---|---|
| With pyridine; thionyl chloride In dichloromethane at 0℃; | 90% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
67-64-1
acetone

-
-
59779-75-8
diethyl (4R,5R)-2,2-dimethyl-1,3-dioxolane-4,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With hydrogenchloride; orthoformic acid triethyl ester In N,N-dimethyl-formamide for 72h; Ambient temperature; | 89.5% |
| With toluene-4-sulfonic acid In toluene for 15h; Inert atmosphere; Reflux; | 18% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
120-92-3
cyclopentanone

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene for 36h; Heating; | 88% |
| With 10-camphorsulfonic acid In toluene at 150℃; for 24h; |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
79-04-9
chloroacetyl chloride

-
-
226710-54-9
(2S,3S)-2,3-Bis-(2-chloro-acetoxy)-succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With pyridine In chloroform for 5h; Ambient temperature; | 87.3% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
85785-78-0
4-(Phenylsulfonyl)-2-butanon-dimethylacetal

-
-
85785-79-1
(4S,5S)-2-(2-Benzenesulfonyl-ethyl)-2-methyl-[1,3]dioxolane-4,5-dicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene at 80 - 90℃; for 20h; | 87% |
-
-
13811-71-7
diethyl (2S,3S)-tartrate

-
-
100-52-7
benzaldehyde

-
-
21154-16-5, 35572-31-7, 89015-57-6, 141042-56-0
diethyl-2,3-O-benzylidene-D-tartrate

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In cyclohexane for 16h; Catalytic behavior; Reflux; enantioselective reaction; | 87% |
(-)-Diethyl D-tartrate Chemical Properties
Synonyms : (-)-Diethyl-2,3-dihydroxysuccinate; (2S,3S)(-)-Dihydroxybutane-1,4-dioic acid diethyl ester
Molecular Structure :
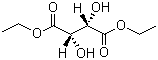
Molecular Formula : C8H14O6
Molecular Weight : 206.19
CAS Registry Number : 13811-71-7
EINECS : 237-458-8
Melting point : 17 oC
Boiling point : 280 oC
Flash point : 93 oC
Density : 1.205
Refractive index : 1.445-1.447
Water solubility : insoluble
(-)-Diethyl D-tartrate Safety Profile
Related Products
- (-)-Diethyl D-tartrate
- 138117-50-7
- 138124-32-0
- 138-12-5
- 138126-04-2
- 13813-19-9
- 13813-22-4
- 13813-23-5
- 13813-24-6
- 13813-40-6
- 13813-41-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View