-
Name
Methyl 4-nitrobenzoate
- EINECS 210-599-2
- CAS No. 619-50-1
- Article Data445
- CAS DataBase
- Density 1.301 g/cm3
- Solubility
- Melting Point 94-96 °C(lit.)
- Formula C8H7NO4
- Boiling Point 302.6 °C at 760 mmHg
- Molecular Weight 181.148
- Flash Point 149.2 °C
- Transport Information
- Appearance light yellow to yellow fine crystalline powder
- Safety 22-24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Benzoicacid, p-nitro-, methyl ester (6CI,8CI);4-Methoxycarbonylnitrobenzene;4-nitrobenzoic acid methyl ester;Methyl 4-nitrobenzoate;Methylp-nitrobenzoate;NSC 4137;p-Carbomethoxynitrobenzene;p-Nitrobenzoic acidmethyl ester;
- PSA 72.12000
- LogP 1.90460
Synthetic route

| Conditions | Yield |
|---|---|
| With dmap; 2-chloro-1-(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,11,11,12,12,12-henicosafluorododecyl)pyridinium trifluoromethanesulfonate; triethylamine In N,N-dimethyl-formamide at 20℃; for 1h; | 100% |
| Stage #1: 4-nitro-benzoic acid With 1H-imidazole; iodine; triphenylphosphine In dichloromethane at 20℃; for 0.0833333h; Garegg-Samuelsson type reaction; Stage #2: methanol In dichloromethane at 20℃; Garegg-Samuelsson type reaction; | 100% |
| With dmap; 2-chloro-1-(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,11,11,12,12,12-henicosafluorododecyl)pyridinium trifluoromethanesulfonate; triethylamine In N,N-dimethyl-formamide at 20℃; for 1h; | 100% |
-
-
636-98-6
p-nitrobenzene iodide

-
-
201230-82-2
carbon monoxide

-
-
1067-21-6
triethylmethoxystannane

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With iodophenylbis(triphenylphosphine)palladium under 760 Torr; for 1h; | 100% |

-
-
18107-18-1
diazomethyl-trimethyl-silane

-
-
62-23-7
4-nitro-benzoic acid

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| In methanol; toluene at 20℃; for 2h; | 100% |
| In ethyl acetate | 60% |
| In methanol; diethyl ether; hexane at 20℃; for 2h; Inert atmosphere; | 178 mg |
-
-
5564-73-8
1-(dimethoxymethyl)pyrrolidine

-
-
62-23-7
4-nitro-benzoic acid

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| In 1,2-dichloro-ethane at 30℃; for 25h; | 100% |
-
-
67-56-1
methanol

-
A
-
619-50-1
4-nitrobenzoic acid methyl ester

-
B
-
154109-07-6
diethyl [N-(hydroxy)imino](p-fluorophenyl)methylphosphonate

| Conditions | Yield |
|---|---|
| for 6h; Inert atmosphere; Reflux; | A 100% B 100% |


| Conditions | Yield |
|---|---|
| With water; iodine; sodium nitrite at 20 - 70℃; chemoselective reaction; | 99% |
| With ammonium peroxydisulfate at 60℃; for 2.5h; | 99% |
| With potassium hydroxide; iodine at 0℃; for 0.0166667h; | 98% |

| Conditions | Yield |
|---|---|
| With sodium carbonate; palladium; silver(l) oxide at 80℃; for 48h; Molecular sieve; | 99% |
| With dihydrogen peroxide for 3h; Irradiation; | 95% |
| With oxygen; potassium carbonate at 60℃; under 750.075 Torr; for 20h; | 94% |

| Conditions | Yield |
|---|---|
| scandium tris(trifluoromethanesulfonate) at 64℃; for 168h; | 98% |
| With dipotassium hydrogenphosphate for 5h; Reflux; | 98% |
| With potassium ethoxide | |
| acrylonitrile; triphenylphosphine at 25℃; for 24h; | 71 % Chromat. |
-
-
88470-26-2
N,N-dimethoxyamine

