-
Name
Dimethyldimethoxysilane
- EINECS 214-189-4
- CAS No. 1112-39-6
- Article Data63
- CAS DataBase
- Density 0.841 g/cm3
- Solubility decomposes in water
- Melting Point -80 °C
- Formula C4H12 O2Si
- Boiling Point 81.999 °C at 760 mmHg
- Molecular Weight 120.224
- Flash Point -34.808 °C
- Transport Information UN 1993 3/PG 2
- Appearance Colorless clear liquid
- Safety 16-26-33-36/37/39
- Risk Codes 11-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, F
F
- Synonyms AY 43-004;Dimethyldimethoxysilane;LS 520;NSC 93882;TSL 8112;TSL 8117;
- PSA 18.46000
- LogP 0.98100
Synthetic route

| Conditions | Yield |
|---|---|
| In methanol at 18 - 57℃; for 3.5h; | 99% |
| With diethyl ether |
-
-
67-56-1
methanol

-
-
77214-38-1
1-(methoxydimethylsilyl)-3-(3-methoxy-1,1,3,3-tetramethyldisilazanyl)-2,2,4,4-tetramethyldisilazane

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
13270-85-4
1,3-bis(methoxydimethylsilyl)-2,2,4,4-tetramethylcyclodisilazane

| Conditions | Yield |
|---|---|
| In hexane | A 90.5% B 96.5% |


| Conditions | Yield |
|---|---|
| In benzene-d6 | A n/a B 96% |

-
-
67-56-1
methanol

-
-
14850-05-6
1-(chlorodimethylsilyl)-3-(3-chloro-1,1,3,3-tetramethyl-1-disilazanyl)-2,2,4,4-tetramethylcyclodisilazane

-
-
1112-39-6
dimethyldimethoxysilan

| Conditions | Yield |
|---|---|
| for 0.5h; | 95% |
-
-
541-05-9
Hexamethylcyclotrisiloxane

-
-
616-38-6
carbonic acid dimethyl ester

-
-
1112-39-6
dimethyldimethoxysilan

| Conditions | Yield |
|---|---|
| With aluminum oxide; potassium fluoride at 400℃; | 94% |
-
-
67-56-1
methanol

-
-
2329-10-4
1,3-bis(chlorodimethylsilyl)-2,2,4,4-tetramethylcyclodisilazane

-
-
1112-39-6
dimethyldimethoxysilan

| Conditions | Yield |
|---|---|
| In hexane for 0.5h; | 92% |
-
-
7595-34-8
bis(ethylmercapto)dimethylsilane

-
-
122-51-0
orthoformic acid triethyl ester

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
6267-24-9
tris(ethylsulfanyl)methane

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In toluene at 0℃; for 5h; | A 80% B 90% |


| Conditions | Yield |
|---|---|
| With C6H5N(CH3)2; CH3OH | 90% |
| With methanol; N,N-dimethyl-aniline | 90% |
| With C6H5N(CH3)2; CH3OH | 40-50 |
-
-
1455-13-6
deuteromethanol

-
-
302841-08-3
2-methyl-3-triphenylphosphonio-2-silabutane-2-thiolate

-
A
-
1112-39-6
dimethyldimethoxysilan

| Conditions | Yield |
|---|---|
| Isomerization; Elimination; | A 80% B 30% C 40% D 30% |

-
-
1833-51-8
chloromethyldimethylphenylsilane

-
-
124-41-4
sodium methylate

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
17881-88-8
methoxydimethylphenylsilane

-
C
-
17876-91-4
dimethyl-phenyl-methoxymethylsilane

| Conditions | Yield |
|---|---|
| In methanol at 65℃; for 12h; Product distribution; variation of reactants, solvents, temperature and reaction time; | A 6% B 5% C 79% |
| In methanol at 65℃; for 12h; | A 6% B 5% C 79% |

-
-
16709-86-7
chloromethyldimethylvinylsilane

-
-
124-41-4
sodium methylate

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
16546-47-7
vinyldimethylmonomethoxysilane

