-
Name
6-PHENYL-2-THIOURACIL
- EINECS 253-227-4
- CAS No. 36822-11-4
- Article Data26
- CAS DataBase
- Density 1.37 g/cm3
- Solubility
- Melting Point 263-264.5 °C(Solv: water (7732-18-5))
- Formula C10H8N2OS
- Boiling Point 462.3 °C at 760 mmHg
- Molecular Weight 204.252
- Flash Point 233.4 °C
- Transport Information
- Appearance
- Safety 24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Uracil,6-phenyl-2-thio- (6CI,7CI,8CI);2-Mercapto-6-phenylpyrimidin-4-ol;6-Phenyl-2-thiouracil;6-Phenylthiouracil;Fenucil;NSC 163991;NSC 42600;
- PSA 84.81000
- LogP 2.13790
4(1H)-Pyrimidinone,2,3-dihydro-6-phenyl-2-thioxo- Specification
The 4(1H)-Pyrimidinone,2,3-dihydro-6-phenyl-2-thioxo-, with the CAS registry number 36822-11-4 and EINECS registry number 253-227-4, has the systematic name of 6-phenyl-2-thioxo-2,3-dihydropyrimidin-4(1H)-one. It belongs to the following product categories: Biochemistry; Nucleobases and their analogs; Nucleosides, Nucleotides & Related Reagents; Biochemicals and Reagents; Nucleoside Analogs; Nucleosides, Nucleotides, Oligonucleotides. And the molecular formula of the chemical is C10H8N2OS.
The characteristics of 4(1H)-Pyrimidinone,2,3-dihydro-6-phenyl-2-thioxo- are as followings: (1)ACD/LogP: 1.92; (2)# of Rule of 5 Violations: 0; (3)ACD/BCF (pH 5.5): 1; (4)ACD/BCF (pH 7.4): 1; (5)ACD/KOC (pH 5.5): 1; (6)ACD/KOC (pH 7.4): 1; (7)#H bond acceptors: 3; (8)#H bond donors: 2; (9)#Freely Rotating Bonds: 1; (10)Polar Surface Area: 55.64 Å2; (11)Index of Refraction: 1.702; (12)Molar Refractivity: 57.44 cm3; (13)Molar Volume: 148.1 cm3; (14)Polarizability: 22.77×10-24cm3; (15)Surface Tension: 68.7 dyne/cm; (16)Density: 1.37 g/cm3; (17)Flash Point: 233.4 °C; (18)Enthalpy of Vaporization: 75.1 kJ/mol; (19)Boiling Point: 462.3 °C at 760 mmHg; (20)Vapour Pressure: 3.62E-09 mmHg at 25°C.
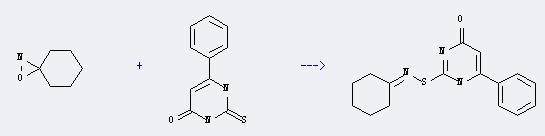
Uses of 4(1H)-Pyrimidinone,2,3-dihydro-6-phenyl-2-thioxo-: It can react with 1-oxa-2-aza-spiro[2.5]octane to produce 2-(Cyclohexylidenaminothio)-6-phenylpyrimidin-4-on. This reaction will need reagent toluene and dimethylformamide. The reaction time is 12 hours with temperature of 20°C, and the yield is about 53%.
Addtionally, the following datas could be converted into the molecular structure:
(1)SMILES: S=C2NC(/c1ccccc1)=C\C(=O)N2
(2)InChI: InChI=1/C10H8N2OS/c13-9-6-8(11-10(14)12-9)7-4-2-1-3-5-7/h1-6H,(H2,11,12,13,14)
(3)InChIKey: XEKNACRTWJHOCE-UHFFFAOYAZ
Related Products
- 4(1H)-Pyrimidinone, 6-amino-2-chloro- (9CI)
- 4(1H)-Pyrimidinone,2,3-dihydro-5-(hydroxymethyl)-2-thioxo-
- 4(1H)-Pyrimidinone,2,3-dihydro-5-propyl-2-thioxo-
- 4(1H)-Pyrimidinone,2,3-dihydro-6-phenyl-2-thioxo-
- 4(1H)-Pyrimidinone,5-butyl-2,3-dihydro-6-propyl-2-thioxo-
- 4(1H)-Pyrimidinone,6-amino-5-(2,2-diethoxyethyl)-2,3-dihydro-2-thioxo-
- 4(1H)-Pyrimidinone,6-chloro-2-methyl-5-nitro-
- 4(1H)-Pyrimidinone,6-fluoro-2-(methylamino)-
- 4(1H)-Pyrimidinone,6-hydroxy-2-methyl-
- 3682-26-6
- 3682-35-7
- 36823-84-4
- 36823-88-8
- 3682-43-7
- 3682-47-1
- 36824-73-4
- 36825-36-2
- 36830-74-7
- 3683-12-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View