-
Name
Benzyl chloroformate
- EINECS 207-925-0
- CAS No. 501-53-1
- Article Data53
- CAS DataBase
- Density 1.244 g/cm3
- Solubility Soluble in most organic solvents; usually use in water, dichloromethane, THF, ether or toluene
- Melting Point -20 °C
- Formula C8H7ClO2
- Boiling Point 223.984 °C at 760 mmHg
- Molecular Weight 170.595
- Flash Point 73.488 °C
- Transport Information UN 2924 3/PG 2
- Appearance light yellow liquid
- Safety 53-26-36/37/39-45-60-61-62-46
- Risk Codes 45-20-34-48/22-50/53-67-65-63-48/20-11
-
Molecular Structure
-
Hazard Symbols
 T,
T,  C,
C,  N,
N,  F
F
- Synonyms Benzyl chlorocarbonate;Benzyloxycarbonyl chloride;Benzylcarbonyl chloride;Z-Cl;Benzyl carbonochloridate;Carbobenzoxy chloride;Chloroformic acid, benzyl ester;Formic acid, chloro-, benzyl ester;Carbonochloridic acid, phenylmethyl ester;Benzyl Chlorofomate;n-Amyl chloroformate,n-pentyl chloroformate;CBZ-CL;Benzyl chloroformate (CBZ-Cl);Carboobenzoxy Chloride;Cbz-Cl (Carboobenzoxy Chloride);
- PSA 26.30000
- LogP 2.56200
Synthetic route
-
-
22426-83-1
O-benzyl S-methyl carbonothioate

-
-
501-53-1
benzyl chloroformate

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride at 0 - 20℃; for 1h; | 100% |
| With sulfuryl dichloride at 20℃; for 1h; | 72% |
| With sulfuryl dichloride at 0 - 20℃; for 1h; | 72% |

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at -5 - 10℃; for 13h; Reagent/catalyst; Temperature; Solvent; | 99% |
| With pyridine In dichloromethane | 98% |
| With pyridine In dichloromethane | 98% |
-
-
110177-67-8
S-ethyl O-benzyl carbonothioate

-
-
501-53-1
benzyl chloroformate

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride at 20℃; for 1h; | 81% |
| With sulfuryl dichloride at 0 - 20℃; for 1h; | 81% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In tetrahydrofuran at 0 - 20℃; | 76% |

| Conditions | Yield |
|---|---|
| With toluene anfangs unter Kuehlung; | |
| at -8℃; | |
| With 2,3-Dimethylaniline |
-
-
39608-52-1
benzyl carbonochloridate

-
-
501-53-1
benzyl chloroformate

| Conditions | Yield |
|---|---|
| With thionyl chloride at 90℃; for 3h; | |
| With thionyl chloride for 6h; Chlorination; Heating; | |
| With thionyl chloride for 4h; Reflux; | |
| With thionyl chloride at 90℃; for 2h; |

| Conditions | Yield |
|---|---|
| With pyrographite In tetrahydrofuran at 20℃; for 1h; Condensation; | |
| With dmap In toluene at 20℃; for 24h; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: sulfur; DBU / dimethylsulfoxide / 5 h / 20 °C / 760 Torr 1.2: 67 percent / dimethylsulfoxide / 1 h / 20 °C 2.1: 100 percent / SO2Cl2 / 1 h / 0 - 20 °C View Scheme |

-
-
125178-42-9
Thiocarbonic acid O-benzyl ester

-
-
501-53-1
benzyl chloroformate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrahydrofuran / 2 h / 20 °C / 750.06 Torr 2: 81 percent / sulfuryl chloride / 1 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 1.62 g / tetrahydrofuran / 16 h / 20 °C / 750.06 Torr 2: 72 percent / sulfuryl chloride / 1 h / 0 - 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: DBU / tetrahydrofuran / 1 h / 20 °C / 750.06 Torr 2: tetrahydrofuran / 2 h / 20 °C / 750.06 Torr 3: 81 percent / sulfuryl chloride / 1 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: DBU / tetrahydrofuran / 1 h / 20 °C / 750.06 Torr 2: 1.62 g / tetrahydrofuran / 16 h / 20 °C / 750.06 Torr 3: 72 percent / sulfuryl chloride / 1 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: sulfur; DBU / tetrahydrofuran / 6 h / 80 °C / 7500.6 Torr 2: tetrahydrofuran / 2 h / 20 °C / 750.06 Torr 3: 81 percent / sulfuryl chloride / 1 h / 0 - 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane |
-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
100-53-8
phenylmethanethiol

