-
Name
(S)-3-N-Boc-aminopiperidine
- EINECS 626-980-5
- CAS No. 216854-23-8
- Article Data3
- CAS DataBase
- Density 1.02 g/cm3
- Solubility
- Melting Point 122-127 °C
- Formula C10H20N2O2
- Boiling Point 304.8 °C at 760 mmHg
- Molecular Weight 200.281
- Flash Point 138.2 °C
- Transport Information
- Appearance
- Safety 26-39
- Risk Codes 37/38-41
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Carbamicacid, (3S)-3-piperidinyl-, 1,1-dimethylethyl ester (9CI);(S)-(-)-3-(tert-Butoxycarbonylamino)piperidine;(S)-3-tert-Butoxycarbonylaminopiperidine;(S)-tert-ButylN-[piperidin-3-yl]carbamate;tert-Butyl ((3S)-piperidin-3-yl)carbamate;
- PSA 50.36000
- LogP 1.98280
Synthetic route
-
-
92235-39-7
3(S)-<(butoxycarbonyl)amino>-2-oxopiperidine

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| With borane-THF In tetrahydrofuran at 0 - 25℃; for 6.75h; | 30% |
-
-
607376-86-3
(S)-2-(tert-butoxycarbonylamino)pentane-1,5-diyl dimethanesulfonate

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| With ammonium hydroxide In acetonitrile for 48h; Ambient temperature; Yield given; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: H2SO4 / 2 h / Heating 2: Et3N / dioxane; H2O / 2 h / 0 °C 3: NaBH4, CaCl2 / ethanol / 2 h / 0 °C 4: Et3N / CH2Cl2 / 1 h / -10 - 0 °C 5: aq. NH4OH / acetonitrile / 48 h / Ambient temperature View Scheme |
-
-
6525-53-7, 16422-27-8, 40149-68-6
L-glutamic acid dimethyl ester

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: Et3N / dioxane; H2O / 2 h / 0 °C 2: NaBH4, CaCl2 / ethanol / 2 h / 0 °C 3: Et3N / CH2Cl2 / 1 h / -10 - 0 °C 4: aq. NH4OH / acetonitrile / 48 h / Ambient temperature View Scheme |
-
-
59279-60-6, 130622-05-8
dimethyl (2S)-2-[(tert-butoxycarbonyl)amino]pentanedioate

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: NaBH4, CaCl2 / ethanol / 2 h / 0 °C 2: Et3N / CH2Cl2 / 1 h / -10 - 0 °C 3: aq. NH4OH / acetonitrile / 48 h / Ambient temperature View Scheme |
-
-
162955-48-8
[(1S)-4-hydroxy-1-(hydroxymethyl)butyl]carbamic acid tert-butyl ester

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Et3N / CH2Cl2 / 1 h / -10 - 0 °C 2: aq. NH4OH / acetonitrile / 48 h / Ambient temperature View Scheme |
-
-
1188914-98-8
1-[4-chloro-6-(4-morpholinyl)-1,3,5-triazin-2-yl]-2-(difluoromethyl)-4-methoxy-1H-benzimidazole

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

-
-
1188915-59-4
tert-butyl (R)-1-[4-[2-(difluoromethyl)-4-methoxy-1H-benzimidazol-1-yl]-6-(4-morpholinyl)-1,3,5-triazin-2-yl]piperidin-3-ylcarbamate

| Conditions | Yield |
|---|---|
| at 20℃; | 100% |
-
-
1527525-60-5
1-[5-(difluoromethyl)-6-fluoro-2-pyridyl]-6-(6-methylpyrazin-2-yl)pyrazolo[4,3-c]pyridine

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

-
-
1527525-62-7
tert-butyl N-[(3S)-1-[3-(difluoromethyl)-6-[6-(6-methylpyrazin-2-yl)pyrazolo[4,3-c]pyridin-1-yl]-2-pyridyl]-3-piperidyl]carbamate

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine In 1-methyl-pyrrolidin-2-one at 100℃; Sealed tube; | 100% |
-
-
23680-84-4
2-chloro-6,7-dimethoxyquinazolin-4-amine

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| In i-Amyl alcohol at 130℃; under 760.051 Torr; for 0.833333h; Microwave irradiation; | 100% |
-
-
1188914-98-8
1-[4-chloro-6-(4-morpholinyl)-1,3,5-triazin-2-yl]-2-(difluoromethyl)-4-methoxy-1H-benzimidazole

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

-
-
1188915-61-8
tert-butyl (S)-1-[4-[2-(difluoromethyl)-4-methoxy-1H-benzimidazol-1-yl]-6-(4-morpholinyl)-1,3,5-triazin-2-yl]piperidin-3-ylcarbamate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; | 100% |
-
-
122-78-1
phenylacetaldehyde

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

-
-
1361322-88-4
(S)-tert-butyl 1-phenethylpiperidin-3-ylcarbamate

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; acetic acid In dichloromethane at 20℃; for 4.25h; | 99% |
-
-
501-53-1
benzyl chloroformate

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

-
-
876379-22-5
benzyl (3S)-3-[(2-methylpropan-2-yl)oxycarbonylamino]piperidine-1-carboxylate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; | 99% |
| With triethylamine In dichloromethane at 0℃; for 1h; | 95% |
| With triethylamine In ethyl acetate at 0℃; for 1h; | 95% |
| With triethylamine In dichloromethane at 0 - 20℃; Product distribution / selectivity; | 89% |
| With triethylamine In tetrahydrofuran at 20℃; for 0.5h; |
-
-
776-74-9
Bromodiphenylmethane

