-
Name
1,4-Dithiane
- EINECS 208-007-2
- CAS No. 505-29-3
- Article Data73
- CAS DataBase
- Density 1.14 g/cm3
- Solubility slightly soluble in water, soluble in hot ethanol and oil
- Melting Point 107-113 °C
- Formula C4H8S2
- Boiling Point 200.5 °C at 760 mmHg
- Molecular Weight 120.24
- Flash Point 82.1 °C
- Transport Information
- Appearance white solid with an extremely unpleasant smell
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms p-Dithiane(7CI,8CI);1,4-Dithiacyclohexane;1,4-Dithiin, tetrahydro-;NSC 24178;p-Dithiane;Diethylene disulfide;p-Dithiin, tetrahydro-;CCRIS 3240;HSDB 7426;
- PSA 50.60000
- LogP 1.46640
Synthetic route

| Conditions | Yield |
|---|---|
| With 1-pentyl-3-methylimidazolium bromide at 60℃; for 5.5h; | 92% |
| With sodium ethanolate In ethanol for 2.5h; Cyclization; Heating; | 64% |

| Conditions | Yield |
|---|---|
| With 25percent soude; tetrabutylammomium bromide In benzene at 80℃; for 4h; Product distribution; other concentration of catalyst and soude, time; | 87.5% |
| With 1-pentyl-3-methylimidazolium bromide at 60℃; for 0.133333h; Irradiation; microwave; | 81% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene at 150℃; for 0.333333h; microwave irradiation; | 81% |
-
-
25423-55-6
3,6-dithiaoctan-1,8-dithiol

-
-
3563-36-8
1,2-bis(2-chloroethylthio)ethane

-
A
-
505-29-3
1,4-Dithiane

-
B
-
296-41-3
1,4,7,10,13,16-hexathiacyclooctadecan

-
C
-
25423-56-7
1,4,7,10-tetrathiacyclododecane

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 50℃; | A n/a B 70% C 5% |

-
-
16892-64-1
2,2,4,4,6,6-hexamethyl-1,3,5,2,4,6-trithiatristanninane

-
-
106-93-4
ethylene dibromide

-
A
-
505-29-3
1,4-Dithiane

-
C
-
2767-47-7
dimethyltin dibromide

| Conditions | Yield |
|---|---|
| In further solvent(s) soln. of (Me2SnS)3 in BrCH2CH2Br was heated at 140°C for 35 h; excess C2H4Br2 was removed in vac. at room temp., products were sepd. by sublimation at 70°C; dithiane was identified by chromy., elem. anal. for 2Me2SnBr2*S(CH2CH2)2S; | A n/a B 58% C 18% |

-
-
113322-95-5
{(C5H5)Ru(P(C6H5)3)2(SC2H4)}(1+)*CF3SO3(1-)*0.5CH2Cl2={(C5H5)Ru(P(C6H5)3)2(SC2H4)}(OSO2CF3)*0.5CH2Cl2

-
-
2001-45-8
tetraphenyl phosphonium chloride

-
A
-
505-29-3
1,4-Dithiane

-
B
-
32993-05-8
chloro(cyclopentadienyl)bis(triphenylphosphine)ruthenium (II)

| Conditions | Yield |
|---|---|
| In dichloromethane-d2 byproducts: (C2H4S)n; PPh4Cl added to CD2Cl2-soln. of Ru-thiiran complex at 0°C; decompn. by warming to room temp.; | A n/a B 50% |

-
-
50-00-0
formaldehyd

-
-
540-63-6
ethane-1,2-dithiol

-
A
-
4829-04-3
1,3-dithiolane

-
B
-
505-29-3
1,4-Dithiane

-
C
-
1449678-80-1
3-methylideneamino-1,5,3-dithiazepane

-
D
-
154729-78-9
1,3,6,8-tetrathiecane

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydrazine hydrate In water at 20℃; pH=0.45 - 0.5; | A 30% B 9% C 49% D 12% |