-
-
598-74-3
2-amino-3-methylbutane

-
-
501-53-1
benzyl chloroformate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane |

-
-
74137-84-1
9,10-dihydro-3-methyl-4H-thieno[3,4-b][1,5]benzodiazepin-10-one

-
B
-
501-53-1
benzyl chloroformate
-
-
1449331-28-5
benzyl 2-oxo-2-(pyren-1-yl)ethyl carbonate

-
-
501-53-1
benzyl chloroformate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: water / acetonitrile / pH 7.5 / Photolysis; Inert atmosphere 2: sodium carbonate / toluene / 6 h / 0 °C View Scheme |

| Conditions | Yield |
|---|---|
| With oxygen at 20℃; for 3h; Irradiation; |
-
-
61-54-1
tryptamine

-
-
501-53-1
benzyl chloroformate

-
-
38750-13-9
(2-indol-3-yl-ethyl)-carbamic acid benzyl ester

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In chloroform at 20℃; for 1h; | 100% |
| With N-ethyl-N,N-diisopropylamine In chloroform at 0 - 20℃; for 1h; | 100% |
| With sodium hydroxide In tetrahydrofuran | 97% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; for 1h; | 100% |
| In benzene for 0.5h; Ambient temperature; | 95% |
| With triethylamine In dichloromethane at 20℃; for 6h; Large scale; | 95.3% |
-
-
107-09-5
2-bromoethylamine

-
-
501-53-1
benzyl chloroformate

-
-
53844-02-3
benzyl N-(2-bromoethyl)carbamate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; for 6h; | 100% |
| With sodium hydroxide | |
| In sodium hydroxide; acetone | |
| With triethylamine In N,N-dimethyl-formamide at 0℃; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 0 - 20℃; | 100% |
| With sodium hydroxide In water at 0 - 20℃; for 5h; | 99% |
| With sodium carbonate In water at 0 - 20℃; for 24h; | 98% |
-
-
2835-82-7
3-aminobutyric acid

-
-
501-53-1
benzyl chloroformate

-
-
51440-81-4
3-benzyloxycarbonylaminobutyric acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; | 100% |
| With sodium hydroxide In tetrahydrofuran for 16h; Ambient temperature; | 97% |
| With sodium hydroxide In water; acetone for 1h; Ambient temperature; | 91% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water Inert atmosphere; | 100% |
| With sodium hydroxide In water at 0 - 20℃; | 100% |
| With potassium carbonate In water at 0 - 2℃; pH=1.5 - Ca. 2; Reagent/catalyst; pH-value; Temperature; Solvent; | 99% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In diethyl ether; water at 0 - 20℃; for 6h; | 100% |
| With sodium hydroxide In water at 0℃; for 24h; | 94% |
| With sodium hydrogencarbonate for 4h; Ambient temperature; | 93% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 0 - 20℃; | 100% |
| With sodium hydroxide at 0℃; for 1h; | 99% |
| With sodium hydroxide In water at 20℃; for 2h; | 92% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In diethyl ether for 5h; | 100% |
| In ethyl acetate Heating; | 31% |
| With sodium hydroxide at 0℃; | |
| Schotten-Baumann reaction; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide a) 0 deg C, 30 min, b) 20 deg C, 2 h; | 100% |
| With sodium hydroxide In water for 0.5h; | 100% |
| With sodium hydroxide In 1,4-dioxane; water at 20℃; for 1h; | 82% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 0 - 20℃; | 100% |
| Stage #1: L-phenylalanine; benzyl chloroformate With sodium hydroxide In water at 0 - 20℃; for 1.5h; Stage #2: With hydrogenchloride In water pH=4; | 99% |
| With sodium hydroxide | 99% |
-
-
1080-06-4
L-Tyr-OMe