-
C
-
30535-30-9
allyl(methoxy)dimethylsilane

-
D
-
98582-92-4
(methoxymethyl)dimethylvinylsilane

| Conditions | Yield |
|---|---|
| In methanol at 65℃; Yields of byproduct given; | A n/a B n/a C n/a D 76% |

-
-
1833-51-8
chloromethyldimethylphenylsilane

-
-
124-41-4
sodium methylate

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
36094-19-6
Benzyl-dimethyl-methoxysilan

-
C
-
17881-88-8
methoxydimethylphenylsilane

-
D
-
60592-88-3, 86962-11-0, 86962-12-1
ethyl(methoxy)methylphenylsilane

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 30℃; for 168h; | A 3% B 76% C 3% D 14% |
| In methanol at 30℃; for 168h; Product distribution; variation of reactants, solvents, temperature and reaction time; | A 3% B 76% C 3% D 14% |

-
-
1833-51-8
chloromethyldimethylphenylsilane

-
-
124-41-4
sodium methylate

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
17881-88-8
methoxydimethylphenylsilane

-
C
-
60592-88-3, 86962-11-0, 86962-12-1
ethyl(methoxy)methylphenylsilane

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether In 1,4-dioxane at 30℃; for 18h; | A 76% B 10% C 14% |
| In 1,4-dioxane at 100℃; for 12h; | A 44% B 12% C 32% |

-
-
67-56-1
methanol

-
-
14850-05-6
1-(chlorodimethylsilyl)-3-(3-chloro-1,1,3,3-tetramethyl-1-disilazanyl)-2,2,4,4-tetramethylcyclodisilazane

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
18790-13-1
Tris-(methoxy-dimethylsilyl)-amin

| Conditions | Yield |
|---|---|
| With triethylamine for 0.5h; | A 35% B 73% |

-
-
1066-35-9
dimethylmonochlorosilane

-
-
1067-52-3
tributyltin methoxide

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
1461-22-9
tributyltin chloride

-
C
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| 2:1; room temp.; | A 68% B n/a C n/a |

-
-
1833-51-8
chloromethyldimethylphenylsilane

-
-
13106-69-9
caesium methoxide

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
17881-88-8
methoxydimethylphenylsilane

-
C
-
17876-91-4
dimethyl-phenyl-methoxymethylsilane

| Conditions | Yield |
|---|---|
| In methanol at 65℃; for 2h; | A 66% B 20% C 8% |

-
-
1833-51-8
chloromethyldimethylphenylsilane

-
-
865-33-8
potassium methanolate

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
17881-88-8
methoxydimethylphenylsilane

-
C
-
17876-91-4
dimethyl-phenyl-methoxymethylsilane

| Conditions | Yield |
|---|---|
| In methanol at 65℃; for 12h; | A 46% B 14% C 28% |

-
-
16709-86-7
chloromethyldimethylvinylsilane

-
-
124-41-4
sodium methylate

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
16546-47-7
vinyldimethylmonomethoxysilane

-
C
-
30535-30-9
allyl(methoxy)dimethylsilane

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 100℃; | A 19% B 9% C 45% |
| In 1,4-dioxane at 100℃; Product distribution; other solvent, temperature and reactants; | A 19% B 9% C 45% |

-
-
10124-62-6
1,2-dimethoxy-1,1,2,2-tetramethyldisilane

-
-
78-79-5
isoprene

-
A
-
1112-39-6
dimethyldimethoxysilan

| Conditions | Yield |
|---|---|
| at 475℃; | A 39% B 31% |

-
-
675833-17-7
Cp*(OC)(pyridine)FeSiMe2OMe

-
A
-
1112-39-6
dimethyldimethoxysilan

| Conditions | Yield |
|---|---|
| In toluene heated at 90°C for 24 h; | A 24% B 18% |
-
-
462-95-3
formaldehyde diethyl acetal

