-
Name
3-METHOXYPHENYLMAGNESIUM BROMIDE
- EINECS
- CAS No. 36282-40-3
- Article Data11
- CAS DataBase
- Density 1.013 g/mL at 25 °C
- Solubility
- Melting Point
- Formula C7H7BrMgO
- Boiling Point 65-67 °C
- Molecular Weight 211.341
- Flash Point −4 °F
- Transport Information UN 2924 3/PG 2
- Appearance Clear colorless to brown solution
- Safety 16-26-36/37/39-45
- Risk Codes 11-14-19-20/21/22-34
-
Molecular Structure
-
Hazard Symbols
 F;
F;  Xi
Xi
- Synonyms (m-Methoxyphenyl)magnesiumbromide (6CI);Benzene, methoxy-, magnesium complex;3-Anisylmagnesium bromide;3-Methoxyphenylmagnesium bromide;m-Anisylmagnesium bromide;
- PSA 9.23000
- LogP 2.34100
Synthetic route
-
-
491-37-2
2,3-dihydro-4H-1-benzopyran-4-one

-
-
2398-37-0
3-methoxyphenyl bromide

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

| Conditions | Yield |
|---|---|
| With hydrogenchloride In tetrahydrofuran |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran |
-
-
2398-37-0
3-methoxyphenyl bromide

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

| Conditions | Yield |
|---|---|
| With magnesium; iodine In tetrahydrofuran at 20℃; Heating / reflux; | |
| With magnesium In tetrahydrofuran Inert atmosphere; | |
| With iodine; magnesium In tetrahydrofuran for 2h; Inert atmosphere; Reflux; |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
108-94-1
cyclohexanone

-
-
1884-42-0
1-(m-Methoxyphenyl)cyclohexanol

| Conditions | Yield |
|---|---|
| Stage #1: (3-methoxyphenyl)magnesium bromide; cyclohexanone In diethyl ether at 0 - 20℃; for 4h; Stage #2: With water at 0℃; | 100% |
| In tetrahydrofuran for 0.333333h; | |
| In tetrahydrofuran; toluene at 0℃; for 5.5h; Inert atmosphere; |
-
-
3132-99-8
m-bromobenzoic aldehyde

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
76778-33-1
3-bromo-α-(3-methoxyphenyl)benzenemethanol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 0.25h; | 100% |
| Stage #1: m-bromobenzoic aldehyde; (3-methoxyphenyl)magnesium bromide In tetrahydrofuran at 80℃; for 3h; Grignard reaction; Inert atmosphere; Stage #2: With water; sodium chloride In tetrahydrofuran at 20℃; |

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

| Conditions | Yield |
|---|---|
| Stage #1: 3-(4-methoxy-benzyl)-3-aza-bicyclo[3.2.1]octan-8-one; (3-methoxyphenyl)magnesium bromide In tetrahydrofuran at -78 - 20℃; Stage #2: With hydrogenchloride In water pH=1; | 100% |
-
-
150356-32-4
ethyl 3-(3,4-methylenedioxyphenyl)-1-oxoindene-2-carboxylate

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
150356-38-0
Ethyl (1RS)-1-Hydroxy-1-(3-methoxyphenyl)-3-(3,4-methylenedioxyphenyl)indene-2-carboxylate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran | 100% |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
85909-08-6
tert-butyl 2-oxopyrrolidine-1-carboxylate

-
-
914954-34-0
tert-butyl (4-(3-methoxyphenyl)-4-oxobutyl)carbamate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -40 - 0℃; for 3h; | 100% |
| In tetrahydrofuran at -78 - 20℃; for 14h; Inert atmosphere; Schlenk technique; | 45% |
-
-
22399-01-5
diethyl 2-(4-bromobenzylidene)malonate

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

| Conditions | Yield |
|---|---|
| Stage #1: diethyl 2-(4-bromobenzylidene)malonate; (3-methoxyphenyl)magnesium bromide In tetrahydrofuran; diethyl ether at 0℃; for 3.3h; Stage #2: With water; ammonium chloride | 100% |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
1122-91-4
4-bromo-benzaldehyde