-
-
501-53-1
benzyl chloroformate

-
-
13512-31-7
N-(benzyloxycarbonyl)-L-tyrosine methyl ester

| Conditions | Yield |
|---|---|
| With sodium carbonate In water; acetone at 20℃; for 2h; | 100% |
| With sodium carbonate In dichloromethane; water at 0 - 20℃; for 2.5h; | 98% |
| With sodium hydrogencarbonate In chloroform at 25℃; for 3h; | 94% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In tetrahydrofuran at 0 - 20℃; | 100% |
| With sodium hydrogencarbonate In tetrahydrofuran at 0 - 20℃; | 100% |
| With sodium hydrogencarbonate In tetrahydrofuran; water at 0℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 0 - 20℃; | 100% |
| With potassium carbonate In tetrahydrofuran; water at 20℃; for 12h; | 96% |
| Stage #1: glycine With sodium hydroxide In water Inert atmosphere; Stage #2: benzyl chloroformate In water at 0℃; for 0.833333h; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water for 13.5h; Ambient temperature; | 100% |
| With sodium carbonate at 0 - 20℃; for 3h; | 100% |
| With sodium hydroxide at 0℃; for 0.166667h; | 97% |
-
-
501-53-1
benzyl chloroformate

-
-
2584-71-6
cis-hydroxy-D-proline

-
-
130930-25-5
(R,R)-4-hydroxy-1-(benzyloxycarbonyl)pyrrolidine-2-carboxylic acid

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water; toluene at 20℃; for 16.25h; | 100% |
| With sodium hydrogencarbonate In 1,4-dioxane; water | 100% |
| With sodium hydrogencarbonate In water; toluene for 16.5h; Ambient temperature; | 96% |
-
-
501-53-1
benzyl chloroformate

-
-
51-35-4
4R-4-hydroxyproline

-
-
13504-85-3
(2S,4R)-1-(benzyloxycarbonyl)-4-hydroxypyrrolidine-2-carboxylic acid

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In diethyl ether Acylation; | 100% |
| With sodium hydroxide at 0 - 20℃; for 5h; | 100% |
| With sodium hydrogencarbonate In water; toluene at 20℃; for 12.25h; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In tetrahydrofuran; water at 20℃; for 4h; Inert atmosphere; Cooling with ice; | 100% |
| With N-ethyl-N,N-diisopropylamine | 96% |
| With sodium hydroxide In tetrahydrofuran; water at 20℃; for 12h; Cooling with ice; | 95% |

| Conditions | Yield |
|---|---|
| With sodium carbonate In 1,4-dioxane; water at 20℃; for 20h; | 100% |
| With sodium carbonate In 1,4-dioxane; water at 0 - 20℃; for 18h; | 97% |
| With hydrogenchloride; sodium hydroxide In 1,4-dioxane; toluene at 0 - 20℃; | 95% |

| Conditions | Yield |
|---|---|
| With sodium carbonate In 1,4-dioxane; water at 20℃; | 100% |
| With sodium carbonate In 1,4-dioxane; water 1.) 0 deg C, 1.5 h, 2.) room temperature, 11 h; | 99% |
| With sodium hydroxide In water at 0 - 20℃; for 16h; | 94% |
-
-
20859-02-3
L-tert-Leucine

-
-
501-53-1
benzyl chloroformate

-
-
62965-10-0
(S)-N-carbobenzoxy-tert-butylleucine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 1,4-dioxane at 0℃; for 18h; | 100% |
| With sodium hydroxide In water for 3h; Inert atmosphere; | 100% |
| With sodium hydroxide In water at 5 - 20℃; for 14h; pH=10 - 10.5; Product distribution / selectivity; | 97% |
-
-
21075-83-2
1-methyl-1-tert-butoxycarbonylhydrazine

-
-
501-53-1
benzyl chloroformate

-
-
57699-92-0
2-benzyl 1-tert-butyl 1-methylhydrazine-1,2-dicarboxylate

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In ethyl acetate at 0 - 1℃; for 1h; | 100% |
| With sodium hydroxide In dichloromethane; water at 20℃; for 2h; | 81% |
| With sodium hydroxide In 1,4-dioxane; water at 25℃; for 1h; | 69.54% |
-
-
5680-80-8
methyl (2S)-2-amino-3-hydroxypropanoate hydrochloride

-
-
501-53-1
benzyl chloroformate

-
-
1676-81-9
N-benzyloxycarbonyl-L-serine methyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran; water 0 deg C -> room temperature, 4 h; | 100% |
| With sodium hydrogencarbonate In dichloromethane; water at 0 - 20℃; for 6h; | 97% |
| With sodium hydrogencarbonate | 93% |
-
-
5619-04-5
methyl 2-amino-3-hydroxypropanoate hydrochloride

