-
Name
7,7-DICHLOROBICYCLO[3.2.0]HEPT-2-EN-6-ONE
- EINECS 226-165-0
- CAS No. 5307-99-3
- Article Data38
- CAS DataBase
- Density 1.44g/cm3
- Solubility
- Melting Point
- Formula C7H6Cl2O
- Boiling Point 274.8 °C at 760 mmHg
- Molecular Weight 177.03
- Flash Point 114.1 °C
- Transport Information
- Appearance clear brown liquid
- Safety 26-36/37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Dichlorobicycloheptenone;
- PSA 17.07000
- LogP 1.93530
Synthetic route
-
-
4342-33-0
tributyl(trichloroacetoxy)stannane

-
-
542-92-7
cyclopenta-1,3-diene

-
A
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
B
-
1461-22-9
tributyltin chloride

-
C
-
791-28-6
Triphenylphosphine oxide

| Conditions | Yield |
|---|---|
| With triphenylphosphine | A 30% B 90% C 92% |
| With P(C6H5)3 | A 30% B 90% C 92% |

-
-
79-36-7
dichloroacethyl chloride

-
-
542-92-7
cyclopenta-1,3-diene

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With triethylamine In pentane for 4h; Heating; | 88% |
| With triethylamine In hexane; n-heptane at 20℃; | 86% |
| With triethylamine In hexane at 20℃; | 85% |
-
-
20618-08-0
β,β-dichloroacryloyl chloride

-
-
542-92-7
cyclopenta-1,3-diene

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With triethylamine In hexane at 26 - 30℃; for 9h; | 85% |
-
-
4591-28-0
dichloroketene

-
-
542-92-7
cyclopenta-1,3-diene

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With triethylamine In pentane | 79% |
-
-
542-92-7
cyclopenta-1,3-diene

-
-
76-02-8
Trichloroacetyl chloride

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With zinc In diethyl ether at 0℃; for 1h; | 61% |
-
-
78133-61-6
7,7-dichloro-2,4-exo-bis(trimethylsilyl)bicyclo<3.2.0>hept-2-en-6-one

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With sulfuric acid In methanol for 18h; Ambient temperature; | 37% |
-
-
25436-07-1
trimethylsilyl trichloroacetate

-
-
542-92-7
cyclopenta-1,3-diene

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With triphenylphosphine |
-
-
33630-76-1
5,5-bis(trimethylsilyl)cyclopentadiene

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 72 percent / triethylamine / hexane / 1.) ice-cooling, 2 h, 2.) 0 - 5 deg C, 12 h 2: 37 percent / conc. H2SO4 / methanol / 18 h / Ambient temperature View Scheme |
-
-
77-73-6, 933-60-8, 1755-01-7
bi(cyclopentadiene)

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 200 °C 2: triethylamine / pentane View Scheme |

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Heating 2: triethylamine / n-heptane; hexane / 20 °C View Scheme |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
92998-64-6
7,7-dichloro-6-hydroxybicyclo<3.2.0>hept-2-ene

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol for 21h; Ambient temperature; | 96% |
| With lithium aluminium tetrahydride In diethyl ether for 1h; Ambient temperature; |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
110097-55-7, 110171-08-9
7,7-dichloro-6-hydroxybicyclo<3.2.0>hept-2-ene-6-d

| Conditions | Yield |
|---|---|
| With sodium borodeuteride In methanol for 15h; Ambient temperature; | 95% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
533-75-5
2-hydroxy-2,4,6-cycloheptatrien-1-one

| Conditions | Yield |
|---|---|
| With acetic acid; sodium hydroxide | 95% |
| With sodium hydroxide; acetic acid at 130℃; for 7h; Rearrangement; | 87% |
| With hydrogenchloride; sodium hydroxide; acetic acid for 8h; pH=1; Heating; | 77% |
| With sodium hydroxide; acetic acid for 8h; Heating; | 77% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
3886-69-9
(R)-1-phenyl-ethyl-amine

-
-
1192489-59-0
2-(dichloromethyl)-N-[(1R)-1-phenylethyl]cyclopent-3-ene-1-carboxamide

| Conditions | Yield |
|---|---|
| In benzene at 20℃; for 4h; | 95% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
925211-06-9, 925211-07-0
bicyclo(3.2.0)hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With zinc In acetic acid at 70℃; for 0.666667h; | 94% |
| With acetic acid; zinc at 70℃; for 48h; | 83% |
| With zinc In acetic acid | 81% |

