-
Name
5-Chloro-1-(4-piperidyl)-2-benzimidazolinone
- EINECS 258-771-6
- CAS No. 53786-28-0
- Article Data8
- CAS DataBase
- Density 1.323 g/cm3
- Solubility 549mg/L at 20℃
- Melting Point 228-231 °C(lit.)
- Formula C12H14ClN3O
- Boiling Point
- Molecular Weight 251.716
- Flash Point
- Transport Information
- Appearance
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 4-(6-Chloro-1,2-dihydro-2-oxobenzo[d]imidazol-3-yl)piperidine;5-Chloro-1-(4-piperidinyl)-1,3-dihydro-2H-benzimidazol-2-one;5-Chloro-1-(piperidin-4-yl)-1H-benzimidazol-2(3H)-one;5-Chloro-1-piperidin-4-yl-1,3-dihydrobenzimidazol-2-one;R 29676;
- PSA 49.82000
- LogP 2.23630
Synthetic route
-
-
53786-46-2
4-(5-chloro-2-oxo-2,3-dihydro-benzoimidazol-1-yl)-piperidine-1-carboxylic acid ethyl ester

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide for 8h; Heating; | 96% |
| With sodium hydroxide at 100℃; | |
| With ammonium chloride In water |
-
-
58859-46-4
ethyl 4-aminopiperidine-1-carboxylate

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: Na2CO3; KI / cyclohexanol / 0.17 h / 180 °C / microwave irradiation 2: Zn; aq. HCl / methanol / 50 °C 3: acetonitrile 4: aq. NaOH / 100 °C View Scheme |
-
-
53786-45-1
ethyl 4-[(2-amino-4-chlorophenyl)amino]piperidine-1-carboxylate

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: acetonitrile 2: aq. NaOH / 100 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 90 percent / Et3N / CH2Cl2 / 0.5 h / 0 °C 2: 96 percent / 10percent NaOH / 8 h / Heating View Scheme |
-
-
53786-44-0
4-(4-chloro-2-nitro-anilino)-piperidine-1-carboxylic acid ethyl ester

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Zn; aq. HCl / methanol / 50 °C 2: acetonitrile 3: aq. NaOH / 100 °C View Scheme | |
| Multi-step reaction with 3 steps 1: 93 percent / H2 / Raney Ni / ethanol; tetrahydrofuran / 1 h / Ambient temperature 2: 90 percent / Et3N / CH2Cl2 / 0.5 h / 0 °C 3: 96 percent / 10percent NaOH / 8 h / Heating View Scheme |
-
-
58859-46-4
ethyl 4-aminopiperidine-1-carboxylate

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 65 percent / Na2CO3, KI / various solvent(s) / 28 h / 160 °C 2: 93 percent / H2 / Raney Ni / ethanol; tetrahydrofuran / 1 h / Ambient temperature 3: 90 percent / Et3N / CH2Cl2 / 0.5 h / 0 °C 4: 96 percent / 10percent NaOH / 8 h / Heating View Scheme |
-
-
89-61-2
2,5-dichloronitrobenzene

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 65 percent / Na2CO3, KI / various solvent(s) / 28 h / 160 °C 2: 93 percent / H2 / Raney Ni / ethanol; tetrahydrofuran / 1 h / Ambient temperature 3: 90 percent / Et3N / CH2Cl2 / 0.5 h / 0 °C 4: 96 percent / 10percent NaOH / 8 h / Heating View Scheme |
-
-
87120-72-7
1-(tert-butoxycarbonyl)-4-aminopiperidine

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium carbonate / dimethyl sulfoxide / 20 °C / Inert atmosphere 2: hydrazine hydrate / water; ethanol / 0.17 h / 45 °C 3: tetrahydrofuran / 20 °C / Inert atmosphere 4: trifluoroacetic acid / dichloromethane / 20 °C View Scheme |
-
-
345-18-6
2-fluoro-5-chloronitrobenzene

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium carbonate / dimethyl sulfoxide / 20 °C / Inert atmosphere 2: hydrazine hydrate / water; ethanol / 0.17 h / 45 °C 3: tetrahydrofuran / 20 °C / Inert atmosphere 4: trifluoroacetic acid / dichloromethane / 20 °C View Scheme |
-
-
460047-89-6
tert-butyl 4-((4-chloro-2-nitrophenyl)amino)piperidine-1-carboxylate