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 60℃; for 14h; | 94% |
-
-
35432-36-1
2-methylpropane-1-sulfonyl chloride

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

-
-
1270496-69-9
[(S)-1-(2-methyl-propane-1-sulfonyl)-piperidin-3-yl]-carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; | 93% |
-
-
110651-92-8
7-chloro-6-nitrothieno[3,2-b]pyridine

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

-
-
1620011-13-3
tert-butyl [(3S)-1-(6-nitrothieno[3,2-b]pyridin-7-yl)piperidin-3-yl]carbamate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In butan-1-ol at 110℃; for 12h; | 93% |

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In 1,4-dioxane at 90℃; for 1h; | 92% |
-
-
1200130-66-0
4-chloro-9-(2-trimethylsilanyl-ethoxymethyl)-9H-dipyrido[2,3-b;4',3'-d]pyrrole-6-carbonitrile

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

-
-
1312753-95-9
(S)-tert-butyl 1-(6-cyano-9-((2-(trimethylsilyl)ethoxy)methyl)-9H-dipyrido[2,3-b;4',3'-d]pyrrol-4-yl)piperidin-3-ylcarbamate

| Conditions | Yield |
|---|---|
| In N,N-dimethyl acetamide at 100℃; for 2h; | 90% |
-
-
870703-61-0
di-tert-butyl (6-bromo-2-pyridinyl)imidodicarbonate

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| Stage #1: di-tert-butyl (6-bromo-2-pyridinyl)imidodicarbonate; tert-butyl (S)-piperidin-3-yl-carbamate With tris-(dibenzylideneacetone)dipalladium(0); caesium carbonate; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In N,N-dimethyl-formamide at 95℃; for 7h; Inert atmosphere; Stage #2: With hydrogenchloride In 1,4-dioxane at 20℃; for 7h; | 90% |
-
-
50-00-0
formaldehyd

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

-
-
902152-77-6
(S)-tert-butyl (1-methylpiperidin-3-yl)carbamate

| Conditions | Yield |
|---|---|
| With formic acid In 2-methyltetrahydrofuran; water at 80℃; for 0.75h; | 90% |

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In ethanol at 100℃; for 15h; | 90% |
-
-
1620012-10-3
5-iodo-6-nitro-1,2,3,4-tetrahydro-1,8-naphthyridine

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

-
-
1620012-11-4
tert-butyl [(3S)-1-(3-nitro-5,6,7,8-tetrahydro-1,8-naphthyridin-4-yl)piperidin-3-yl]carbamate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In butan-1-ol at 140℃; for 14h; | 89% |
-
-
41019-45-8
5-(4-chlorophenyl)-2-furoic acid

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate In N,N-dimethyl-formamide at 20℃; for 1h; | 89% |

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In 1,4-dioxane at 90℃; for 0.5h; | 88% |
| With N-ethyl-N,N-diisopropylamine In 1,4-dioxane at 90℃; for 0.5h; | 88% |

-
-
216854-23-8
tert-butyl (S)-piperidin-3-yl-carbamate

| Conditions | Yield |
|---|---|
| With dmap; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In N,N-dimethyl-formamide at 40℃; for 8h; | 88% |
(S)-3-N-Boc-aminopiperidine Chemical Properties
IUPAC Name: tert-butyl N-[(3S)-piperidin-3-yl]carbamate
Synonyms: (S)-TERT-BUTYL-PIPERIDIN-3-YLCARBAMATE;Carbamic acid, (3S)-3-piperidinyl-, 1,1-dimethylethyl ester (9CI);(S)-PIPERIDIN-3-YL-CARBAMIC ACID TERT-BUTYL ESTER;;(S)-3-N-BOC-AMINOPIPERIDINE;(S)-3-BOC-AMINOPIPERIDINE;CARBAMIC ACID, (3S)-3-PIPERIDINYL-, 1,1-DIMETHYLETHYL ESTER;(S)-(-)-3-TERT-BUTOXYCARBONYLAMINOPIPERIDINE;(S)-3-(TERT-BUTOXYCARBONYLAMINO)PIPERIDINE
The Molecular formula of (S)-3-N-Boc-aminopiperidine(216854-23-8): C10H20N2O2
The Molecular Weight of (S)-3-N-Boc-aminopiperidine(216854-23-8): 200.28
Molecular Structure : 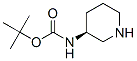
Melting point: 122-127°C
alpha: -3 o (c=1, DMF)
(S)-3-N-Boc-aminopiperidine Safety Profile
The Hazard Codes of (S)-3-N-Boc-aminopiperidine(216854-23-8):  Xi
Xi
The Risk Statements information of (S)-3-N-Boc-aminopiperidine(216854-23-8):
41: Risk of serious damage to eyes
37/38: Irritating to respiratory system and skin
The Safety Statements information of (S)-3-N-Boc-aminopiperidine(216854-23-8):
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
39: Wear eye/face protection
WGK Germany: 3
Related Products
- (S)-3-N-Boc-aminopiperidine
- 216854-24-9
- 216854-53-4
- 216854-55-6
- 21685-47-2
- 2168-93-6
- 2168-96-9
- 21691-41-8
- 21691-44-1
- 2169-44-0
- 216955-61-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View