-
Name
DIBENZO(A,I)PYRENE
- EINECS
- CAS No. 191-30-0
- Article Data12
- CAS DataBase
- Density 1.28g/cm3
- Solubility
- Melting Point 228.05°C
- Formula C24H14
- Boiling Point 630.6°C at 760 mmHg
- Molecular Weight 302.375
- Flash Point 349.8°C
- Transport Information
- Appearance
- Safety Confirmed carcinogen with experimental tumorigenic data. When heated to decomposition it emits acrid smoke and irritating fumes.
- Risk Codes 40
-
Molecular Structure
-
Hazard Symbols

- Synonyms 1,2,9,10-Dibenzopyrene;1,2:3,4-Dibenzopyrene; 2,3:4,5-Dibenzpyrene; 4,5,6,7-Dibenzpyrene; Ba51-090462; Dibenzo[a,l]pyrene; NSC 90324
- PSA 0.00000
- LogP 6.89040
Synthetic route

-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Stage #1: N-[2-(5,6-dihydro-4H-benz[de]anthracen-7-yl)benzylidene]aniline With potassium hydroxide In N,N-dimethyl-formamide at 100℃; for 1h; Stage #2: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In benzene for 0.5h; Heating; | 88% |
-
-
1013933-57-7
9-(2-methoxy-vinyl)-benzo[g]chrysene

-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| With methanesulfonic acid In dichloromethane at 20℃; for 2.5h; | 75% |
-
-
13322-54-8
Pentahelicen-3-carbonsaeure-methylester

-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| ueber Dibenzo<def,p>chrysen-8-ol; |

| Conditions | Yield |
|---|---|
| With aluminium trichloride; ortho-toluoyl chloride |
-
-
10383-87-6
1-Phenylbenzanthracen

-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| With aluminium trichloride; tin(IV) chloride |
-
-
17181-10-1
C24H14O

-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| With sodium chloride; zinc(II) chloride; zinc |

| Conditions | Yield |
|---|---|
| With aluminium trichloride |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: 86 percent / Br2 / CCl4 / 2 h / Heating 2.1: 61 percent / CsF / Pd(PPh3)4 / dimethylformamide / 24 h 3.1: 96 percent / molecular sieves 4 Angstroem / CH2Cl2 / 4 h 4.1: KOH / dimethylformamide / 1 h / 100 °C 4.2: 88 percent / DDQ / benzene / 0.5 h / Heating View Scheme |
-
-
681215-15-6
7-bromo-5,6-dihydro-4H-benz[de]anthracene

-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: 61 percent / CsF / Pd(PPh3)4 / dimethylformamide / 24 h 2.1: 96 percent / molecular sieves 4 Angstroem / CH2Cl2 / 4 h 3.1: KOH / dimethylformamide / 1 h / 100 °C 3.2: 88 percent / DDQ / benzene / 0.5 h / Heating View Scheme |
-
-
681215-16-7
7-(2-formylphenyl)-5,6-dihydro-4H-benz[de]anthracene

-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: 96 percent / molecular sieves 4 Angstroem / CH2Cl2 / 4 h 2.1: KOH / dimethylformamide / 1 h / 100 °C 2.2: 88 percent / DDQ / benzene / 0.5 h / Heating View Scheme |
-
-
29568-49-8
1-Phenyl-3,4-dihydro-benzo[a]anthracene

-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: DDQ 2: AlCl3, SnCl4 View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: (i), (ii) (heating) 2: DDQ 3: AlCl3, SnCl4 View Scheme |

| Conditions | Yield |
|---|---|
| In acetic acid; benzene at 40℃; for 4h; | 86% |
| In acetic acid; benzene at 40℃; Rate constant; Mechanism; |
-
-
191-30-0
Dibenzo[a,l]pyrene