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: hydrazine hydrate / water; ethanol / 0.17 h / 45 °C 2: tetrahydrofuran / 20 °C / Inert atmosphere 3: trifluoroacetic acid / dichloromethane / 20 °C View Scheme |
-
-
1429415-80-4
tert-butyl 4-((2-amino-4-chlorophenyl)amino)piperidine-1-carboxylate

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrahydrofuran / 20 °C / Inert atmosphere 2: trifluoroacetic acid / dichloromethane / 20 °C View Scheme |
-
-
460047-90-9
tert-butyl 4-(5-chloro-2-oxo-2,3-dihydro-1H-benzo[d]imidazole-1-yl)piperidine-1-carboxylate

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; |
-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With nitric acid In o-xylene at 60℃; | 95% |
| With nitric acid In 1,2-dichloro-benzene at 60℃; for 2h; | 64% |
| With nitric acid In 1,2-dichloro-benzene at 20 - 60℃; for 2h; | 64% |
| With nitric acid In o-xylene at 60℃; for 2h; |
-
-
1146300-32-4
7-chloro-1-(2,4-difluorophenyl)-1,4-dihydro-8-methyl-6-nitro-4-oxoquinolone-3-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1146300-60-8
7-(4-(6-chloro-1,2-dihydro-2-oxobenzo[d]imidazol-3-yl)piperidin-1-yl)-1-(2,4-difluorophenyl)-1,4-dihydro-8-methyl-6-nitro-4-oxoquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide Microwave irradiation; | 91% |
-
-
1146300-33-5
1-tert-butyl-7-chloro-1-1,4-dihydro-8-methyl-6-nitro-4-oxoquinolone-3-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1146300-77-7
1-tert-butyl-7-(4-(6-chloro-1,2-dihydro-2-oxobenzo[d]imidazol-3-yl)piperidin-1-yl)-1,4-dihydro-8-methyl-6-nitro-4-oxoquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide Microwave irradiation; | 89% |
-
-
527-72-0
2-thiophenylcarboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With pyridine; 1,3,5-trichloro-2,4,6-triazine In acetonitrile at 140℃; for 0.0833333h; Microwave irradiation; | 89% |
-
-
40133-08-2
5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-2-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With pyridine; 1,3,5-trichloro-2,4,6-triazine In acetonitrile at 140℃; for 0.0833333h; Microwave irradiation; | 85% |
-
-
1146300-31-3
7-chloro-1-cyclopropyl-1,4-dihydro-8-methyl-6-nitro-4-oxoquinolone-3-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1146300-43-7
7-(4-(6-chloro-1,2-dihydro-2-oxobenzo[d]imidazol-3-yl)piperidin-1-yl)-1-cyclopropyl-1,4-dihydro-8-methyl-6-nitro-4-oxoquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide Microwave irradiation; | 82% |
-
-
960313-86-4
1-(4-fluorophenyl)-1,4-dihydro-7-methoxy-6-nitro-4-oxo-1,8-naphthyridine-3-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide microwave irradiation; | 78% |
-
-
236743-93-4
(S)-9,10-difluoro-3-methyl-8-nitro-7-oxo-2,3-dihydro-7H-[1,4]oxazino[2,3,4-ij]quinoline-6-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide microwave irradiation; | 78% |
-
-
112811-77-5
1-cyclopropyl-6,7-difluoro-1,4-dihydro-5-nitro-8-methoxy-4-oxoquinoline-3-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1018937-91-1
C26H23ClFN5O7

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide for 0.0333333h; microwave irradiation; | 78% |
-
-
368839-20-7
7-chloro-1-(4-fluorophenyl)-1,4-dihydro-6-nitro-4-oxoquinoline-3-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1119087-61-4
7-(4-(6-chloro-1,2-dihydro-2-oxobenzo[d]imidazol-3-yl)piperidin-1-yl)-1-(4-fluorophenyl)-1,4-dihydro-6-nitro-4-oxoquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide Microwave irradiation; | 78% |
-
-
1585-11-1
5-chloro-2,2-diphenylpentanenitrile

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With sodium carbonate; sodium iodide In acetonitrile at 160℃; under 12901 Torr; for 0.25h; microwave irradiation; | 76% |
-
-
960313-85-3
1-cyclopropyl-1,4-dihydro-7-methoxy-6-nitro-4-oxo-1,8-naphthyridine-3-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide microwave irradiation; | 76% |
-
-
15936-81-9
5‑bromo‑7‑methylindole