-
-
62-23-7
4-nitro-benzoic acid

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| In benzene for 3h; Heating; | 98% |
| In benzene Product distribution; Mechanism; Heating; other nucleophiles; var. reaction conditions; | 98% |
| In benzene for 3h; Mechanism; Heating; | 98% |
-
-
4525-33-1
dimethyl dicarbonate

-
-
62-23-7
4-nitro-benzoic acid

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| magnesium(II) perchlorate In nitromethane at 40℃; for 16h; | 98% |
| With 2,6-dimethylpyridine; fac-tris(2-phenylpyridinato-N,C2')iridium(III) In N,N-dimethyl-formamide at 20℃; for 48h; Sealed tube; Inert atmosphere; Irradiation; |
-
-
67-56-1
methanol

-
-
636-98-6
p-nitrobenzene iodide

-
-
201230-82-2
carbon monoxide

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With palladium diacetate; caesium carbonate; 1,3,5,7-tetramethyl-8-phenyl-2,4,6-trioxa-8-phosphatricyclo[3.3.1.13,7]decane In N,N-dimethyl-formamide at 80℃; under 2327.23 Torr; | 98% |
| With triethylamine at 100℃; under 3750.38 Torr; for 1.5h; Inert atmosphere; | 95% |
| With palladium diacetate; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In N,N-dimethyl-formamide at 100℃; under 5250.53 Torr; continuous flow reactor; | 81% |

-
-
80-11-5
N-methyl-N-nitrosotoluene-p-sulfonamide

-
-
62-23-7
4-nitro-benzoic acid

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With potassium hydroxide In tetrahydrofuran; methanol; water at 20℃; | 98% |
| Stage #1: N-methyl-N-nitrosotoluene-p-sulfonamide With potassium hydroxide In methanol; water; N,N-dimethyl-formamide Flow reactor; Stage #2: 4-nitro-benzoic acid In methanol; diethyl ether; water; N,N-dimethyl-formamide at 0 - 25℃; Sonication; | 90% |
-
-
67-56-1
methanol

-
-
636-97-5
N-amino-(4-nitrophenyl)carboxamide

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate at 20℃; for 0.25h; Esterification; | 97% |
| With Oxone at 20℃; for 6h; oxidative esterification; | 84% |

| Conditions | Yield |
|---|---|
| In 1,2-dimethoxyethane for 16h; | 97% |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In acetonitrile | 97% |
-
-
681-84-5
tetramethylorthosilicate

-
-
555-16-8
4-nitrobenzaldehdye

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride; (t-Bu2POH)2PdCl2 In acetonitrile at 50℃; for 24h; | 97% |
-
-
121-45-9
phosphorous acid trimethyl ester

-
-
62-23-7
4-nitro-benzoic acid

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With diethylazodicarboxylate In tetrahydrofuran for 2h; | 96% |
| With diethylazodicarboxylate In tetrahydrofuran for 2h; Mechanism; other phosphite, other carboxylic acid, phenols, imides and C-H acids; | 92% |
-
-
67-56-1
methanol

-
-
50-00-0
formaldehyd

-
-
120-46-7
1,3-diphenylpropanedione

-
A
-
619-50-1
4-nitrobenzoic acid methyl ester

-
B
-
21600-84-0
bis-1,5(4-nitrophenyl)-1,5-pentanedione

| Conditions | Yield |
|---|---|
| hydroxocobalt(III) Schiff base at 60℃; for 1.5h; Product distribution; Mechanism; var. 4-subst. educts; var. reaction conditions; | A 92% B 96% |


| Conditions | Yield |
|---|---|
| With N,N-dimethyl-formamide dimethyl acetal for 16h; Heating; | 95% |
-
-
17302-46-4
methyl 2-hydroxy-5-nitrobenzoate