-
-
2040-67-7, 89124-15-2
trans-2,3-dichloro-1,4-dithiane

-
A
-
505-29-3
1,4-Dithiane

-
B
-
6784-47-0
2,2'-bi-1,3-dithiolanyl

-
C
-
23230-01-5
2,3-dihydro-1,4-dithiin

-
D
-
106691-43-4
2,3-dihydro-5,6-dichloro-1,4-dithiin

| Conditions | Yield |
|---|---|
| With disodium 1,2-ethanedithiolate In ethanol for 12h; | A 2% B n/a C 15% D 6% |


| Conditions | Yield |
|---|---|
| With aluminum oxide; sulfide ion In toluene at 140℃; for 20h; | A 1.5% B 1.8% |

| Conditions | Yield |
|---|---|
| With aluminum oxide; hydrogen sulfide at 220℃; |

| Conditions | Yield |
|---|---|
| With potassium pyrosulfate at 160 - 210℃; | |
| With aluminium silicate at 240℃; | |
| With cation exchanger at 180℃; | |
| With toluene-4-sulfonic acid; acetaldehyde; benzene Unter Entfernen des entstehenden Wassers und Erhitzen des Reaktionsprodukts auf 200grad; | |
| at 180℃; in Gegenwart eines sauren Kationen-Austauschers; |

| Conditions | Yield |
|---|---|
| With aluminum oxide at 225℃; | |
| With hydrogen bromide; phenol |

| Conditions | Yield |
|---|---|
| at 125 - 200℃; |
-
-
856365-85-0
1-(2-chloro-ethylsulfanyl)-2-propylsulfanyl-ethane

-
A
-
505-29-3
1,4-Dithiane

-
B
-
540-54-5
1-Chloropropane

| Conditions | Yield |
|---|---|
| beim Erhitzen; |
-
-
159859-35-5
1-butylsulfanyl-2-(2-chloro-ethylsulfanyl)-ethane

-
A
-
505-29-3
1,4-Dithiane

-
B
-
109-69-3
n-Butyl chloride

| Conditions | Yield |
|---|---|
| beim Erhitzen; |
-
-
14440-77-8
3,6,9-trithia-1,11-undecanediol

-
-
505-29-3
1,4-Dithiane

| Conditions | Yield |
|---|---|
| With hydrogen bromide; phenol |
-
-
64-17-5
ethanol

-
-
505-60-2
bis (2-chloroethyl) sulphide

-
-
1001-58-7
3-mercaptopropionitrile

-
A
-
505-29-3
1,4-Dithiane

-
B
-
111-97-7
3,3'-thiobis-propanenitrile

| Conditions | Yield |
|---|---|
| Reagiert des Natriumsalzes; |


| Conditions | Yield |
|---|---|
| With sulfur dichloride at 0℃; | |
| With sodium sulfide | |
| With phenol at 180℃; im Rohr; | |
| With sulfur at 200℃; |

| Conditions | Yield |
|---|---|
| at 180℃; | |
| at 180℃; Gleichgewicht; |
-
-
505-60-2
bis (2-chloroethyl) sulphide

-
A
-
505-29-3
1,4-Dithiane

-
B
-
74-85-1
ethene

-
C
-
107-06-2
1,2-dichloro-ethane

| Conditions | Yield |
|---|---|
| at 220℃; Prod.5:HCl; | |
| at 180℃; Prod.5:HCl; |

-
-
98026-19-8
2-(2-mercapto-ethylsulfanyl)-ethanol

-
-
505-29-3
1,4-Dithiane

| Conditions | Yield |
|---|---|
| With hydrogenchloride Erhitzen des Reaktionsprodukts in Phenol bis auf 180grad; |
-
-
125787-98-6
1-Chloro-2-<2-(methylthio)ethylthio>ethane

-
A
-
505-29-3
1,4-Dithiane

-
B
-
74-87-3
methylene chloride

| Conditions | Yield |
|---|---|
| beim Erhitzen; |

| Conditions | Yield |
|---|---|
| With potassium pyrosulfate under 20 Torr; | |
| With potassium pyrosulfate; phosphoric acid; zinc(II) chloride at 200℃; |
-
-
37460-04-1
2,5,8-trithianonane

-
A
-
505-29-3
1,4-Dithiane

-
B
-
74-93-1
methylthiol

-
C
-
22322-43-6
2-methylsulfanyl-ethanethiol

-
D
-
1822-74-8
methyl vinyl sulfide

| Conditions | Yield |
|---|---|
| bei der trockener Destillation;Produkt5:1.2-Bis-<2-methylmercapto-aethylmercapto>-aethan; |


| Conditions | Yield |
|---|---|
| Destillation; | |
| beim Erhitzen; |

| Conditions | Yield |
|---|---|
| With hydrogen bromide; phenol |

| Conditions | Yield |
|---|---|
| at 90℃; |

| Conditions | Yield |
|---|---|
| With aluminum oxide at 300℃; |

| Conditions | Yield |
|---|---|
| With sodium sulfide; ethanol |
-
-
544-40-1
Dibutyl sulfide

