-
Name
N-[3-(DIMETHYLAMINO)PROPYL]LAURAMIDE 9&
- EINECS 221-661-3
- CAS No. 3179-80-4
- Article Data25
- CAS DataBase
- Density 0.883 g/cm3
- Solubility
- Melting Point 34.5-38.5 °C(lit.)
- Formula C17H36N2O
- Boiling Point 418.9 °C at 760 mmHg
- Molecular Weight 284.486
- Flash Point 207.1 °C
- Transport Information
- Appearance
- Safety 26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms (Laurylamidopropyl)dimethylamine;3-Dodecanamidopropyldimethylamine;Lexamine L 13;N-[3-(Dimethylamino)propyl]lauramide;N',N'-Dimethyl-N-dodecanoyl-1,3-diaminopropane;Schercodine L;
- PSA 32.34000
- LogP 4.36610
Synthetic route
-
-
143-07-7
lauric acid

-
-
109-55-7
1-amino-3-(dimethylamino)propane

-
-
3179-80-4
lauric acid dimethylaminopropylamide

| Conditions | Yield |
|---|---|
| With potassium hydroxide; sodium hydroxide In water at 200℃; under 75.0075 Torr; for 1h; Reagent/catalyst; Temperature; | 98.6% |
| With toluene-4-sulfonic acid In 5,5-dimethyl-1,3-cyclohexadiene at 137℃; Dean-Stark; | 92% |
| With toluene-4-sulfonic acid In 5,5-dimethyl-1,3-cyclohexadiene at 137℃; for 24h; | 92% |
-
-
112-16-3
n-dodecanoyl chloride

-
-
109-55-7
1-amino-3-(dimethylamino)propane

-
-
3179-80-4
lauric acid dimethylaminopropylamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; | 93% |
| With triethylamine In dichloromethane at 20℃; | 93% |
| In chloroform at 20℃; for 24h; | 90% |
-
-
143-07-7
lauric acid

-
-
110-95-2
N,N,N'N'-tetramethyl-1,3-propanediamine

-
-
3179-80-4
lauric acid dimethylaminopropylamide

| Conditions | Yield |
|---|---|
| In toluene Inert atmosphere; Darkness; | 89% |
-
-
111-82-0
methyl n-dodecanoate

-
-
109-55-7
1-amino-3-(dimethylamino)propane

-
B
-
3179-80-4
lauric acid dimethylaminopropylamide

| Conditions | Yield |
|---|---|
| at 160 - 200℃; for 9 - 11.8h; Product distribution / selectivity; |

-
-
111-82-0
methyl n-dodecanoate

-
-
109-55-7
1-amino-3-(dimethylamino)propane

-
-
3179-80-4
lauric acid dimethylaminopropylamide

| Conditions | Yield |
|---|---|
| at 120 - 130℃; for 24h; |
-
-
3179-80-4
lauric acid dimethylaminopropylamide

-
-
61792-31-2
3-dodecanamido-N,N-dimethylpropan-1-amine oxide

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In ethanol at 50℃; for 24h; | 97% |
| With dihydrogen peroxide In water at 75 - 85℃; for 7h; Product distribution / selectivity; |
-
-
74-96-4
ethyl bromide

-
-
3179-80-4
lauric acid dimethylaminopropylamide

-
-
94036-25-6
(3-Dodecanoylamino-propyl)-ethyl-dimethyl-ammonium; bromide

| Conditions | Yield |
|---|---|
| In isopropyl alcohol for 8h; Heating; | 95% |
| In acetone Inert atmosphere; Reflux; | 78% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In isopropyl alcohol at 80℃; for 24h; | 93.5% |
| In ethanol at 80℃; for 48h; | 88% |

| Conditions | Yield |
|---|---|
| Stage #1: lauric acid dimethylaminopropylamide; 1.3-chlorobromopropane With potassium hydroxide In acetonitrile at 75℃; for 12h; pH=9.6; Stage #2: With sodium diselenide In water-d2 at 70℃; for 18h; | 91% |
-
-
3179-80-4
lauric acid dimethylaminopropylamide

-
-
100-44-7
benzyl chloride

-
-
52513-11-8
N-(3-(dimethylbenzylammonium)propyl)lauramide chloride

| Conditions | Yield |
|---|---|
| In isopropyl alcohol for 8h; Heating; | 90% |
| In ethanol at 78℃; Reflux; | |
| In ethanol at 78℃; Reflux; |
-
-
3179-80-4
lauric acid dimethylaminopropylamide

-
-
616-38-6
carbonic acid dimethyl ester

-
-
1380723-45-4
laurylamidopropyltrimethylammonium methyl carbonate

| Conditions | Yield |
|---|---|
| In methanol at 150℃; under 10501.1 Torr; for 10h; | 90% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 80℃; for 12h; | 80% |
| In isopropyl alcohol for 12h; Solvent; Reflux; | 70% |
-
-
3179-80-4
lauric acid dimethylaminopropylamide

-
-
1433993-67-9
4-(hydroxymethyl)-1,3,2-dioxathiolane-2,2-dioxide

-
-
1434911-77-9
C20H42N2O6S

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20 - 34℃; for 0.833333h; | 78% |
| In tetrahydrofuran Reflux; | |
| In tetrahydrofuran Reflux; |

| Conditions | Yield |
|---|---|
| In acetone for 65h; Inert atmosphere; Reflux; | 74% |
| In ethanol Reflux; |

| Conditions | Yield |
|---|---|
| In acetone for 68h; Inert atmosphere; Reflux; | 72% |

| Conditions | Yield |
|---|---|
| In ethanol at 80℃; for 48h; | 71% |

| Conditions | Yield |
|---|---|
| In ethanol at 80℃; for 50h; | 68.2% |

| Conditions | Yield |
|---|---|
| With sodium carbonate In xylenes for 72h; Heating / reflux; |
-
-
3179-80-4
lauric acid dimethylaminopropylamide

