-
Name
alclofenac epoxide
- EINECS
- CAS No. 70319-10-7
- Density 1.401g/cm3
- Solubility
- Melting Point
- Formula C11H11ClO4
- Boiling Point 411.2°Cat760mmHg
- Molecular Weight 242.67
- Flash Point 202.5°C
- Transport Information
- Appearance
- Safety Mutation data reported. When heated to decomposition it emits toxic fumes of Cl−.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms
- PSA 59.06000
- LogP 1.74470
Alclofenac epoxide Chemical Properties
Molecular Structure of Alclofenac epoxide (CAS NO.70319-10-7):
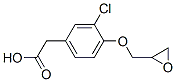
IUPAC: [3-Chloro-4-(oxiran-2-ylmethoxy)phenyl]acetic acid
Molecular Formula: C11H11ClO4
Molecular Weight: 242.6556
Density: 1.401 g/cm3
Boiling point: 411.2oC at 760 mmHg
Flash point: 202.5oC
Index of Refraction: 1.581
Surface Tension: 56.7 dyne/cm
Enthalpy of Vaporization: 69.97 kJ/mol
Vapour Pressure: 1.69E-07 mmHg at 25oC
SMILES: O=C(O)Cc2cc(Cl)c(OCC1OC1)cc2
InChI: InChI=1/C11H11ClO4/c12-9-3-7(4-11(13)14)1-2-10(9)16-6-8-5-15-8/h1-3,8H,4-6H2,(H,13,14)
InChIKey: FQBFPTMSLNQVNW-UHFFFAOYAA
Alclofenac epoxide Toxicity Data With Reference
| 1. | mmo-sat 100 µg/plate | JJATDK JAT, Journal of Applied Toxicology. 3 (1983),230. | ||
| 2. | otr-ham:emb 125 mg/L | CBINA8 Chemico-Biological Interactions. 34 (1981),95. |
Alclofenac epoxide Safety Profile
Mutation data reported. When heated to decomposition it emits toxic fumes of Cl−.
Alclofenac epoxide Specification
Alclofenac epoxide with cas registry number of 70319-10-7 is also known as (3-beta,5-alpha,15-beta)-19-Oxo-3,14,15-trihydroxycard-20(22)-enolide ; Alloglaucotoxigenin ; Alloglucotoxigenin ; 5-alpha-Card-20(22)-enolide, 3-beta,14,15-beta-trihydroxy-19-oxo- (8CI) ; Card-20(22)-enolide, 19-oxo-3,14,15-trihydroxy-, (3-beta,5-alpha,15-beta)- . Alclofenac epoxide was shown to inhibit the activity of yeast alcohol dehydrogenase and form a conjugate with cysteine. It showed mutagenic effects on strains of Salmonella typhimurium sensitive to alkylating agents, but had no effect on strains sensitive to intercalating agents. Alclofenac epoxide with cas registry number of 70319-10-7 is used as a intermediate.
Related Products
- Alclofenac epoxide
- 70321-63-0
- 70321-72-1
- 70321-75-4
- 7032-17-9
- 70321-85-6
- 70321-86-7
- 70322-21-3
- 703-23-1
- 70324-65-1
- 70331-82-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View