-
-
106-93-4
ethylene dibromide

-
A
-
505-29-3
1,4-Dithiane

-
B
-
109-65-9
1-bromo-butane

-
-
505-29-3
1,4-Dithiane

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: THF; (Ar), organic compound added to soln. of Fe-complex, stirred for 3h (room temp.); filtered, concd. (vacuum), filtered into ether/petroleum ether (2/1), ppt. washed with ether and petroleum ether, dried (vacuum), elem. anal.; | 99% |
-
-
505-29-3
1,4-Dithiane

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: THF; (Ar), organic compound added to soln. of Fe-complex (molar ratio: 1/2), stirred for 12h (room temp.); pptd., filtered, washed with ether and petroleum ether, dried (vacuum), elem. anal.; | 98% |
-
-
505-29-3
1,4-Dithiane

-
-
12092-47-6
di-μ-chloro-bis(1,5-cyclooctadiene)dirhodium

-
-
26042-63-7
silver(I) hexafluorophosphate

-
-
68264-13-1
[Rh(cyclo-octa-1,5-diene)(dithian)]PF6

| Conditions | Yield |
|---|---|
| In dichloromethane N2-atmosphere; addn. of AgPF6 to suspn. of Rh-complex contg. excess of dithian, stirring for 10 min; filtration, evapn. of filtrate (vac.), crystn. (Me2CO/Et2O); elem. anal.; | 98% |

-
-
505-29-3
1,4-Dithiane

| Conditions | Yield |
|---|---|
| In acetone N2-atmosphere; stirring for 15 min; evapn. (reduced pressure), crystn. (Me2CO/Et2O); elem. anal.; | 95% |
-
-
505-29-3
1,4-Dithiane

-
-
79-08-3
bromoacetic acid

-
-
30843-64-2
1,4-Dithianium-essigsaeure-betain-hydrobromid

| Conditions | Yield |
|---|---|
| In nitromethane at 40℃; | 93% |
-
-
505-29-3
1,4-Dithiane

-
-
19087-70-8
1,4-dithiane 1-oxide

| Conditions | Yield |
|---|---|
| With nitric acid; Bu4(1+)*AuCl4(1-) In nitromethane; water for 2h; Ambient temperature; | 93% |
| With N-(tert-butyl)-N-chloro-cyanamide In water; acetonitrile at 25℃; | 90% |
| With sodium periodate In water; acetone at 20℃; for 24h; | 38.3% |
-
-
505-29-3
1,4-Dithiane

-
-
26042-63-7
silver(I) hexafluorophosphate

-
-
68246-34-4
[Pd(η3-1-phenylallyl)(dithian)]PF6

| Conditions | Yield |
|---|---|
| In acetone N2-atmosphere; addn. of AgPF6 to suspn. of Pd-complex contg. excess of dithian, stirring for 10 min; filtration, evapn. of filtrate (vac.), crystn. (Me2CO/Et2O); elem. anal.; | 92% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) Me2SnCl2 and 1,4-dithiane were heated at 140°C in a sealed tube under vac. for 12 h; sublimation at 70°C/0.5 mm Hg; elem. anal.; | 90% |
-
-
505-29-3
1,4-Dithiane

-
-
100203-27-8
CH3Co(HONC(CH3)C(CH3)NO)2(C4H8S2)Co(HONC(CH3)C(CH3)NO)2CH3

| Conditions | Yield |
|---|---|
| In dichloromethane (CH3Co(dmgH)2)2 and dithiane are stirred under N2 in CH2Cl2 at room temp. until all dissolved; ether added, ppt. collected by filtration, washed with ether and dried in vacuo at room temp.; elem. anal.; | 90% |

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane addn. of a soln. of 3 equivs. of 1,4-dithiane in CH2Cl2 to a soln. of 2 equivs. of silver salt in methanol, stirring at room temp. for 1 h; filtration, washing with CH2Cl2, drying in vac.; elem. anal.; | 90% |
-
-
505-29-3
1,4-Dithiane

-
-
26042-63-7
silver(I) hexafluorophosphate

-
-
12257-42-0
[(norbornadiene)rhodium(I)chloride]2

-
-
68264-15-3
[Rh(norbornadiene)(dithian)]PF6

| Conditions | Yield |
|---|---|
| In dichloromethane N2-atmosphere; addn. of AgPF6 to suspn. of Rh-complex contg. excess of dithian, stirring for 10 min; filtration, evapn. of filtrate (vac.), crystn. (Me2CO/Et2O); elem. anal.; | 90% |


| Conditions | Yield |
|---|---|
| With copper-iron mixed oxide In dichloromethane; acetonitrile at 140℃; for 24h; Sealed tube; | 90% |
-
-
505-29-3
1,4-Dithiane