-
-
134446-26-7
(4-bromo-phenyl)-(3-methoxy-phenyl)-methanol

| Conditions | Yield |
|---|---|
| Stage #1: (3-methoxyphenyl)magnesium bromide; 4-bromo-benzaldehyde In tetrahydrofuran at 80℃; for 3h; Grignard reaction; Inert atmosphere; Stage #2: With water; sodium chloride In tetrahydrofuran at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one; palladium diacetate; tricyclohexylphosphine In tetrahydrofuran at 20℃; for 20h; Kumada coupling; | 99% |
| With 1,3-bis(mesityl)imidazolium chloride; palladium diacetate In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 20℃; for 1h; Kumada reaction; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: C17H16O3; (3-methoxyphenyl)magnesium bromide In tetrahydrofuran at 0 - 20℃; for 0.5h; Inert atmosphere; Stage #2: With water In tetrahydrofuran at 0℃; diastereoselective reaction; | 99% |
-
-
137495-63-7
3-iodo-1,2-dicarba-closo-dodecaborane

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
B
-
1159494-39-9
3-meta-methoxyphenyl-o-carborane

| Conditions | Yield |
|---|---|
| With copper(l) iodide; bis-triphenylphosphine-palladium(II) chloride In tetrahydrofuran Ar, a soln. of Mg compd. (8.5 equiv.) added dropwise to a soln. of B compd., CuI and catalyst, refluxed for 34 h; quenched (dilute HCl), extd. (ethyl acetate), washed (water), dried (MgSO4), concd., chromy. (silica gel); | A 0% B 99% |

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

| Conditions | Yield |
|---|---|
| at 20℃; for 13h; Inert atmosphere; | 99% |
-
-
30404-50-3
8-methoxy-3,4-dihydroisocoumarin

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
891829-94-0
[2-(2-hydroxy-ethyl)-6-methoxy-phenyl]-(3-methoxy-phenyl)-methanone

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; dichloromethane at -78 - 20℃; | 98% |
-
-
1711-09-7
3-bromobenzoyl chloride

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
750633-66-0
(3-bromophenyl)-(3-methoxyphenyl)-methanone

| Conditions | Yield |
|---|---|
| Stage #1: 3-bromobenzoyl chloride With tributylphosphine In tetrahydrofuran at 20℃; for 0.333333h; Stage #2: (3-methoxyphenyl)magnesium bromide In tetrahydrofuran at 20℃; for 0.166667h; | 98% |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
521270-76-8
(2-isocyanophenyl)(phenyl)methanone

-
-
1253396-21-2
2-(3-methoxyphenyl)-3-phenyl-3H-indol-3-ol

| Conditions | Yield |
|---|---|
| Stage #1: (3-methoxyphenyl)magnesium bromide; (2-isocyanophenyl)(phenyl)methanone In tetrahydrofuran; diethyl ether at 0℃; Stage #2: With water; ammonium chloride In tetrahydrofuran; diethyl ether at 0℃; | 98% |
-
-
426265-40-9
quinolin-6-yl 4-methylbenzene-1-sulfonate

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
1023531-31-8
6-(3'-methoxyphenyl)quinoline

| Conditions | Yield |
|---|---|
| Stage #1: (3-methoxyphenyl)magnesium bromide With C24H52Cl2O12P4Pd2 In tetrahydrofuran; 1,4-dioxane at 20℃; for 0.0833333h; Kumada-Corriu cross-coupling reaction; Inert atmosphere; Stage #2: quinolin-6-yl 4-methylbenzene-1-sulfonate In tetrahydrofuran; 1,4-dioxane at 80℃; for 24h; Kumada-Corriu cross-coupling reaction; Inert atmosphere; regioselective reaction; | 98% |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
75209-54-0
(E)-1-(3-methoxyphenyl)cyclohept-1-en

| Conditions | Yield |
|---|---|
| With N,N'-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene hydrochloride; palladium diacetate In tetrahydrofuran at 50℃; for 3h; Schlenk technique; Inert atmosphere; Sealed tube; | 98% |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

| Conditions | Yield |
|---|---|
| With copper acetylacetonate In tetrahydrofuran at -18 - 0℃; for 2h; enantiospecific reaction; | 98% |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
106-95-6
allyl bromide