-
-
7595-34-8
bis(ethylmercapto)dimethylsilane

-
A
-
54699-20-6
1-ethoxy-1-(ethylthio)methane

-
B
-
1112-39-6
dimethyldimethoxysilan

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In toluene at 0℃; for 5h; | A 12% B 7% |

-
-
109-87-5
Dimethoxymethane

-
-
7595-34-8
bis(ethylmercapto)dimethylsilane

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
77454-95-6
(ethylthio)methoxymethane

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In toluene at 0℃; for 5h; Product distribution; other temperature; | A 4% B 10% |
| With zinc(II) chloride In toluene at 0℃; for 5h; | A 4% B 10% |

-
-
681-84-5
tetramethylorthosilicate

-
-
676-58-4
methylmagnesium chloride

-
-
1112-39-6
dimethyldimethoxysilan

| Conditions | Yield |
|---|---|
| With diethyl ether |

| Conditions | Yield |
|---|---|
| With N,N-dimethyl-aniline | |
| With N,N-diethylaniline; Petroleum ether | |
| With aniline |
-
-
151237-85-3
methoxydimethylsilyl azide

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
4774-73-6
diazidodimethylsilane

| Conditions | Yield |
|---|---|
| With aluminium trichloride at 25℃; Equilibrium constant; Thermodynamic data; |
-
-
67-56-1
methanol

-
-
14850-05-6
1-(chlorodimethylsilyl)-3-(3-chloro-1,1,3,3-tetramethyl-1-disilazanyl)-2,2,4,4-tetramethylcyclodisilazane

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
18790-13-1
Tris-(methoxy-dimethylsilyl)-amin

-
C
-
77214-38-1
1-(methoxydimethylsilyl)-3-(3-methoxy-1,1,3,3-tetramethyldisilazanyl)-2,2,4,4-tetramethyldisilazane

| Conditions | Yield |
|---|---|
| With triethylamine In hexane for 0.5h; Product distribution; Heating; without triethylamine; |

-
-
67-56-1
methanol

-
-
13270-82-1
1,3-bis(aminodimethylsilyl)-2,2,4,4-tetramethylcyclodisilazane

-
A
-
1112-39-6
dimethyldimethoxysilan

-
B
-
18790-13-1
Tris-(methoxy-dimethylsilyl)-amin

-
C
-
13270-85-4
1,3-bis(methoxydimethylsilyl)-2,2,4,4-tetramethylcyclodisilazane

| Conditions | Yield |
|---|---|
| With triethylamine for 18h; Product distribution; Heating; other reaction time;; |

-
-
67-56-1
methanol

-
-
82881-21-8
1,3-Di-tert-butyl-2-(di-tert-butylamino)-4,4-dimethyl-1,3,2,4-diazaphosphasiletidin

-
A
-
21981-37-3
diisobutylamine

-
B
-
1112-39-6
dimethyldimethoxysilan

-
C
-
75-64-9
tert-butylamine

-
D
-
121-45-9
phosphorous acid trimethyl ester

| Conditions | Yield |
|---|---|
| for 1h; Product distribution; Ambient temperature; |

-
-
1719-57-9
Chloro(chloromethyl)dimethylsilane

-
-
124-41-4
sodium methylate

-
A
-
18143-33-4
(chloromethyl)methoxydimethylsilane

-
B
-
1112-39-6
dimethyldimethoxysilan

-
C
-
67965-21-3
methoxydimethyl(methoxymethyl)silane

| Conditions | Yield |
|---|---|
| at 140℃; Product distribution; Mechanism; var. temp., other alkali metals alkoxides, chloromethyldimethylhalosilanes, and chloromethyldimethylmethoxysilane; |

-
-
1112-39-6
dimethyldimethoxysilan

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
17980-39-1
1,1,3,3,5,5-hexamethyl-1,5-divinyltrisiloxane

| Conditions | Yield |
|---|---|
| With iron(III) chloride at 70 - 80℃; for 4h; Temperature; Reagent/catalyst; | 94.2% |
| With trifluorormethanesulfonic acid; acetic anhydride; acetic acid at 50℃; for 2.5h; | 51.7% |
-
-
1123-07-5
4,4,6-trimethyl-[1,3]dioxane

