-
Name
5-Amino-4,6-dichloropyrimidine
- EINECS 226-503-7
- CAS No. 5413-85-4
- Article Data31
- CAS DataBase
- Density 1.606 g/cm3
- Solubility slightly soluble in water
- Melting Point 145-148 °C(lit.)
- Formula C4H3Cl2N3
- Boiling Point 267.1 °C at 760 mmHg
- Molecular Weight 163.994
- Flash Point 115.3 °C
- Transport Information
- Appearance Yellowish-brown crystals or powder
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 5-Pyrimidinamine, 4,6-dichloro-;4,6-Dichloro-5-aminopyrimidine;Pyrimidine, 5-amino-4,6-dichloro-;4,6-dichloropyrimidin-5-amine;4,6-Dichloropyrimidin-5-ylamine;
- PSA 51.80000
- LogP 1.94680
Synthetic route

| Conditions | Yield |
|---|---|
| With tin(ll) chloride In ethanol for 1h; Heating; | 99% |
| With tin(ll) chloride In ethanol for 0.333333h; Heating; | 91% |
| With palladium 10% on activated carbon; hydrogen In ethyl acetate at 20℃; for 4h; | 86% |

-
-
5413-85-4
5-amino-4,6-dichloropyridimine

| Conditions | Yield |
|---|---|
| 64% |
-
-
69340-97-2
5-amino-4,6-dihydroxy-pyrimidine

-
-
5413-85-4
5-amino-4,6-dichloropyridimine

| Conditions | Yield |
|---|---|
| With tetraethylammonium chloride; trichlorophosphate at 80 - 110℃; for 3h; | 54.1% |
| With trichlorophosphate at 110℃; for 48h; Inert atmosphere; Sealed tube; | 44 mg |
| With trichlorophosphate at 110℃; for 48h; Sealed tube; Inert atmosphere; | 44 mg |
-
-
64-18-6
formic acid

-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
123240-66-4
4,6-dichloro-5-formamidopyrimidine

| Conditions | Yield |
|---|---|
| With acetic anhydride at 20℃; for 2h; | 100% |
| With acetic anhydride at 0 - 20℃; for 2.5h; | 100% |
| With acetic acid at 0 - 30℃; Autoclave; Large scale; | 89.7% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| With thionyl chloride at 0 - 20℃; for 1h; Inert atmosphere; | 100% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
138405-07-9
(+/-)-(1β,3α,4β)-3,4-bis(hydroxymethyl)-1-cyclobutylamine

-
-
138405-08-0
(+/-)-5-amino-4-chloro-6-<<(1β,3α,4β)-3,4-bis(hydroxymethyl)cyclopentan-1-yl>amino>pyrimidine

| Conditions | Yield |
|---|---|
| With triethylamine In butan-1-ol for 48h; Heating; | 99% |
| With triethylamine In butan-1-ol at 100℃; for 48h; | 48% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
57260-71-6
1-t-Butoxycarbonylpiperazine

-
-
853679-43-3
tert-butyl 4-(5-amino-6-chloropyrimidin-4-yl)piperazine-1-carboxylate

| Conditions | Yield |
|---|---|
| In triethylamine; toluene for 12h; Heating / reflux; | 99% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In 1,4-dioxane; water Reflux; | 99% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
88474-32-2
4-chloro-5-cyano-1,2,3-triazole

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium nitrite In water 1.)-5 deg C, 1.5 h 2.)20 deg C, 1.5 h; | 98% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
106-47-8
4-chloro-aniline

-
-
103505-49-3
6-chloro-N4-(4-chlorophenyl)-4,5-pyrimidinediamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; water at 20 - 82℃; for 79h; | 98% |
| With hydrogenchloride In ethanol; water at 20 - 82℃; for 79h; | 98% |
| With hydrogenchloride In ethanol; water at 20 - 82℃; for 79h; | 98% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
536-90-3
m-Anisidine

-
-
870257-99-1
6-chloro-N4-(3-methoxy-phenyl)-pyrimidine-4,5-diamine

| Conditions | Yield |
|---|---|
| In ethanol | 98% |
| In ethanol at 80℃; Acidic conditions; | 80% |
| With hydrogenchloride In ethanol; water for 24h; Heating; | 68% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 50℃; for 16h; | 98% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
637-89-8
4-sulfanylphenol