-
-
501-53-1
benzyl chloroformate

-
-
14464-15-4
methyl 2-(benzyloxycarbonylamino)-3-hydroxypropanoate

| Conditions | Yield |
|---|---|
| With sodium carbonate In tetrahydrofuran at 20℃; for 12h; | 100% |
| Stage #1: methyl 2-amino-3-hydroxypropanoate hydrochloride; benzyl chloroformate With sodium hydrogencarbonate In water at 0 - 20℃; for 24.75h; Inert atmosphere; Stage #2: With hydrogenchloride In water pH=1; | 75% |
| With triethylamine In dichloromethane for 5h; Ambient temperature; | 70% |
-
-
501-53-1
benzyl chloroformate

-
-
107-15-3
ethylenediamine

-
-
18807-67-5
1,2-Ethanediylbis(phenylmethyl carbamate)

| Conditions | Yield |
|---|---|
| With potassium hydroxide | 100% |
| With sodium hydroxide |
-
-
501-53-1
benzyl chloroformate

-
-
78-96-6, 1674-56-2
3-amino-2-propanol

-
-
65935-10-6
1-Benzyloxycarbonylamino-2,4-propanediol

| Conditions | Yield |
|---|---|
| With sodium carbonate In water at 0℃; for 3h; | 100% |
| With sodium carbonate In water at 0℃; for 3h; | 100% |
| With sodium carbonate In water at 0℃; for 3h; | 96% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at -65℃; for 1h; | 100% |
| With sodium tetrahydroborate In methanol at -75℃; for 3h; | 96% |
| With sodium tetrahydroborate In ethanol at -78℃; for 1.5h; | 74% |
-
-
694-05-3
1,2,3,6-tetrahydropyridine

-
-
501-53-1
benzyl chloroformate

-
-
66207-23-6
1-benzyloxycarbonyl-1,2,3,6-tetrahydropyridine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 16h; | 100% |
| With sodium carbonate at 0℃; for 2h; | 99% |
| With sodium carbonate In water at 5 - 20℃; Cooling with ice; | 99% |
-
-
616-45-5
2-pyrrolidinon

-
-
501-53-1
benzyl chloroformate

-
-
14468-80-5
N-benzyloxycarbonylpyrrolidin-2-one

| Conditions | Yield |
|---|---|
| Stage #1: 2-pyrrolidinon With n-butyllithium In tetrahydrofuran Stage #2: benzyl chloroformate In tetrahydrofuran | 100% |
| Stage #1: 2-pyrrolidinon With sodium hydroxide In toluene for 6h; Dean-Stark; Reflux; Stage #2: benzyl chloroformate In toluene at 5 - 12℃; for 2h; | 95.2% |
| Stage #1: 2-pyrrolidinon With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; for 0.5h; Inert atmosphere; Stage #2: benzyl chloroformate In tetrahydrofuran at -78 - -40℃; for 3h; Inert atmosphere; | 91% |
-
-
498-94-2
isonipecotic acid