-
-
1112-39-6
dimethyldimethoxysilan

-
-
77181-39-6
2,2,4,4,6-Pentamethyl-1,3-dioxa-2-silacyclohexane

| Conditions | Yield |
|---|---|
| With tin(IV) chloride at 65℃; | 94% |
-
-
1112-39-6
dimethyldimethoxysilan

-
-
14879-83-5
2,2-dimethyl-[1,3,2]dioxasilinane

-
A
-
109-87-5
Dimethoxymethane

-
B
-
77181-39-6
2,2,4,4,6-Pentamethyl-1,3-dioxa-2-silacyclohexane

| Conditions | Yield |
|---|---|
| With tin(IV) chloride at 65℃; Product distribution; other reaction conditions; also with 1,3-dioxane and 4,4-dimethyl-1,3-dioxane; | A n/a B 94% |

-
-
930-36-9
1-methyl-1H-pyrazole

-
-
1112-39-6
dimethyldimethoxysilan

-
-
375-72-4
Nonafluorobutanesulfonyl fluoride

-
B
-
353-66-2
dimethyldifluorosilane

| Conditions | Yield |
|---|---|
| In diethyl ether 35°C , 24 h; | A 94% B n/a |
| In diethyl ether 35°C , 24 h; | A 94% B n/a |

-
-
616-47-7
1-methyl-1H-imidazole

-
-
1112-39-6
dimethyldimethoxysilan

-
-
375-72-4
Nonafluorobutanesulfonyl fluoride

| Conditions | Yield |
|---|---|
| In diethyl ether 20°C, slight warming; | 93.9% |
| In diethyl ether 20°C, slight warming; | 93.9% |

-
-
1112-39-6
dimethyldimethoxysilan

-
-
5301-78-0
4-(hydroxymethyl)-1-oxido-2,6,7-trioxa-1-phosphabicyclo [2.2.2] octane

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 100℃; for 8h; Solvent; Temperature; Inert atmosphere; | 93.6% |

| Conditions | Yield |
|---|---|
| With tris(pentafluorophenyl)borate In toluene | 92% |
-
-
1112-39-6
dimethyldimethoxysilan

-
-
35189-96-9
3-methyl-2-butenylmagnesium chloride

-
-
75732-21-7
(γ,γ-dimethylallyl)dimethylmethoxysilane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -20 - 20℃; for 19h; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| With iodine; magnesium; potassium iodide for 5h; Inert atmosphere; Reflux; | 91.51% |
-
-
1112-39-6
dimethyldimethoxysilan

-
-
107-05-1
3-chloroprop-1-ene

-
-
30535-30-9
allyl(methoxy)dimethylsilane

| Conditions | Yield |
|---|---|
| With iodine; magnesium; potassium iodide In acetyl chloride for 5h; Inert atmosphere; Reflux; | 91.51% |
-
-
1112-39-6
dimethyldimethoxysilan

-
-
768-87-6
4-hydroxymethyl-2,6,7-trioxa-1λ5-phosphabicyclo<2.2.2>octane-1-sulfide

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 100℃; for 6h; Temperature; Solvent; Inert atmosphere; | 91.2% |
-
-
103-83-3
N,N'-dimethylbenzylamine

-
-
1112-39-6
dimethyldimethoxysilan

-
-
375-72-4
Nonafluorobutanesulfonyl fluoride

-
-
25628-16-4
trimethylbenzylammoniumperfluorobutylsulfonate

| Conditions | Yield |
|---|---|
| In diethyl ether 20°C, slight warming, 20 h; | 90.2% |
| In diethyl ether 20°C, slight warming, 20 h; | 90.2% |

