-
Name
Octyl-beta-D-glucopyranoside
- EINECS 249-887-8
- CAS No. 29836-26-8
- Article Data51
- CAS DataBase
- Density 1.18 g/cm3
- Solubility 3 g/10 mL in water
- Melting Point 105 °C
- Formula C14H28O6
- Boiling Point 454.1 °C at 760 mmHg
- Molecular Weight 292.373
- Flash Point 228.4 °C
- Transport Information
- Appearance
- Safety 24/25
- Risk Codes 36/37/38
-
Molecular Structure
- Hazard Symbols
- Synonyms Glucopyranoside,octyl, b-D- (8CI);1-O-Octyl-b-D-glucopyranose;1-O-Octyl-b-D-glucopyranoside;1-O-n-Octyl-b-D-glucopyranoside;1-Octyl b-D-glucopyranoside;1-Octyl b-D-glucoside;1-n-Octyl b-D-glucopyranoside;BTB 11967;Octyl glucoside;Octyl b-D-glucoside;Octyl b-D-glycopyranoside;Octyl b-glucopyranoside;n-Octyl b-D-glucopyranoside;n-Octyl b-D-glucoside;b-D-Octyl glucoside;b-Octyl monoglucoside;
- PSA 99.38000
- LogP 0.16340
Synthetic route
-
-
38954-67-5
octyl (2,3,4,6-tetra-O-acetyl)-β-D-glucopyranoside

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 20℃; for 3h; Zemplen reaction; | 100% |
| With sodium methylate In methanol for 2h; Ambient temperature; | 99% |
| With sodium methylate In methanol at 20℃; Inert atmosphere; | 91% |

| Conditions | Yield |
|---|---|
| With citrate buffer; Aerosol-OT; β-glucosidase In octane at 20℃; for 120h; pH=5.5; | 40% |
| With almond meal In water at 50℃; for 168h; Thermodynamic data; Equilibrium constant; Enzymatic reaction; | 4.05% |
| With almond meal cross-linked with glutaraldehyde In water at 50℃; for 168h; Equilibrium constant; Enzymatic reaction; |
-
-
2280-44-6
D-Glucose

-
-
111-87-5
octanol

-
A
-
29781-80-4
octyl alpha-D-glucopyranoside

-
B
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With sulfuric acid In 1,4-dioxane Yields of byproduct given; | A 30% B n/a |
| With boron trifluoride diethyl etherate In tetrahydrofuran for 55h; Ambient temperature; Yield given. Yields of byproduct given; | |
| With acetyl chloride at 120℃; for 0.333333h; microwave irradiation; |


| Conditions | Yield |
|---|---|
| With immobilized β-glucosidase G 0395 from almonds (EC 3.2.1.21) In water at 20℃; for 144h; | 18% |
| With almond β-glucosidase immobilised with ENTP-4000 In water at 50℃; for 96h; | 8.2% |
| In water; acetonitrile at 40℃; for 72h; 90percent CH3CN, almond β-D-glucosidase; Yield given; | |
| With almond β-D-glycosidase immobilized on Amberlite XAD-4; sodium chloride In acetonitrile at 50℃; Kinetics; Further Variations:; Solvents; Reaction partners; Enzymatic reaction; | |
| With almond β-glucosidase immobilized on Amberlite XAD-4; barium(II) chloride In N,N-dimethyl-formamide at 50℃; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With Amberlite IR-120 (H+) resin for 0.133333h; Microwave irradiation; | 15% |
| With β-glucosidase immobilized on polyamine microspheres In aq. phosphate buffer; tert-butyl alcohol at 50℃; for 48h; pH=6; Catalytic behavior; Kinetics; Concentration; Reagent/catalyst; Solvent; Enzymatic reaction; | |
| With recombinant β-glucosidase from Microcerotermes annandalei In aq. phosphate buffer at 30℃; for 144h; |
-
-
133-99-3, 490-37-9, 2152-98-9, 4482-75-1, 5965-66-2, 13299-27-9, 13360-52-6, 14641-93-1, 15548-43-3, 16462-44-5, 16984-36-4, 16984-38-6, 20869-27-6, 22688-72-8, 27452-49-9, 29276-55-9, 30608-12-9, 34481-13-5, 37169-60-1, 37169-64-5, 64234-01-1, 64234-02-2, 75281-88-8, 80446-85-1, 84413-57-0, 92344-56-4, 93221-87-5, 93301-77-0, 94799-29-8, 100427-98-3, 100428-00-0, 101312-82-7, 102046-24-2, 102046-25-3, 109432-00-0, 109432-02-2, 109432-04-4, 109432-08-8, 135268-49-4, 138231-26-2, 140461-59-2, 145414-22-8, 145414-26-2, 146339-75-5, 146339-76-6, 149116-55-2, 528-50-7
Cellobiose

-
-
111-87-5
octanol

-
A
-
2280-44-6
D-Glucose

-
B
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With almond β-glucosidase In acetate buffer; acetonitrile at 50℃; for 72h; pH=4.4; | A n/a B 14.4% |


| Conditions | Yield |
|---|---|
| With phosphate buffer; lactase from Kluyveromyces lactis In nitrobenzene at 50℃; for 1h; | 13% |
-
-
111-87-5
octanol