-
-
22313-60-6
tert-butyl 4-methylbenzoperoxoate

| Conditions | Yield |
|---|---|
| With copper-iron mixed oxide In dichloromethane; acetonitrile at 140℃; for 24h; Sealed tube; | 89% |
-
-
505-29-3
1,4-Dithiane

-
-
78670-75-4
pentacarbonyl(tetrafluoroborato)rhenium

-
-
118504-51-1
{((CO)5Re)(C4H8S2)}(1+)*BF4(1-)={((CO)5Re)(C4H8S2)}BF4

| Conditions | Yield |
|---|---|
| In dichloromethane (Ar); addn. of 1,4-dithiane to a suspn. of one equiv. of (CO)5ReFBF3 and stirring at room temp. for 24 h;; sepn. of the residue; washing with CH2Cl2; vacuum drying; elem. anal.; | 87% |

| Conditions | Yield |
|---|---|
| In water; acetone addn. of a soln. of 1,4-dithiane in acetone to a soln. of silver salt inwater; elem. anal.; | 87% |
-
-
505-29-3
1,4-Dithiane

-
-
78670-75-4
pentacarbonyl(tetrafluoroborato)rhenium

| Conditions | Yield |
|---|---|
| In dichloromethane (Ar); addn. of 1,4-dithiane to a suspn. of two equiv. of (CO)5ReFBF3 and stirring at room temp. for 24 h;; sepn. of the residue; washing with CH2Cl2; vacuum drying; elem. anal.; | 85% |

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane addn. of a soln. of 1,4-dithiane in CH2Cl2 to a soln. of silver salt in methanol; elem. anal.; | 85% |
| In ethanol | |
| In ethanol |
-
-
505-29-3
1,4-Dithiane

-
-
243643-58-5
mer-[(1,4-bis(diphenylphosphino)butane)aquatrichlororuthenium(III)]

-
-
890526-28-0
mer-[RuCl3(1,4-bis(diphenylphosphino)butane)(1,4-dithiane)]

| Conditions | Yield |
|---|---|
| In dichloromethane all manipulations under Ar atm.; Ru and S compds. dissolved in CH2Cl2 with stirring at room temp. for 12 h; concd.; pptd. with Et2O,filtered, washed with Et2O, dried in vac.; elem.anal.; | 85% |

| Conditions | Yield |
|---|---|
| With copper-iron mixed oxide In dichloromethane; acetonitrile at 140℃; for 24h; Sealed tube; | 85% |
-
-
505-29-3
1,4-Dithiane

-
-
957188-75-9
[N-(trifluoromethylsulfonyl)imino][4-(trifluoromethyl)phenyl]-λ3-bromane

-
-
1251955-12-0
C5H8F3NO2S3

| Conditions | Yield |
|---|---|
| In dichloromethane at -78 - 0℃; for 2h; Inert atmosphere; | 84% |
-
-
505-29-3
1,4-Dithiane

-
-
43084-97-5
tert-butyl perester of 4-methoxybenzoic acid

| Conditions | Yield |
|---|---|
| With copper-iron mixed oxide In dichloromethane; acetonitrile at 140℃; for 24h; Sealed tube; | 83% |
-
-
505-29-3
1,4-Dithiane

| Conditions | Yield |
|---|---|
| In pentane a pentane solution of Pd(F6acac)2 was added to a solution of 1,4-C4H8S2 in pentane with stirring; filtrated and vac. dried; | 82% |
-
-
505-29-3
1,4-Dithiane

-
-
26042-63-7
silver(I) hexafluorophosphate

-
-
68246-32-2
[Pd(η3-2-methylallyl)(dithian)]PF6

| Conditions | Yield |
|---|---|
| In acetone N2-atmosphere; addn. of AgPF6 to suspn. of Pd-complex contg. excess of dithian, stirring for 10 min; filtration, evapn. of filtrate (vac.), crystn. (Me2CO/Et2O); elem. anal.; | 82% |

-
-
505-29-3
1,4-Dithiane

-
-
88496-72-4
RuCl2(1,4-bis(diphenylphosphino)butane)(PPh3)

-
-
890526-27-9
trans-[RuCl2(1,4-bis(diphenylphosphino)butane)(1,4-dithiane)]

| Conditions | Yield |
|---|---|
| In dichloromethane all manipulations under Ar atm.; mixt. of Ru and S compds. stirred in CH2Cl2 at room temp. for 30 min; concd.; pptd. with Et2O,filtered, washed with Et2O, dried in vac.; elem.anal.; | 80% |
-
-
505-29-3
1,4-Dithiane