-
-
24743-14-4
1-allyl-3-methoxybenzene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 97% |
| In tetrahydrofuran; toluene at 0 - 20℃; for 18h; | 81% |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
591-31-1
3-methoxy-benzaldehyde

-
-
55360-45-7
bis(3-methoxyphenyl)methanol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 3h; Grignard Reaction; | 97% |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
123-62-6
propionic acid anhydride

-
-
37951-49-8
1-(3-methoxyphenyl)propan-1-one

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether at -70℃; | 97% |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
389838-05-5
(1E)-4,4-bis(methylthio)-2-nitro-1-pyrrolidino-1,3-butadiene

-
-
467421-88-1
(E)-4-(3-methoxyphenyl)-1,1-bis(methylthio)-3-nitro-1,3-butadiene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -78℃; | 97% |
-
-
40705-22-4
4-benzyloxy-6-bromo-3-methoxybenzaldehyde

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
1569316-45-5
[4-(benzyloxy)-2-bromo-5-methoxyphenyl](3-methoxyphenyl)methanol

| Conditions | Yield |
|---|---|
| In diethyl ether at 0 - 20℃; for 3h; Inert atmosphere; | 97% |
-
-
20469-65-2
1-bromo-3,5-dimethoxybenzene

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
33580-34-6
3,3',5-trimethoxy-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With nickel(II) fluoride; 1,3-bis(3,6-diisopropylphenyl)imidazolium hydrochloride In tetrahydrofuran at 0 - 80℃; for 12.5h; Inert atmosphere; | 97% |
-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
6161-50-8
3,3'-dimethoxybiphenyl

| Conditions | Yield |
|---|---|
| With iron(III) chloride; 1,2-dichloro-ethane In tetrahydrofuran at 20℃; for 0.166667h; | 96% |
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In tetrahydrofuran at 66℃; for 0.166667h; Inert atmosphere; | 87% |
| With 4-oxo-2,2,6,6-tetramethylpiperidin-oxyl In tetrahydrofuran at 66℃; for 0.166667h; Inert atmosphere; | 87% |

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 0.5h; Grignard reaction; | 96% |
-
-
14243-64-2
(triphenylphosphine)gold(I) chloride

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
870485-34-0
(3-methoxyphenyl)(triphenyl-λ5-phosphanyl)gold

| Conditions | Yield |
|---|---|
| In tetrahydrofuran 3-methoxyphenylmagnesium bromide added dropwise at -78°C to soln.ClAuPPh3 in THF in Schlenk techniques under Ar; mixt. warmed to room te mp., mixed for 16 h; treated with satd. soln. of NH4Cl, extd. with Et2O; org. layers washed with satd. soln. of NaCl, dried over MgSO4; solvents removed under vac.; crystals obtained via crystn. from CHCl3; | 96% |

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

| Conditions | Yield |
|---|---|
| Stage #1: (1RS,5RS,6RS,7RS)-3-methoxy-6-methyl-7-phenylbicyclo[3.2.1]-oct-3-ene-2,8-dione; (3-methoxyphenyl)magnesium bromide In tetrahydrofuran at 0 - 20℃; for 0.5h; Inert atmosphere; Stage #2: With water In tetrahydrofuran at 0℃; diastereoselective reaction; | 96% |
-
-
108-85-0
1-bromocyclohexane