-
-
873106-32-2
5-amino-4,6-bis(p-hydroxyphenylthio)pyrimidine

| Conditions | Yield |
|---|---|
| With triethylamine In butan-1-ol Heating; | 98% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In ethanol; water; toluene Suzuki-Miyaura Coupling; Reflux; | 98% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In ethanol; water; toluene for 20h; Inert atmosphere; Reflux; | 98% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In 1,4-dioxane; water at 80℃; for 16h; Suzuki-Miyaura Coupling; Inert atmosphere; regioselective reaction; | 91% |

| Conditions | Yield |
|---|---|
| With ammonia In water at 100℃; for 46h; | 97% |
| With ammonia at 100℃; for 46h; | 97% |
| With ammonia In water at 100℃; for 46h; | 97% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

| Conditions | Yield |
|---|---|
| With triethylamine In butan-1-ol at 120℃; for 48h; | 97% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
56777-11-8
4-chloro-<5-<14N>,6-<15N>>diaminopyrimidine

| Conditions | Yield |
|---|---|
| With ammonia In ethanol at 120 - 130℃; under 5171.5 Torr; for 7h; | 97% |
| With ammonia at 120 - 140℃; under 5171.5 Torr; for 7h; | 95% |
| With [15N]ammonia In ethanol at 140℃; for 4h; | 81% |
| With (15N)-ammonium chloride; potassium hydrogencarbonate In dimethyl sulfoxide at 80℃; for 50h; | 4.3 mmol |

-
-
5413-85-4
5-amino-4,6-dichloropyridimine

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol at 105℃; for 144h; | 97% |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In butan-1-ol at 117℃; for 48h; | 97% |
-
-
107-10-8
propylamine

-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
195252-56-3
4,5-diamino-6-chloro-N4-propylpyrimidine

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol at 80℃; for 8h; | 96% |
| With triethylamine In butan-1-ol for 16h; Heating; | 95% |
| In water; isopropyl alcohol at 80℃; for 16h; | |
| With N-ethyl-N,N-diisopropylamine In 1,4-dioxane at 100℃; for 16h; |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
103-72-0
phenyl isothiocyanate

-
-
871266-78-3
(7-chloro[1,3]thiazolo[5,4-d]pyrimidin-2-yl)(phenyl)amine

| Conditions | Yield |
|---|---|
| With potassium fluoride on basic alumina In acetonitrile at 60℃; for 5h; Reagent/catalyst; Solvent; Temperature; Time; | 96% |
| In acetonitrile at 60℃; for 5h; | 96% |
| With caesium carbonate In acetonitrile at 50℃; for 16h; | 92% |
| With caesium carbonate In acetonitrile at 50℃; for 12h; |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
106-54-7
p-Chlorothiophenol

-
-
873106-30-0
5-amino-4,6-bis(p-chlorophenylthio)pyrimidine

| Conditions | Yield |
|---|---|
| With triethylamine In butan-1-ol Heating; | 96% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 18h; | 96% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen; palladium on activated charcoal In diethyl ether at 25℃; under 1551.44 Torr; | 95% |
| With sodium hydroxide; hydrogen; palladium 10% on activated carbon In diethyl ether; water at 20℃; for 72h; | 31.9% |
| With palladium on activated charcoal Hydrogenation; |

| Conditions | Yield |
|---|---|
| 95% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

| Conditions | Yield |
|---|---|
| 95% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
64-04-0
phenethylamine

-
-
73840-54-7
6-chloro-N4-phenethylpyrimidine-4,5-diamine

| Conditions | Yield |
|---|---|
| With triethylamine In butan-1-ol at 100℃; for 12h; | 95% |
| With triethylamine In butan-1-ol for 5h; Inert atmosphere; Schlenk technique; | 92% |
| With triethylamine In butan-1-ol at 120℃; for 4h; Heating / reflux; | 90% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 50℃; for 16h; | 95% |
| With potassium fluoride on basic alumina In acetonitrile at 60℃; for 5h; | 91% |
| In acetonitrile at 60℃; for 5h; | 91% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
108-98-5
thiophenol

-
-
873106-27-5
5-amino-4,6-bis(phenylthio)pyrimidine

| Conditions | Yield |
|---|---|
| With triethylamine In butan-1-ol Heating; | 95% |
| With triethylamine In butan-1-ol Heating / reflux; | 95% |
-
-
5413-85-4
5-amino-4,6-dichloropyridimine