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1248590-98-8
5-chloro-1-(1-(7-methyl-1H-indol-5-yl)piperidin-4-yl)-1H-benzo[d]imidazol-2(3H)-one

| Conditions | Yield |
|---|---|
| With RuPhos palladacycle; lithium hexamethyldisilazane; ruphos In tetrahydrofuran at 65℃; for 4h; Inert atmosphere; Sealed vial; | 76% |
-
-
5394-18-3
2-(4-bromobutyl)isoindoline-1,3-dione

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate; sodium iodide In acetonitrile Reflux; | 75.2% |
-
-
102614-43-7
2-chloro-3-fluoro-5,12-dihydro-5-oxobenzothiazolo<3,2-a>quinoline-6-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1028203-07-7
C28H20ClFN4O4S

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide microwave irradiation; | 75% |
-
-
1028202-77-8
2-chloro-3-nitro-5,12-dihydro-5-oxobenzothiazolo[3,2-a]quinoline-6-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1028203-08-8
C28H20ClN5O6S

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide microwave irradiation; | 74% |
-
-
960313-87-5
1-tert-butyl-1,4-dihydro-7-methoxy-6-nitro-4-oxo-1,8-naphthyridine-3-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide microwave irradiation; | 72% |
-
-
112811-72-0
1-cyclopropyl-6,7-difluoro-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1018937-88-6
C26H24ClFN4O5

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide for 0.0333333h; microwave irradiation; | 70% |
-
-
129722-34-5
7-(4-bromobutoxy)-3,4-dihydro-2(1H)-quinolinone

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1314032-14-8
7-(4-(4-(5-chloro-2-oxo-2,3-dihydro-1H-benzo[d]imidazol-1-yl)piperidin-1-yl)butoxy)-3,4-dihydroquinolin-2(1H)-one

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In acetonitrile Reflux; Inert atmosphere; | 70% |
-
-
494204-73-8
1-tert-butyl-7-chloro-1,4-dihydro-6-nitro-4-oxoquinoline-3-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1119088-12-8
1-tert-butyl-7-(4-(6-chloro-1,2-dihydro-2-oxobenzo[d]imidazol-3-yl)piperidin-1-yl)-1,4-dihydro-6-nitro-4-oxoquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide Microwave irradiation; | 69% |
-
-
100986-89-8
levofloxacin Q-acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide microwave irradiation; | 67% |
-
-
93107-29-0
1-cyclopropyl-6-nitro-4-oxo-7-chloro-1,4-dihydroquinoline-3-carboxylic acid

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1119087-10-3
7-(4-(6-chloro-1,2-dihydro-2-oxobenzo[d]imidazol-3-yl)piperidin-1-yl)-1-cyclopropyl-1,4-dihydro-6-nitro-4-oxoquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide Microwave irradiation; | 67% |

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| Stage #1: 1-(3-iodopropyl)-2,3-dihydro(4,5,6,7-D4)-1H-1,3-benzodiazol-2-one; 5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one In tetrahydrofuran; N,N-dimethyl-formamide for 2.16667h; Inert atmosphere; Stage #2: With potassium carbonate In tetrahydrofuran; N,N-dimethyl-formamide at 20℃; for 48h; | 64% |
-
-
57848-46-1
4-bromo-2-fluorobenzaldehyde

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; acetic acid In 1,2-dichloro-ethane at 20℃; Inert atmosphere; | 63% |
-
-
2164-34-3
2-(bromomethyl)-2,3-dihydrobenzo[1,4]dioxine

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
85076-06-8
5-Chloro-1-[1-(2,3-dihydro-benzo[1,4]dioxin-2-ylmethyl)-piperidin-4-yl]-1,3-dihydro-benzoimidazol-2-one

| Conditions | Yield |
|---|---|
| With triethylamine; sodium iodide In N,N-dimethyl-formamide for 60h; Ambient temperature; | 60% |
-
-
1089210-20-7
2,3-dihydro-2,2-dimethyl-5-chloro-7-(oxiranyl-methoxy)-benzofuran

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
1089209-50-6
1-((2,3-dihydro-2,2-dimethyl-5-chloro-benzofuran-7-yl)-oxy)-3-(4-(2,3-dihydro-2-oxo-5-chloro-benzoimidazol-1-yl)-piperidin-1-yl)-propan-2-ol

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 72h; | 58% |
-
-
107617-97-0
7-fluoro-2-<(tosyloxy)methyl>-1,4-benzodioxan