-
-
1112-39-6
dimethyldimethoxysilan

-
-
13315-17-8
4,4'-bis(dimethylsilyl)diphenyl ether

| Conditions | Yield |
|---|---|
| With tris(pentafluorophenyl)borate In toluene | 90% |
-
-
103-83-3
N,N'-dimethylbenzylamine

-
-
1112-39-6
dimethyldimethoxysilan

-
-
375-72-4
Nonafluorobutanesulfonyl fluoride

-
A
-
353-66-2
dimethyldifluorosilane

-
B
-
25628-16-4
trimethylbenzylammoniumperfluorobutylsulfonate

| Conditions | Yield |
|---|---|
| In diethyl ether 35°C , 20 h; | A n/a B 90% |
| In diethyl ether 35°C , 20 h; | A n/a B 90% |


| Conditions | Yield |
|---|---|
| Stage #1: N,N-dimethylaminomethylferrocene With tert.-butyl lithium In diethyl ether; pentane at 20℃; Inert atmosphere; Schlenk technique; Stage #2: dimethyldimethoxysilan In diethyl ether; pentane at -60 - 20℃; for 12h; Inert atmosphere; Schlenk technique; | 90% |
-
-
109-02-4
4-methyl-morpholine

-
-
1112-39-6
dimethyldimethoxysilan

-
-
375-72-4
Nonafluorobutanesulfonyl fluoride

-
-
25622-97-3
N,N-dimethylmorpholiniumperfluorobutylsulfonate

| Conditions | Yield |
|---|---|
| In diethyl ether 20°C, slight warming, 24 h; | 88.2% |
| In diethyl ether 20°C, slight warming, 24 h; | 88.2% |

-
-
1112-39-6
dimethyldimethoxysilan

-
-
1493-13-6
trifluorormethanesulfonic acid

-
-
27607-78-9
bis(trifluoromethanesulfonyloxy)dimethylsilane

| Conditions | Yield |
|---|---|
| at 0℃; | 88% |
-
-
120-94-5
1-Methylpyrrolidine

-
-
1112-39-6
dimethyldimethoxysilan

-
-
375-72-4
Nonafluorobutanesulfonyl fluoride

-
A
-
26601-00-3
N,N-dimethylpyrrolidiniumperfluorobutylsulfonate

-
B
-
353-66-2
dimethyldifluorosilane

| Conditions | Yield |
|---|---|
| In diethyl ether 35°C , 24 h; | A 88% B n/a |
| In diethyl ether 35°C , 24 h; | A 88% B n/a |

-
-
307-35-7
perfluorooctyl sulfofluorure

-
-
1112-39-6
dimethyldimethoxysilan

-
-
30427-51-1
diethyl-octadecyl-amine

-
-
25628-16-4
trimethylbenzylammoniumperfluorobutylsulfonate

| Conditions | Yield |
|---|---|
| In diethyl ether 20°C, slight warming; | 88% |
| In diethyl ether 20°C, slight warming; | 88% |

-
-
109-02-4
4-methyl-morpholine

-
-
1112-39-6
dimethyldimethoxysilan

-
-
375-72-4
Nonafluorobutanesulfonyl fluoride

-
A
-
25622-97-3
N,N-dimethylmorpholiniumperfluorobutylsulfonate

-
B
-
353-66-2
dimethyldifluorosilane

| Conditions | Yield |
|---|---|
| In diethyl ether 35°C , 24 h; | A 88% B n/a |
| In diethyl ether 35°C , 24 h; | A 88% B n/a |

-
-
1112-39-6
dimethyldimethoxysilan

-
-
927-77-5
n-propylmagnesium bromide

-
-
18182-14-4
methoxydimethyl(n-propyl)silane

| Conditions | Yield |
|---|---|
| In diethyl ether at 0 - 20℃; for 19h; Inert atmosphere; | 85% |
-
-
307-35-7
perfluorooctyl sulfofluorure