-
-
62-23-7
4-nitro-benzoic acid

-
A
-
619-50-1
4-nitrobenzoic acid methyl ester

-
B
-
96-97-9
5-nitrosalicylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitro-benzoic acid With potassium carbonate In N,N-dimethyl acetamide at 110℃; for 0.5h; Stage #2: methyl 2-hydroxy-5-nitrobenzoate at 110℃; for 24h; Temperature; | A 95% B n/a |


| Conditions | Yield |
|---|---|
| With C26H28N3O3S(1+)*F6P(1-); nickel diacetate; potassium carbonate; triethylamine at 25℃; for 2h; | 95% |
-
-
616-38-6
carbonic acid dimethyl ester

-
-
62-23-7
4-nitro-benzoic acid

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid at 80 - 85℃; for 9.5h; Neat (no solvent); | 94.2% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In N,N-dimethyl-formamide for 0.0180556h; microwave irradiation; | 90% |
| With potassium carbonate In dimethyl sulfoxide at 90℃; for 16h; Inert atmosphere; Green chemistry; | 86% |

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane for 0.166667h; | 94% |
| With pyridine at 70℃; for 3h; | 91% |
| at 25℃; Thermodynamic data; ΔH; |

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride at 20℃; for 2.5h; Esterification; | 94% |
-
-
67-56-1
methanol

-
-
201230-82-2
carbon monoxide

-
-
586-78-7
para-nitrophenyl bromide

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With palladium diacetate; caesium carbonate; 1,3,5,7-tetramethyl-8-phenyl-2,4,6-trioxa-8-phosphatricyclo[3.3.1.13,7]decane In N,N-dimethyl-formamide at 80℃; under 2327.23 Torr; | 94% |
| With palladium diacetate; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In 1,4-dioxane at 100℃; under 11251.1 Torr; continuous flow reactor; | 77% |
| With palladium diacetate; triethylamine; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene; hydrazine In tetrahydrofuran; 1,4-dioxane at 100℃; under 11251.1 Torr; Temperature; Flow reactor; | 77% |
| With ruthenium(III) trichloride hydrate; 1,4-bis(2'-diphenylphosphino-3'-methylimidazolium)butane triflate; triethylamine In 1-methyl-pyrrolidin-2-one at 140℃; under 15001.5 Torr; for 16h; Autoclave; | 80 %Chromat. |

-
-
67-56-1
methanol

-
-
54914-77-1
1-(4-nitrophenyl)-3-phenyl-1-propanone

-
A
-
104-53-0
3-phenyl-propionaldehyde

-
B
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With N,N-dimethyl-formamide dimethyl acetal for 16h; Heating; | A n/a B 93% |

-
-
186581-53-3, 908094-01-9
diazomethane

-
-
619-73-8
4-nitrobenzyl chloride

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitrobenzyl chloride With sodium chlorite; sodium dihydrogenphosphate; Me-AZADO+Cl- In dichloromethane; water at 25℃; for 1.5h; Stage #2: With 2-methyl-but-2-ene In dichloromethane; water Stage #3: diazomethane With hydrogenchloride; sodium hydroxide Product distribution / selectivity; more than 3 stages; | 93% |
| Stage #1: 4-nitrobenzyl chloride With sodium chlorite; sodium dihydrogenphosphate; 2,2,6,6-tetramethylpiperidin-1-oxoammonium chloride In dichloromethane; water at 25℃; for 1.5h; Stage #2: With 2-methyl-but-2-ene In dichloromethane; water Stage #3: diazomethane With hydrogenchloride; sodium hydroxide Product distribution / selectivity; more than 3 stages; | 63% |
-
-
99768-12-4
4-methoxycarbonylphenylboronic acid

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With 1,1,1,3',3',3'-hexafluoro-propanol; N–nitrosaccharin at 60℃; for 19h; Reagent/catalyst; Inert atmosphere; Schlenk technique; | 93% |
| With Iron(III) nitrate nonahydrate In toluene at 80℃; for 18h; Inert atmosphere; | 86% |
| With dipotassium peroxodisulfate; bismuth (III) nitrate pentahydrate In benzene at 70℃; for 12h; Inert atmosphere; | 74% |
| With copper(I) oxide; tetrabutylammonium nitrite In acetonitrile at 20℃; | 53% |
-
-
67-56-1
methanol