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
107617-74-3
5-Chloro-1-[1-(7-fluoro-2,3-dihydro-benzo[1,4]dioxin-2-ylmethyl)-piperidin-4-yl]-1,3-dihydro-benzoimidazol-2-one

| Conditions | Yield |
|---|---|
| With triethylamine; potassium iodide In dimethyl sulfoxide at 80℃; for 20h; | 57% |
-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
625446-22-2
4-bromo-2-fluoroacetophenone

| Conditions | Yield |
|---|---|
| Stage #1: 5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one; 4-bromo-2-fluoroacetophenone With titanium(IV) isopropylate at 60℃; for 2h; Inert atmosphere; Stage #2: With sodium tris(acetoxy)borohydride In tetrahydrofuran at 20℃; Inert atmosphere; | 56% |
-
-
3635-70-9
6,7-dimethyl-2-(toluene-4-sulfonyloxymethyl)-2,3-dihydro-benzo[1,4]dioxine

-
-
53786-28-0
5-chloro-1-(piperidin-4-yl)-1,3-dihydro-2H-benzo[d]imidazol-2-one

-
-
85076-21-7
1-[(6,7-Dimethyl-benzo-1,4-dioxan-2-yl)-methyl]-4-(5-chlorobenzimidazol-2-on-1-yl)-piperidine

| Conditions | Yield |
|---|---|
| With triethylamine; potassium iodide In dimethyl sulfoxide at 80℃; for 20h; | 55% |
5-Chloro-1-(4-piperidyl)-2-benzimidazolinone Chemical Properties
Molecule structure of 5-Chloro-1-(4-piperidyl)-2-benzimidazolinone (CAS NO.53786-28-0):
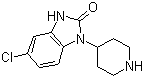
IUPAC Name: 6-Chloro-3-piperidin-4-yl-1H-benzimidazol-2-one
Molecular Weight: 251.71206 [g/mol]
Molecular Formula: C12H14ClN3O
Index of Refraction: 1.605
Molar Refractivity: 65.59 cm3
Molar Volume: 190.2 cm3
Polarizability: 26×10-24 cm3
Melting Point: 228-231 °C(lit.)
XLogP3: 2.6
H-Bond Donor: 2
H-Bond Acceptor: 2
Rotatable Bond Count: 1
Tautomer Count: 2
Exact Mass: 251.08254
MonoIsotopic Mass: 251.08254
Topological Polar Surface Area: 44.4
Heavy Atom Count: 17
Complexity: 306
Canonical SMILES: C1CNCCC1N2C3=C(C=C(C=C3)Cl)NC2=O
InChI: InChI=1S/C12H14ClN3O/c13-8-1-2-11-10(7-8)15-12(17)16(11)9-3-5-14-6-4-9/h1-2,7,9,14H,3-6H2,(H,15,17)
InChIKey: DOAYWDKFDPSTSV-UHFFFAOYSA-N
EINECS: 258-771-6
Product Categories of 5-Chloro-1-(4-piperidyl)-2-benzimidazolinone (CAS NO.53786-28-0): Benzimidazoles; BenzimidazolesHeterocyclic Building Blocks; Building Blocks; Halogenated Heterocycles; Heterocyclic Building Blocks
5-Chloro-1-(4-piperidyl)-2-benzimidazolinone Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
WGK Germany: 3
5-Chloro-1-(4-piperidyl)-2-benzimidazolinone Specification
5-Chloro-1-(4-piperidyl)-2-benzimidazolinone (CAS NO.53786-28-0) is also named as 5-Chloro-1,3-dihydro-1-(4-piperidinyl)-2H-benzimidazol-2-one ; 2H-Benzimidazol-2-one,5-chloro-1,3-dihydro-1-(4-piperidinyl)- ; 4-(6-Chloro-1,2-dihydro-2-oxobenzo[d]imidazol-3-yl)piperidine ; 5-Chloro-1-(4-piperidinyl)-1,3-dihydro-2H-benzimidazol-2-one ; 5-Chloro-1-(piperidin-4-yl)-1H-benzimidazol-2(3H)-one ; 5-Chloro-1-piperidin-4-yl-1,3-dihydrobenzimidazol-2-one ; R 29676 . 5-Chloro-1-(4-piperidyl)-2-benzimidazolinone (CAS NO.53786-28-0) is white solid.
Related Products
- 5-Chloro-1-(4-piperidyl)-2-benzimidazolinone
- 53786-98-4
- 53788-49-1
- 53789-78-9
- 53790-47-9
- 53790-72-0
- 5379-16-8
- 537-91-7
- 53-79-2
- 537-92-8
- 53797-35-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View