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| at 120 - 130℃; for 6h; | 93% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| Stage #1: (+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one With sodium tetrahydroborate Stage #2: With ozone In methanol; dichloromethane at -78℃; for 0.166667h; Stage #3: With thiourea In methanol; dichloromethane at -78 - 20℃; Further stages.; | 93% |
-
-
64-17-5
ethanol

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
87274-25-7
(Z)-7,7-Dichloro-hepta-4,6-dienoic acid ethyl ester

| Conditions | Yield |
|---|---|
| In benzene Irradiation; irradiation with a Hanovia 450W high-pressure mercury arc lamp; | 90% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
38432-39-2
2-diethylamino-1,3,2-dioxaphosphorinane

| Conditions | Yield |
|---|---|
| at 120 - 130℃; for 6h; | 87% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
124-41-4
sodium methylate

-
-
412026-46-1
methyl 2-(dichloromethyl)cyclopent-3-ene-1-carboxylate

| Conditions | Yield |
|---|---|
| In methanol at -5 - 0℃; for 2h; | 83% |
-
-
1189-24-8
diisobutyl phosphite

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
128530-26-7
(7,7-Dichloro-6-hydroxy-bicyclo[3.2.0]hept-2-en-6-yl)-phosphonic acid diisobutyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In benzene Ambient temperature; | 70% |
-
-
1809-19-4
dibutyl hydrogen phosphite

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
128530-25-6
(7,7-Dichloro-6-hydroxy-bicyclo[3.2.0]hept-2-en-6-yl)-phosphonic acid dibutyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In benzene Ambient temperature; | 70% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
51592-73-5
7,7-dideuteriobicyclo<3.2.0>hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With deuteroacetic acid; zinc | 67% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
1809-21-8
di-n-propyl phosphonate

-
-
128530-23-4
(7,7-Dichloro-6-hydroxy-bicyclo[3.2.0]hept-2-en-6-yl)-phosphonic acid dipropyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In benzene Ambient temperature; | 67% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
19296-95-8, 19296-96-9, 92998-64-6, 101758-65-0, 130796-66-6, 130796-68-8
(+/-)-endo-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-ol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 20℃; for 1.5h; | 65% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
868-85-9
Dimethyl phosphite

-
-
128530-21-2
dimethyl 6-hydroxy-7,7-dichlorobicyclo<3.2.0>hept-2-en-6-ylphosphonate

| Conditions | Yield |
|---|---|
| With triethylamine In benzene for 5h; Ambient temperature; other dialkyl phosphites; | 62% |
| With triethylamine In benzene for 5h; Ambient temperature; | 62% |
-
-
31121-06-9
2-methoxy-[1,3,2]dioxaphosphinane

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| at 120 - 130℃; for 6h; | 61% |
-
-
1809-20-7
diisopropyl hydrogenphosphonate

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
128530-24-5
(7,7-Dichloro-6-hydroxy-bicyclo[3.2.0]hept-2-en-6-yl)-phosphonic acid diisopropyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In benzene Ambient temperature; | 60% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
762-04-9
phosphonic acid diethyl ester

-
-
128530-22-3
(7,7-Dichloro-6-hydroxy-bicyclo[3.2.0]hept-2-en-6-yl)-phosphonic acid diethyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In benzene Ambient temperature; | 60% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
33892-95-4
4-methyl-2-methoxy-1,3,2-dioxaphosphorinane

| Conditions | Yield |
|---|---|
| at 120 - 130℃; for 6h; | 60% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
30758-76-0
(+/-)-2-formylcyclopent-2-enecarboxylic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 60℃; for 1h; Solvent; Temperature; Concentration; Green chemistry; | 59.4% |
| Stage #1: (+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one With water; triethylamine In tert-butyl alcohol for 3h; Reflux; Solvolysis; Stage #2: With potassium carbonate In tert-butyl alcohol at 10℃; for 0.5h; | 52% |
| With sodium hydrogencarbonate at 75℃; for 5h; Hydrolysis; |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; sodium hydroxide In methanol; water at -5 - 30℃; | 57% |
-
-
16352-21-9
1,3,2-dioxaphosphinan-2-oxide