-
-
501-53-1
benzyl chloroformate

-
-
10314-98-4
1-benzyloxycarbonylpiperidine-4-carboxylic acid

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; sodium carbonate In water; acetonitrile at 0 - 20℃; for 2h; pH=10 - 11; | 100% |
| With potassium carbonate In water at 22℃; for 58h; | 97% |
| With sodium hydrogencarbonate In tetrahydrofuran; water at 0 - 20℃; | 96% |
Benzyl chloroformate Specification
The Benzyl chloroformate, with the CAS registry number 501-53-1, is also known as Carbonochloridic acid, phenylmethyl ester. It belongs to the product categories of Pharmaceutical Intermediates; Benzene derivatives; Amino Acid Derivatives; Aromatic Esters; Chloroformates; N-Protecting Reagents; Biochemistry; Peptide Synthesis; Protection & Derivatization Reagents (for Synthesis); Protective Reagents (Peptide Synthesis); Synthetic Organic Chemistry; Cbz-Amino acid series. Its EINECS number is 207-925-0. This chemical's molecular formula is C8H7ClO2 and molecular weight is 170.59. What's more, its systematic name is Benzyl carbonochloridate. It should be sealed and stored in a ventilated and dry place. Its storage temperature is 2 - 8°C. This chemical is used in organic synthesis for the introduction of the carboxybenzyl (Cbz,Z) protecting group for amines. It is also a carcinogen.
Physical properties of Benzyl chloroformate are:
(1)ACD/LogP: 1.862; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.86; (4)ACD/LogD (pH 7.4): 1.86; (5)ACD/BCF (pH 5.5): 15.32; (6)ACD/BCF (pH 7.4): 15.32; (7)ACD/KOC (pH 5.5): 245.54; (8)ACD/KOC (pH 7.4): 245.54; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 26.3 Å2; (13)Index of Refraction: 1.531; (14)Molar Refractivity: 42.426 cm3; (15)Molar Volume: 137.118 cm3; (16)Polarizability: 16.819×10-24cm3; (17)Surface Tension: 41.04 dyne/cm; (18)Density: 1.244 g/cm3; (19)Flash Point: 73.488 °C; (20)Enthalpy of Vaporization: 46.048 kJ/mol; (21)Boiling Point: 223.984 °C at 760 mmHg; (22)Vapour Pressure: 0.093 mmHg at 25°C.
Preparation of Benzyl chloroformate:
This chemical can be prepared by phosgene and benzyl alcohol. The yield is about 91-94%. It can also be prepared by benzyl alcohol and trichloromethyl chloroformate. In laboratory, it can be prepared by benzyl alcohol and phosgene in the present of toluene.
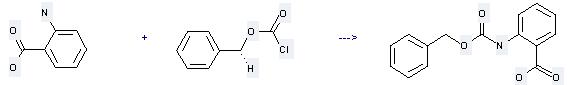
Uses of Benzyl chloroformate:
It can be used to produce N-benzyloxycarbonyl-anthranilic acid at the ambient temperature. It will need reagent 1N NaOH and solvent dioxane. The yield is about 72%.
Safety information of Benzyl chloroformate:
When you are using Benzyl chloroformate, please be cautious about it as the following:This chemical is harmful as it has a danger of serious damage to health by prolonged exposure through inhalation or if swallowed. It is very toxic to aquatic organisms as it may cause long-term adverse effects in the aquatic environmen. Moreover, it may cause cancer. This substance is harmful by inhalation and is harmful as it may cause lung damage if swallowed. If swallowed, it will not induce vomiting, but you should seek medical advice immediately and show this container or label. It can cause burns and is highly flammable. It has a possible risk of harm to the unborn child. Its Vapours may cause drowsiness and dizziness. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need to wear suitable protective clothing, gloves and eye/face protection. In case of accident or if you feel unwell, you should seek medical advice immediately (show the label where possible). It must be avoided exposure, and you need to obtain special instructions before use. This material and its container must be disposed of as hazardous waste. You should avoid releasing to the environment, and you need to refer to special instructions/safety data sheet.
You can still convert the following datas into molecular structure:
(1)SMILES: ClC(=O)OCc1ccccc1
(2)Std. InChI: InChI=1S/C8H7ClO2/c9-8(10)11-6-7-4-2-1-3-5-7/h1-5H,6H2
(3)Std. InChIKey: HSDAJNMJOMSNEV-UHFFFAOYSA-N
The toxicity data of Benzyl chloroformate is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LC50 | inhalation | 590mg/m3/4H (590mg/m3) | National Technical Information Service. Vol. OTS0539682, | |
| rat | LD50 | oral | 3gm/kg (3000mg/kg) | National Technical Information Service. Vol. OTS0539682, |
Related Products
- Benzyl (3R)-3-aminopiperidine-1-carboxylate
- Benzyl (3R)-piperidin-3-ylcarbamate
- Benzyl (3R,4R,5S)-3-hydroxy-4-(4-hydroxyphenyl)-5-(triisopropylsilanyloxy)piperidine-1-carboxylate
- Benzyl (3S)-1,2,3,4-tetrahydroisoquinoline-3-carboxylate
- Benzyl [(3S)-2,5-dioxopyrrolidin-3-yl]carbamate
- Benzyl [1-(hydroxymethyl)cyclopropyl]carbamate
- Benzyl [1-[4-[[(4-fluorobenzyl)amino]carbonyl]-5-hydroxy-1-methyl-6-oxo-1,6-dihydropyrimidin-2-yl]-1-methylethyl]carbamate
- Benzyl 1-(2-(dimethylamino)propyl)-pyrrol-2-yl, citrate ketone
- Benzyl 1-piperazinecarboxylate
- Benzyl 2,2,2-trichloroacetimidate
- 5015-38-3
- 501653-37-8
- 501-65-5
- 501684-22-6
- 501684-93-1
- 501698-31-3
- 501-75-7
- 50-18-0
- 501-81-5
- 5018-30-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View