-
-
1112-39-6
dimethyldimethoxysilan

-
-
124-28-7
N,N-dimethyl-n-octadecylamine

-
-
25628-21-1
trimethylstearylammoniumperfluorooctylsulfonate

| Conditions | Yield |
|---|---|
| In diethyl ether 20°C, slight warming, 5 h; | 84.4% |
| In diethyl ether 20°C, slight warming, 5 h; | 84.4% |
| In diethyl ether |

Dimethoxydimethylsilane Specification
Dimethoxydimethylsilane, with the CAS registry number 1112-39-6, is also named as Silane,dimethoxydimethyl-. It belongs to the product categories of Dialkoxysilanes; Functional Materials; Si (Classes of Silicon Compounds); Silane Coupling Agents; Silane Coupling Agents (Intermediates); Si-O Compounds; Alkoxy Silanes; Crosslinkers; Crosslinking Agents; Di-Alkoxy Silanes; Silane Coupling Agents/Adhesion Promoters; Chemical Synthesis; Contact Printing; Materials Science; Micro/NanoElectronics; Organometallic Reagents; Organosilicon. Its EINECS number is 214-189-4. This chemical's molecular formula is C4H12O2Si and molecular weight is 120.22. It is used as constitution controller and chain extender agents. This chemical is stable at common pressure and temperature, and it should be sealed and stored in containers with dry inert gas which are placed in cool, ventilated and dry places. Moreover, it should be protected from oxides and water.
Physical properties of Dimethoxydimethylsilane are: (1)ACD/LogP: -0.409; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.41; (4)ACD/LogD (pH 7.4): -0.41; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 14.27; (8)ACD/KOC (pH 7.4): 14.27; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 18.46 Å2; (13)Index of Refraction: 1.376; (14)Molar Refractivity: 32.788 cm3; (15)Molar Volume: 143.024 cm3; (16)Polarizability: 12.998×10-24cm3; (17)Surface Tension: 16.9 dyne/cm; (18)Density: 0.841 g/cm3; (19)Flash Point: -34.808 °C; (20)Enthalpy of Vaporization: 30.923 kJ/mol; (21)Boiling Point: 81.999 °C at 760 mmHg; (22)Vapour Pressure: 89.0 mmHg at 25°C.
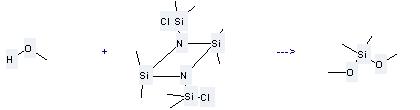
Preparation of Dimethoxydimethylsilane: this chemical can be prepared by N,N'-Bis-(chlordimethylsilyl)-tetramethyl-cyclodisilazan and methanol. This reaction will need solvent hexane with the reaction time of 0.5 hour. The yield is about 92%.
Uses of Dimethoxydimethylsilane: it can be used to produce 2,2-dimethyl-[1,3,6,2]dioxazasilocane by heating. It will need reagent Na and solvent benzene. The yield is about 52%.
![Dimethoxydimethylsilane can be used to produce 2,2-dimethyl-[1,3,6,2]dioxazasilocane by heating](/UserFilesUpload/Uses of Dimethoxydimethylsilane.jpeg)
When you are using this chemical, please be cautious about it as the following:
This chemical is flammable, so you should keep it away from sources of ignition - No smoking. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. You need to take precautionary measures against static discharges. When using it, you need wear suitable protective clothing, gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)SMILES: O(C)[Si](OC)(C)C
(2)Std. InChI: InChI=1S/C4H12O2Si/c1-5-7(3,4)6-2/h1-4H3
(3)Std. InChIKey: JJQZDUKDJDQPMQ-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LC | inhalation | > 21350mg/m3/4 (21350mg/m3) | SENSE ORGANS AND SPECIAL SENSES: LACRIMATION: EYE LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES BEHAVIORAL: EXCITEMENT | National Technical Information Service. Vol. OTS0539962, |
Related Products
- Dimethoxydimethylsilane
- 111-24-0
- 111247-60-0
- 111247-61-1
- 1112-48-7
- 111-25-1
- 111252-36-9
- 1112-54-5
- 1112-55-6
- 1112-56-7
- 111258-23-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View