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With triethylamine In benzene for 12h; | 55% |
-
-
16368-20-0
4-methyl-[1,3,2]dioxaphosphinane 2-oxide

-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

| Conditions | Yield |
|---|---|
| With triethylamine In benzene for 12h; | 55% |
-
-
5307-99-3
(+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one

-
-
3886-69-9
(R)-1-phenyl-ethyl-amine

-
A
-
1040919-29-6
(3R,3aS,6aR)-3-hydroxy-2-[(1R)-1-phenylethyl]-3,3a,6,6a-tetrahydrocyclopenta[c]pyrrol-1(2H)-one

-
B
-
1040919-30-9
(3S,3aR,6aS)-3-hydroxy-2-[(1R)-1-phenylethyl]-3,3a,6,6a-tetrahydrocyclopenta[c]pyrrol-1(2H)-one

| Conditions | Yield |
|---|---|
| Stage #1: (+/-)-7,7-dichlorobicyclo[3.2.0]hept-2-en-6-one; (R)-1-phenyl-ethyl-amine In benzene at 20℃; for 4h; Stage #2: With barium(II) oxide In water; acetonitrile for 20h; Reflux; | A 47% B 46% |

Bicyclo[3.2.0]hept-2-en-6-one,7,7-dichloro- Specification
The Bicyclo[3.2.0]hept-2-en-6-one,7,7-dichloro-, with the CAS registry number 5307-99-3, is also known as Dichlorobicycloheptenone. Its EINECS registry number is 226-165-0. This chemical's molecular formula is C7H6Cl2O and molecular weight is 177.0279. What's more, its systematic name is called 7,7-Dichlorobicyclo[3.2.0]hept-2-en-6-one. Its appearance is clear brown liquid.
Physical properties about this chemical are: (1)ACD/LogP: 0.56; (2)#of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.56; (4)ACD/LogD (pH 7.4): 0.56; (5)ACD/BCF (pH 5.5): 1.56; (6)ACD/BCF (pH 7.4): 1.56; (7)ACD/KOC (pH 5.5): 47.86; (8)ACD/KOC (pH 7.4): 47.86; (9)#H bond acceptors: 1; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 17.07 Å2; (13)Index of Refraction: 1.568; (14)Molar Refractivity: 40.02 cm3; (15)Molar Volume: 122.2 cm3; (16)Surface Tension: 43.6 dyne/cm; (17)Density: 1.44 g/cm3; (18)Flash Point: 114.1 °C; (19)Enthalpy of Vaporization: 51.32 kJ/mol; (20)Boiling Point: 274.8 °C at 760 mmHg; (21)Vapour Pressure: 0.0053 mmHg at 25 °C.
Preparation of Bicyclo[3.2.0]hept-2-en-6-one,7,7-dichloro-: this chemical can be prepared by Cyclopenta-1,3-diene and 3,3-Dichloro-acryloyl chloride.
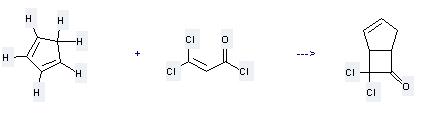
This reaction needs reagent Et3N and solvent Hexane at teperature of 26–30 °C. The reaction time is 9 hours. The yield is 85%.
Uses of Bicyclo[3.2.0]hept-2-en-6-one,7,7-dichloro-: it is used to produce other chemicals. For example, it is used to produce Bicyclo[3.2.0]hept-2-en-6-one.
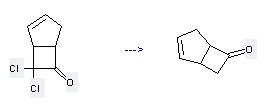
The reaction occurs with reagent Zn and solvent Acetic acid. The yield is 81%.
When you are dealing with this chemical, you should be very careful. This chemical is irritating to eyes, respiratory system and skin. Therefore, you should wear suitable protective clothing, gloves and eye/face protection. And in case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1) SMILES: ClC1(Cl)C(=O)C2C/C=C\C12
(2) InChI: InChI=1/C7H6Cl2O/c8-7(9)5-3-1-2-4(5)6(7)10/h1,3-5H,2H2
(3) InChIKey: JBPBARAOHIDZPU-UHFFFAOYAS
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View