-
-
15617-18-2, 39958-10-6, 14220-64-5
bis(benzonitrile)palladium(II) dichloride

| Conditions | Yield |
|---|---|
| With acetone In acetone N2-atmosphere; stirring for 16 h (pptn.); filtration, washing (Me2CO, Et2O), drying (vac., 56°C); elem. anal.; | 80% |

| Conditions | Yield |
|---|---|
| With bromine In ethanol; water to soln. Ru complex in aq. EtOH Br2 was added and stirred for 20 min, 3-fold excess ligand was added dropwise under Ar and allowed to stand for 1 day; react. mixt. was cooled in ice bath and acetone was added, ppt. was filtered, washed with acetone and Et2O, and dried; elem. anal.; | 80% |

| Conditions | Yield |
|---|---|
| With copper-iron mixed oxide In dichloromethane; acetonitrile at 140℃; for 24h; Sealed tube; | 80% |
-
-
505-29-3
1,4-Dithiane

| Conditions | Yield |
|---|---|
| In acetone byproducts: silver chloride; (N2); stoich. amt. of AgClO4 added to complex soln., stirred in dark for 5 min, slight excess of ligand added; filtered (Kieselguhr), collected over Et2O under N2, ppt. filtered off,washed with Et2O, dried in vac.; elem. anal.; | 79% |

1,4-Dithiane Consensus Reports
Reported in EPA TSCA Inventory.
1,4-Dithiane Specification
The 1,4-Dithiane with CAS registry number of 505-29-3 is also known as p-Dithiane(7CI,8CI). The IUPAC name and product name are the same. It belongs to product categories of Alphabetical Listings; C-D; Flavors and Fragrances; Heterocyclic Building Blocks; S-Containing. Its EINECS registry number is 208-007-2. In addition, the formula is C4H8S2 and the molecular weight is 120.24. This chemical is a white solid with an extremely unpleasant smell.
Physical properties about 1,4-Dithiane are: (1)ACD/LogP: 1.12; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.12; (4)ACD/LogD (pH 7.4): 1.12; (5)ACD/BCF (pH 5.5): 4.2; (6)ACD/BCF (pH 7.4): 4.2; (7)ACD/KOC (pH 5.5): 97.25; (8)ACD/KOC (pH 7.4): 97.25; (9)#H bond acceptors: 0; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 50.6Å2; (13)Index of Refraction: 1.573; (14)Molar Refractivity: 34.77 cm3; (15)Molar Volume: 105.4 cm3; (16)Polarizability: 13.78×10-24cm3; (17)Surface Tension: 43.8 dyne/cm; (18)Density: 1.14 g/cm3; (19)Flash Point: 82.1 °C; (20)Enthalpy of Vaporization: 41.88 kJ/mol; (21)Boiling Point: 200.5 °C at 760 mmHg; (22)Vapour Pressure: 0.459 mmHg at 25 °C
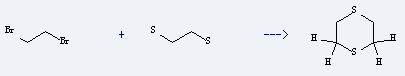
Preparation of 1,4-Dithiane: it is prepared by reaction of ethane-1,2-dithiol with 1,2-dibromo-ethane. The cyclization reaction needs reagent sodium ethoxide and solvent ethanol with other condition of heating for 2.5 hours. The yield is about 64%.
Uses of 1,4-Dithiane: it is used to produce [1,4]dithiane-1-oxide. The reaction occurs with reagent m-chloroperbenzoic acid. The yield is about 36%.
![1,4-Dithiane is used to produce [1,4]dithiane-1-oxide.](/UserFilesUpload/Uses of 1,4-Dithiane.png)
When you are using this chemical, please be cautious about it. As a chemical, it is irritating to eyes, respiratory system and skin. During using it, wear suitable protective clothing. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
1. SMILES: S1CCSCC1
2. InChI: InChI=1/C4H8S2/c1-2-6-4-3-5-1/h1-4H2
3. InChIKey: LOZWAPSEEHRYPG-UHFFFAOYAR
4. Std. InChI: InChI=1S/C4H8S2/c1-2-6-4-3-5-1/h1-4H2
5. Std. InChIKey: LOZWAPSEEHRYPG-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | oral | 2768mg/kg (2768mg/kg) | BEHAVIORAL: TREMOR BEHAVIORAL: ATAXIA LUNGS, THORAX, OR RESPIRATION: DYSPNEA | National Technical Information Service. Vol. AD-A172-647, |
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 50529-33-4
- 50530-12-6
- 50530-43-3
- 5053-08-7
- 5053-24-7
- 505-32-8
- 50-53-3
- 50533-97-6
- 5053-43-0
- 50534-45-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View