-
-
25393-54-8
N-(4-nitrobenzoyl)oxazolidinone

-
-
619-50-1
4-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With [t-Bu2SnCl(OH)]2 In toluene at 85℃; for 14h; | 92% |

| Conditions | Yield |
|---|---|
| With tert-butylamine for 10h; Heating; | 100% |
| Stage #1: ethanol With platinum(IV) oxide; hydrogen at 60℃; under 750.075 - 1500.15 Torr; for 2h; Autoclave; Green chemistry; Stage #2: 4-nitrobenzoic acid methyl ester at 60℃; Autoclave; Inert atmosphere; Green chemistry; chemoselective reaction; | 93% |
| With dichlorobis(1-butylimidazolium) zinc at 70 - 80℃; | 93% |
-
-
619-50-1
4-nitrobenzoic acid methyl ester

-
-
24226-29-7
N-[(4-methoxycarbonyl)phenyl]hydroxylamine

| Conditions | Yield |
|---|---|
| With 5% rhodium-on-charcoal; hydrazine hydrate In tetrahydrofuran at 0℃; for 5h; Inert atmosphere; | 100% |
| With 5% rhodium-on-charcoal; hydrazine hydrate In tetrahydrofuran at 0℃; Inert atmosphere; | 100% |
| With rhodium contaminated with carbon; hydrazine hydrate In tetrahydrofuran at 0℃; for 5h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; polymethylhydrosiloxane; palladium diacetate In tetrahydrofuran; water at 20℃; for 0.5h; | 100% |
| With potassium fluoride; polymethylhydrosiloxane; palladium diacetate In tetrahydrofuran at 20℃; for 0.5h; | 100% |
| With sodium tetrahydroborate; water In ethanol at 25℃; for 1h; Solvent; chemoselective reaction; | 100% |
-
-
619-50-1
4-nitrobenzoic acid methyl ester

-
-
75-05-8
acetonitrile

-
-
79663-14-2
methyl 4-(ethylamino)benzoate

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; ammonium acetate; hydrogen In methanol at 20℃; under 760.051 Torr; for 24h; | 100% |

| Conditions | Yield |
|---|---|
| With AlBrCl3(1-)*C5H5N*H(1+) at 140℃; for 3h; | 99% |
| With methanesulfonic acid; 1-methylimidazolium hydrobromide at 120℃; for 2h; | 96% |
| With iron(III) sulfate; water In toluene at 110℃; for 4h; Ionic liquid; | 95% |

| Conditions | Yield |
|---|---|
| With lanthanum(III) nitrate hexahydrate; TOP at 110℃; for 5h; Reagent/catalyst; Molecular sieve; | 99% |
| With N,N'-biscyclohexyl-imidazol-2-ylidene; 4 A molecular sieve In tetrahydrofuran at 20℃; for 0.25h; | 96% |
| 1,3-dicyclohexyl-imidazol-2-ylidene In tetrahydrofuran at 20℃; for 0.25h; | 96% |
-
-
112-92-5
1-octadecanol

-
-
619-50-1
4-nitrobenzoic acid methyl ester

-
-
56426-96-1
octadecyl 4-nitro-benzoate

| Conditions | Yield |
|---|---|
| Stage #1: 1-octadecanol; 4-nitrobenzoic acid methyl ester In xylene for 0.5h; Heating; Stage #2: With TiO(acac)2 In xylene for 4.5h; Heating; | 99% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In dimethyl sulfoxide at 25℃; for 1.5h; | 99% |
| With magnesium methanolate In N,N-dimethyl-formamide at 80℃; for 8h; | 78% |

| Conditions | Yield |
|---|---|
| With Zn4(OCOCF3)6O In di-isopropyl ether for 40h; Heating; | 99% |

| Conditions | Yield |
|---|---|
| With tetrabutylammonium acetate; oxiranyl-methanol In hexane for 2h; Reflux; | 99% |
-
-
619-50-1
4-nitrobenzoic acid methyl ester