-
-
3926-62-3
sodium monochloroacetic acid

-
-
934425-71-5
laurylamidopropyl-β-alaninebetaine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 85 - 90℃; for 5h; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 98℃; for 5h; pH=7.5; | |
| With sodium hydroxide In water at 98℃; for 5h; pH=7.5; |
-
-
3179-80-4
lauric acid dimethylaminopropylamide

-
-
74-88-4
methyl iodide

-
-
206988-59-2
N,N,N-trimethyl-N-(3-dodecylamidopropyl) ammonium iodide

| Conditions | Yield |
|---|---|
| In diethyl ether at 20℃; for 24h; |

| Conditions | Yield |
|---|---|
| In ethyl acetate at 80℃; |
-
-
3179-80-4
lauric acid dimethylaminopropylamide

-
-
1380723-51-2
laurylamidopropyltrimethylammonium formate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: methanol / 10 h / 150 °C / 10501.1 Torr 2: methanol / 4 h / 35 °C View Scheme |
-
-
3179-80-4
lauric acid dimethylaminopropylamide

-
-
1380723-48-7
laurylamidopropyltrimethylammonium acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: methanol / 10 h / 150 °C / 10501.1 Torr 2: methanol / 4 h / 35 °C View Scheme |

| Conditions | Yield |
|---|---|
| at 40℃; for 4h; |

| Conditions | Yield |
|---|---|
| at 40℃; for 4h; |

| Conditions | Yield |
|---|---|
| In water at 70℃; for 5h; Temperature; Solvent; |

| Conditions | Yield |
|---|---|
| In ethanol at 50℃; for 5h; Solvent; Temperature; | |
| In ethanol for 8h; Solvent; Reflux; | 9.1 g |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 80 - 105℃; for 9h; pH=8.5 - 12; |
Dimethylaminopropyl lauramide Specification
The Dodecanamide,N-[3-(dimethylamino)propyl]- is an organic compound with the formula C17H36N2O. The IUPAC name of this chemical is N-[3-(dimethylamino)propyl]dodecanamide. With the CAS registry number 3179-80-4, it is also named as Dodecanamide, N-(3-(dimethylamino)propyl)-. The product's categories are Amides; Carbonyl Compounds; Organic Building Blocks. Besides, it should be stored in a closed cool and dry place.
Physical properties about Dodecanamide,N-[3-(dimethylamino)propyl]- are: (1)ACD/LogP: 4.64; (2)ACD/LogD (pH 5.5): 1.63; (3)ACD/LogD (pH 7.4): 2.86; (4)ACD/BCF (pH 5.5): 1.96; (5)ACD/BCF (pH 7.4): 32.79; (6)ACD/KOC (pH 5.5): 7.9; (7)ACD/KOC (pH 7.4): 132.34; (8)#H bond acceptors: 3; (9)#H bond donors: 1; (10)#Freely Rotating Bonds: 14; (11)Polar Surface Area: 23.55 Å2; (12)Index of Refraction: 1.459; (13)Molar Refractivity: 88.15 cm3; (14)Molar Volume: 322.3 cm3; (15)Polarizability: 34.94×10-24cm3; (16)Surface Tension: 32 dyne/cm; (17)Density: 0.882 g/cm3; (18)Flash Point: 207.1 °C; (19)Enthalpy of Vaporization: 67.25 kJ/mol; (20)Boiling Point: 418.9 °C at 760 mmHg; (21)Vapour Pressure: 3.17E-07 mmHg at 25°C.
Preparation: this chemical can be prepared by N-[3-(dimethylamino)propyl]dodecanamide. The reaction temperature is 120 - 130 °C. The yield is about 91%.
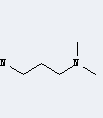
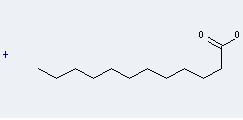
.gif)
Uses of Dodecanamide,N-[3-(dimethylamino)propyl]-: it can be used to produce C19H41N2O(1+)*Br(1-) by heating. It will need reagent propan-2-ol with reaction time of 8 hours. The yield is about 95%.
![]()
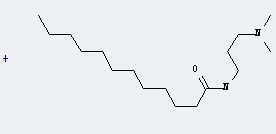
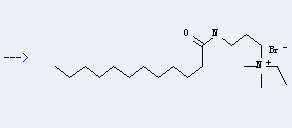
When you are using this chemical, please be cautious about it as the following:
This chemical is irritating to eyes, respiratory system and skin. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(NCCCN(C)C)CCCCCCCCCCC
(2)InChI: InChI=1/C17H36N2O/c1-4-5-6-7-8-9-10-11-12-14-17(20)18-15-13-16-19(2)3/h4-16H2,1-3H3,(H,18,20)
(3)InChIKey: TWMFGCHRALXDAR-UHFFFAOYAR
(4)Std. InChI: InChI=1S/C17H36N2O/c1-4-5-6-7-8-9-10-11-12-14-17(20)18-15-13-16-19(2)3/h4-16H2,1-3H3,(H,18,20)
(5)Std. InChIKey: TWMFGCHRALXDAR-UHFFFAOYSA-N
Related Products
- Dimethylaminopropyl lauramide
- 3179-89-3
- 3179-90-6
- 31799-71-0
- 31799-91-4
- 31805-83-1
- 31807-55-3
- 3180-81-2
- 3181-38-2
- 31823-05-9
- 318237-73-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View