-
-
106-45-6
para-thiocresol

-
-
873106-29-7
5-amino-4,6-bis(p-tolylthio)pyrimidine

| Conditions | Yield |
|---|---|
| With triethylamine In butan-1-ol Heating; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Phenyl-1-propanol With sodium hydride In tetrahydrofuran at 20℃; for 0.666667h; Inert atmosphere; Stage #2: 5-amino-4,6-dichloropyridimine In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen; palladium 10% on activated carbon In tetrahydrofuran at 20℃; for 24h; | 94.3% |
5-Amino-4,6-dichloropyrimidine Specification
The IUPAC name of 5-Amino-4,6-dichloropyrimidine is 4,6-dichloropyrimidin-5-amine. With the CAS registry number 5413-85-4 and EINECS 226-503-7, it is also named as 5-Pyrimidinamine, 4,6-dichloro-. The product's categories are Pyridines, Pyrimidines, Purines and Pteredines; Pyrimidines; Heterocyclic Compounds; Bases & Related Reagents; Heterocycles; Nucleotides; Building Blocks; Heterocyclic Building Blocks. It is yellowish-brown crystals or powder which is stable under normal temperature and pressure. Additionally, this chemical should be sealed in the container, kept away from direct sunshine and stored in the cool and dry place.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 0.76; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.76; (4)ACD/LogD (pH 7.4): 0.76; (5)ACD/BCF (pH 5.5): 2.23; (6)ACD/BCF (pH 7.4): 2.23; (7)ACD/KOC (pH 5.5): 61.73; (8)ACD/KOC (pH 7.4): 61.73; (9)#H bond acceptors: 3; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 29.02 Å2; (13)Index of Refraction: 1.633; (14)Molar Refractivity: 36.46 cm3; (15)Molar Volume: 102 cm3; (16)Polarizability: 14.45×10-24 cm3; (17)Surface Tension: 68 dyne/cm; (18)Density: 1.606 g/cm3; (19)Flash Point: 115.3 °C; (20)Enthalpy of Vaporization: 50.51 kJ/mol; (21)Boiling Point: 267.1 °C at 760 mmHg; (22)Vapour Pressure: 0.00832 mmHg at 25°C.
Preparation of 5-Amino-4,6-dichloropyrimidine: It can be obtained by 4,6-dichloro-5-nitro-pyrimidine. This reaction needs reagent SnCl2*2H2O and solvent ethanol by heating. The reaction time is 20 min. The yield is 91%.
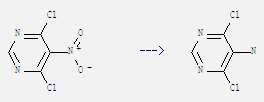
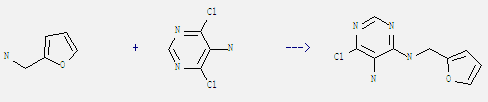
Uses of 5-Amino-4,6-dichloropyrimidine: It is an intermediate in the production of many biological inhibitors. And it can react with furfurylamine to get 6-chloro-N4-furfuryl-pyrimidine-4,5-diyldiamine. This reaction needs reagent triethylamine and solvent butan-1-ol at ambient temperature. The reaction time is 2 hours. The yield is 78%.
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. So people should avoid contact with skin and eyes. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If you want to contact this product, you must wear suitable protective clothing.
People can use the following data to convert to the molecule structure.
1. SMILES:Clc1ncnc(Cl)c1N
2. InChI:InChI=1/C4H3Cl2N3/c5-3-2(7)4(6)9-1-8-3/h1H,7H2
3. InChIKey:NIGDWBHWHVHOAD-UHFFFAOYAN
Related Products
- 5-Amino-4,6-dichloro-2-methylpyrimidine
- 5-Amino-4,6-dichloropyrimidine
- 5-Amino-4,6-dihydroxypyrimidine
- 5-Amino-4-azaindole
- 5-Amino-4-bromo-1-methyl-1H-pyrazole-3-carboxylic acid ethyl ester
- 5-Amino-4-bromobenzothiazole
- 5-Amino-4-chloro-2-methylphenol
- 5-Amino-4-chloro-2-methylphenyl ethyl carbonate
- 5-Amino-4-chloropyrimidine
- 5-Amino-4-chlorosalicylic acid
- 541-41-3
- 5414-19-7
- 5414-21-1
- 541-42-4
- 54142-64-2
- 54143-55-4
- 54143-56-5
- 54143-57-6
- 5414-61-9
- 541-46-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View