-
-
23663-92-5
dimethyl 4,4'-azoxydibenzoate

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; isopropyl alcohol at 30℃; under 760.051 Torr; for 10h; Inert atmosphere; | 98% |
| With manganese; acetic acid In tetrahydrofuran; water at 20℃; for 15h; | 70% |
| With triethylsilane; indium(III) bromide In N,N-dimethyl-formamide at 60℃; for 12h; | 64% |
| With bismuth; sodium hydroxide; sodium tetrahydroborate; oxygen 1.) methanol, room temperature, 0.5 h, 2.) methanol, room temperate, 4 h; Yield given. Multistep reaction; | |
| Multi-step reaction with 2 steps 1: 78 percent / Sb, NaBH4 / methanol / 1 h / Ambient temperature 2: 88 percent / NaOH, O2 / 4 h / Ambient temperature View Scheme |
-
-
619-50-1
4-nitrobenzoic acid methyl ester

-
-
156-87-6
propan-1-ol-3-amine

-
-
90690-76-9
N-(3-Hydroxy-propyl)-4-nitro-benzamid

| Conditions | Yield |
|---|---|
| With ammonium nitrate In neat (no solvent) at 20℃; for 12h; Green chemistry; | 98% |
| In toluene at 97℃; for 4h; Acylation; | 79% |
-
-
619-50-1
4-nitrobenzoic acid methyl ester

-
-
100-36-7
N,N-diethylethylenediamine

-
-
1664-52-4
N-(2-(diethylamino)ethyl)-4-nitrobenzamide

| Conditions | Yield |
|---|---|
| With ammonium nitrate In neat (no solvent) at 20℃; for 12h; Green chemistry; | 98% |

| Conditions | Yield |
|---|---|
| With ammonia In methanol at 70℃; | 98% |
Methyl 4-nitrobenzoate Chemical Properties
MW: 181.15
EINECS: 210-599-2
METHYL-p-NITROBENZOATE,it's melting point is about 94-96 °C(lit.);It's appearance is white or yellow crystal.Soluble in ethanol, Ether, Chloroform, insoluble in water.
Following is the molecular structure of METHYL-p-NITROBENZOATE:
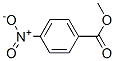
Methyl 4-nitrobenzoate Toxicity Data With Reference
| 1. | ipr-mus LD50:200 mg/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) No. AD691-490 . |
RTECS : DH5775000
Methyl 4-nitrobenzoate Consensus Reports
Methyl 4-nitrobenzoate Safety Profile
Safety Statements: 22-24/25 (Do not breathe dust;Avoid contact with skin and eyes )
WGK Germany: 3
Related Products
- Methyl (2-chloromethyl)oxazole-4-carboxylate
- Methyl (2R)-2-bromo-2-(2-chlorophenyl)acetate
- Methyl (2R,3S)-3-(4-methoxyphenyl)-2-oxiranecarboxylate
- Methyl (2R,3S)-3-(benzoylamino)-2-hydroxy-3-phenylpropanoate
- Methyl (2R,3S)-3-(tert-butoxycarbonylamino)-2-hydroxy-3-phenylpropionate
- Methyl (2S)-1-(1,2-dioxo-3,3-dimethypentyl)-2-pyrrolidinecarboxylate
- Methyl (2S)-2,3-epoxypropanoate
- Methyl (2S)-2-[(tert-butoxycarbonyl)amino]-3-[4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]propanoate
- Methyl (2S)-2-[4-(2,4-dichlorophenoxy)phenoxy]propanoate
- Methyl (2S)-2-amino-2-cyclopentyl-acetate hydrochloride
- 61950-59-2
- 61951-36-8
- 61951-51-7
- 61951-82-4
- 61951-85-7
- 61951-89-1
- 61951-98-2
- 61951-99-3
- 6195-20-6
- 61953-02-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View