-
-
226420-31-1
dibenzo[a,l]pyrene-8,9-quinone

| Conditions | Yield |
|---|---|
| With sodium periodate; rhodium(III) chloride hydrate In dichloromethane; water; acetonitrile at 30 - 40℃; | 58% |
| Multi-step reaction with 2 steps 1.1: OsO4 / pyridine / 144 h / 20 °C 1.2: NaHSO4 / 1 h / 20 °C 2.1: 2,3-dichloro-5,6-dicyano-p-benzoquinone / dioxane / 168 h / under argon atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: OsO4 / pyridine / 168 h / 25 °C 2: DDQ / dioxane / 15 h / Heating View Scheme |
-
-
191-30-0
Dibenzo[a,l]pyrene


| Conditions | Yield |
|---|---|
| With potassium perchlorate In N,N-dimethyl-formamide for 2h; Electrochemical reaction; | A 2% B 5% C 47% D 6% |
| With potassium perchlorate In dimethyl sulfoxide for 2h; Electrochemical reaction; | A 1% B 9% C 44% D 3% |


| Conditions | Yield |
|---|---|
| With potassium perchlorate In N,N-dimethyl-formamide for 2h; Electrochemical reaction; | A 3% B 25% C 26% |


| Conditions | Yield |
|---|---|
| With iodine; silver perchlorate In dimethyl sulfoxide Product distribution; Further Variations:; Reagents; Solvents; Iodine oxidation; nucleophilic substitution; | A 23 % Chromat. B 8 % Chromat. |


| Conditions | Yield |
|---|---|
| With iodine; silver perchlorate In N,N-dimethyl-formamide Product distribution; Further Variations:; Reagents; Solvents; Iodine oxidation; nucleophilic substitution; | 43 % Chromat. |
-
-
191-30-0
Dibenzo[a,l]pyrene


| Conditions | Yield |
|---|---|
| With iodine; silver perchlorate In N,N-dimethyl-formamide Product distribution; Further Variations:; Reagents; Solvents; Iodine oxidation; nucleophilic substitution; | A 1 % Chromat. B 10 % Chromat. C 47 % Chromat. |


| Conditions | Yield |
|---|---|
| With iodine; silver perchlorate In N,N-dimethyl-formamide Product distribution; Further Variations:; Reagents; Solvents; Iodine oxidation; nucleophilic substitution; | 30 % Chromat. |
-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| With osmium(VIII) oxide In pyridine at 25℃; for 168h; |
-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Stage #1: Dibenzo[a,l]pyrene With osmium(VIII) oxide In pyridine at 20℃; for 144h; Stage #2: With sodium hydrogen sulfate at 20℃; for 1h; |
-
-
191-30-0
Dibenzo[a,l]pyrene