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
41876-58-8
1-cyclohexyl-3-methoxybenzene

| Conditions | Yield |
|---|---|
| With Iron(III) amine-bis(phenolate) complex In diethyl ether at 20℃; for 0.5h; Inert atmosphere; | 96% |
| With C54H80Cl2Fe2N2O4 In diethyl ether at 22℃; for 0.5h; Inert atmosphere; | > 95 %Chromat. |
| With [Fe(n-propylamino-N,N-bis(2-methylene-4-tert-butyl-6-methylphenolate))(chloride)]2 In diethyl ether at 22℃; for 0.5h; Catalytic behavior; | > 95 %Chromat. |

| Conditions | Yield |
|---|---|
| Stage #1: 4-vinylbenzyl chloride With cobalt(III) acetylacetonate In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 30℃; for 1h; Stage #2: (3-methoxyphenyl)magnesium bromide In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 30℃; for 0.333333h; | 96% |
-
-
6451-86-1
5-benzyloxy-2-bromo-4-methoxybenzaldehyde

-
-
36282-40-3
(3-methoxyphenyl)magnesium bromide

-
-
1569316-44-4
[5-(benzyloxy)-2-bromo-4-methoxyphenyl](3-methoxyphenyl)methanol

| Conditions | Yield |
|---|---|
| In diethyl ether at 0 - 20℃; for 3h; Inert atmosphere; | 96% |
Magnesium,bromo(3-methoxyphenyl)- Specification
The Magnesium,bromo(3-methoxyphenyl)-, with the CAS registry number 36282-40-3, has the systematic name of bromo(3-methoxyphenyl)magnesium. It is a kind of clear colorless to brown solution, and belongs to the following product categories: Aryl; Grignard Reagents; Organometallic Reagents. And the molecular formula of the chemical is C7H7BrMgO.
The characteristics of Magnesium,bromo(3-methoxyphenyl)- are as followings: (1)#H bond acceptors: 1; (2)#H bond donors: 0; (3)#Freely Rotating Bonds: 2; (4)Polar Surface Area: 9.23Å2.
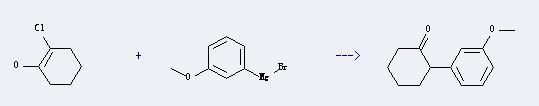
Uses of Magnesium,bromo(3-methoxyphenyl)-: It can react with 2-chloro-cyclohexanone to produce 2-(3-methoxy-phenyl)-cyclohexanone. The yield of the reaction is about 53%.
You should be cautious while dealing with this chemical. It is a kind of highly flammable chemical which is harmful by inhalation, in contact with skin and if swallowed. What's more, it reacts violently with water, and may cause burns. Therefore, you had better take the following instructions: Keep away from sources of ignition - No smoking; Wear suitable gloves and eye/face protection, and if in case of contacting with eyes, rinse immediately with plenty of water and seek medical advice; In case of accident or if you feel unwell, seek medical advice immediately (show label where possible).
Addtionally, the following datas could be converted into the molecular structure:
(1)SMILES: COc1cc([Mg]Br)ccc1
(2)InChI: InChI=1/C7H7O.BrH.Mg/c1-8-7-5-3-2-4-6-7;;/h2-3,5-6H,1H3;1H;/q;;+1/p-1/rC7H7BrMgO/c1-10-7-4-2-3-6(5-7)9-8/h2-5H,1H3
(3)InChIKey: AJKNYEHIJAMGHX-KNINMPIZAP
Related Products
- Magnesium
- Magnesium (2-methylpentyl)-oxide
- Magnesium acetate
- Magnesium acetate tetrahydrate
- Magnesium acetate tetrahydrate
- Magnesium acetylacetonate dihydrate
- Magnesium acrylate
- Magnesium ascorbyl phosphate
- Magnesium ascorbyl phosphate
- Magnesium aspartate tetrahydrate
- 36282-47-0
- 36283-44-0
- 36284-77-2
- 36286-75-6
- 36286-79-0
- 36286-80-3
- 36289-36-8
- 36290-04-7
- 36291-32-4
- 36292-66-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View