-
-
93-61-8
N-methyl-N-phenylformamide

-
-
2869-59-2
dibenzo[def,p]chrysene-10-carboxaldehyde

| Conditions | Yield |
|---|---|
| With trichlorophosphate In 1,2-dichloro-benzene at 90℃; for 8h; Vilsmeier-Haack formylation; |
-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: OsO4 / pyridine / 144 h / 20 °C 1.2: NaHSO4 / 1 h / 20 °C 2.1: 2,3-dichloro-5,6-dicyano-p-benzoquinone / dioxane / 168 h / under argon atmosphere 3.1: 95 percent / NaBH4; O2 / ethanol; H2O / 72 h View Scheme | |
| Multi-step reaction with 3 steps 1: OsO4 / pyridine / 168 h / 25 °C 2: DDQ / dioxane / 15 h / Heating 3: 36 percent / NaBH4; air / propan-2-ol / 24 h View Scheme |
-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: OsO4 / pyridine / 144 h / 20 °C 1.2: NaHSO4 / 1 h / 20 °C 2.1: 2,3-dichloro-5,6-dicyano-p-benzoquinone / dioxane / 168 h / under argon atmosphere 3.1: 95 percent / NaBH4; O2 / ethanol; H2O / 72 h 4.1: 31 percent / pyridine / 24 h / 4 °C / under argon 5.1: 56 percent / sodium methoxide / tetrahydrofuran; methanol / 4 h / 20 °C View Scheme |
-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: OsO4 / pyridine / 144 h / 20 °C 1.2: NaHSO4 / 1 h / 20 °C 2.1: 2,3-dichloro-5,6-dicyano-p-benzoquinone / dioxane / 168 h / under argon atmosphere 3.1: 95 percent / NaBH4; O2 / ethanol; H2O / 72 h 4.1: 38 percent / pyridine / 24 h / 4 °C / under argon 5.1: 29 percent / sodium methoxide / tetrahydrofuran; methanol / 4 h / 20 °C View Scheme |
-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: OsO4 / pyridine / 144 h / 20 °C 1.2: NaHSO4 / 1 h / 20 °C 2.1: 2,3-dichloro-5,6-dicyano-p-benzoquinone / dioxane / 168 h / under argon atmosphere 3.1: 95 percent / NaBH4; O2 / ethanol; H2O / 72 h 4.1: 31 percent / pyridine / 24 h / 4 °C / under argon View Scheme |
-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: OsO4 / pyridine / 144 h / 20 °C 1.2: NaHSO4 / 1 h / 20 °C 2.1: 2,3-dichloro-5,6-dicyano-p-benzoquinone / dioxane / 168 h / under argon atmosphere 3.1: 95 percent / NaBH4; O2 / ethanol; H2O / 72 h 4.1: 38 percent / pyridine / 24 h / 4 °C / under argon View Scheme |
-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: OsO4 / pyridine / 168 h / 25 °C 2: DDQ / dioxane / 15 h / Heating 3: 36 percent / NaBH4; air / propan-2-ol / 24 h 4: tetrahydrofuran / (R,R)-Whelk-O15 chiral stationary phase column View Scheme |
-
-
191-30-0
Dibenzo[a,l]pyrene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: OsO4 / pyridine / 168 h / 25 °C 2: DDQ / dioxane / 15 h / Heating 3: 36 percent / NaBH4; air / propan-2-ol / 24 h 4: tetrahydrofuran / (R,R)-Whelk-O15 chiral stationary phase column View Scheme |
Dibenzo(def,p)chrysene Chemical Properties
Chemical Name: Dibenzo(a,i)pyrene
CAS No.: 191-30-0
EINECS: 205-886-4
Molecular Formula: C24H14
Molecular Weight: 302.37 g/mol
Density: 1.313 g/cm3
Flash Point: 282 °C
Boiling Point: 552.3 °C at 760 mmHg
Storage temp.: 2-8°C
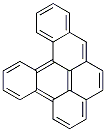
Following is the structure of Dibenzo(def,p)chrysene (191-30-0):
Product Categories about Dibenzo(def,p)chrysene (191-30-0) are Alpha Sort ; D ; DAlphabetic ; DIA - DIC ; Volatiles/ Semivolatiles
The chemical synonymous of Dibenzo(def,p)chrysene (191-30-0) are Dibenzo(A,I)Pyrene ; Dibenz(A,L)Pyrene ; 3,4,9,10-Dibenzopyrene ; 1,2,3,4-Dibenzpyrene ; 1,2,9,10-Dibenzopyrene
Dibenzo(def,p)chrysene Consensus Reports
NTP 10th Report on Carcinogens. IARC Cancer Review: Group 2B IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 7 (1987),p. 56.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Sufficient Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 32 (1983),p. 343.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Limited Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 3 (1973),p. 224.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) .
Dibenzo(def,p)chrysene Safety Profile
Confirmed carcinogen with experimental tumorigenic data. When heated to decomposition it emits acrid smoke and irritating fumes.
Hazard Codes:
Xn: Harmful ![]()
Risk Statements about Dibenzo(def,p)chrysene (191-30-0):
R40 Limited evidence of a carcinogenic effect.
Safety Statements about Dibenzo(def,p)chrysene (191-30-0):
S23 Do not breathe vapour.
S24/25: Avoid contact with skin and eyes.
S36/37: Wear suitable protective clothing and gloves.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View