-
-
5391-18-4, 25320-93-8, 39824-09-4, 95531-15-0, 143289-25-2, 146453-36-3, 148347-27-7
butyl-α-D-glucopyranoside

-
A
-
50-99-7
D-glucose

-
B
-
29781-80-4
octyl alpha-D-glucopyranoside

-
C
-
498-07-7
levoglucosan

-
E
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With Amberlyst 15DRY at 110℃; for 24h; Sealed vial; Ionic liquid; | A 15 %Chromat. B 38 %Chromat. C 10% D 6% E 21 %Chromat. |


| Conditions | Yield |
|---|---|
| With phosphate buffer; β-glucosidase at 30℃; for 17h; pH=5; | 2% |
| With water; almond β-glucosidase In acetate buffer; N,N-dimethyl-formamide at 50℃; pH=5.0; Kinetics; Further Variations:; Solvents; enzyme immobilized on Celite R640<*>; |
-
-
133-99-3, 490-37-9, 2152-98-9, 4482-75-1, 5965-66-2, 13299-27-9, 13360-52-6, 14641-93-1, 15548-43-3, 16462-44-5, 16984-36-4, 16984-38-6, 20869-27-6, 22688-72-8, 27452-49-9, 29276-55-9, 30608-12-9, 34481-13-5, 37169-60-1, 37169-64-5, 64234-01-1, 64234-02-2, 75281-88-8, 80446-85-1, 84413-57-0, 92344-56-4, 93221-87-5, 93301-77-0, 94799-29-8, 100427-98-3, 100428-00-0, 101312-82-7, 102046-24-2, 102046-25-3, 109432-00-0, 109432-02-2, 109432-04-4, 109432-08-8, 135268-49-4, 138231-26-2, 140461-59-2, 145414-22-8, 145414-26-2, 146339-75-5, 146339-76-6, 149116-55-2, 528-50-7
Cellobiose

-
-
111-87-5
octanol

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| In water at 30℃; for 48h; β-glucosidase from Trichoderma viride; Yield given; |
-
-
2280-44-6
D-Glucose

-
-
111-87-5
octanol

-
A
-
29781-80-4
octyl alpha-D-glucopyranoside

-
D
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With iron(III) chloride In tetrahydrofuran for 24h; Product distribution; Ambient temperature; various solvents and promoters investigated; | |
| With H+ ion exchanged zeolite Y at 130℃; Reagent/catalyst; |

-
-
82494-08-4
n-octyl α-D-glucopyranosyl-(1->4)-β-D-glucopyranoside




-
F
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With sodium bromate; phosphate buffer; Ru(azpy)2(H2O)22+ at 60℃; Product distribution; Mechanism; other pH: 3, 4.5, 8.5, 10; |


-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With methanol; sodium methylate | |
| With methanol; barium methoxide |
-
-
185549-56-8
octyl 3,4,6-tri-O-benzyl-β-D-glucopyranoside

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal at 20℃; |

-
A
-
112817-57-9
1,3-bis-[[(6-amino-pyridin-2-yl)amino]carbonyl]benzene

-
B
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
112817-57-9
1,3-bis-[[(6-amino-pyridin-2-yl)amino]carbonyl]benzene

-
B
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
29836-26-8
n-octyl β-D-glucopyranoside

-
B
-
797818-00-9
N,N',N''-tris(6-ethylpyridin-2-yl)benzene-1,3,5-tricarbonamide

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
29836-26-8
n-octyl β-D-glucopyranoside

-
B
-
485831-83-2
1,3,5-tris[(4,6-dimethylpyridin-2-yl)aminomethyl]-2,4,6-trimethylbenzene

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
29836-26-8
n-octyl β-D-glucopyranoside

-
B
-
485831-83-2
1,3,5-tris[(4,6-dimethylpyridin-2-yl)aminomethyl]-2,4,6-trimethylbenzene

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
29836-26-8
n-octyl β-D-glucopyranoside

-
B
-
797818-02-1
1,3,5-tris[(6-aminopyridin-2-yl)aminomethyl]-2,4,6-triethylbenzene

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
29836-26-8
n-octyl β-D-glucopyranoside

-
B
-
797818-02-1
1,3,5-tris[(6-aminopyridin-2-yl)aminomethyl]-2,4,6-triethylbenzene

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
29836-26-8
n-octyl β-D-glucopyranoside

-
B
-
797818-01-0
1,3,5-tris-[(4,6-dimethyl-pyridin-2-yl)-aminomethyl]-2,4,6-triethylbenzene

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
29836-26-8
n-octyl β-D-glucopyranoside

-
B
-
797818-01-0
1,3,5-tris-[(4,6-dimethyl-pyridin-2-yl)-aminomethyl]-2,4,6-triethylbenzene

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |

-
A
-
29836-26-8
n-octyl β-D-glucopyranoside

-
B
-
797818-03-2
2-[3,5-bis[(4,6-dimethylpyridin-2-ylcarbamoyl)methyl]-2,4,6-trimethylphenyl]-N-(4,6-dimethylpyridin-2-yl)acetamide

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 24.85℃; Equilibrium constant; |
-
-
98-88-4
benzoyl chloride

-
-
29836-26-8
n-octyl β-D-glucopyranoside

-
-
1394172-54-3
octyl 4-O-benzoyl-β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With (2S,5S)-2,5-bis[[(2S)-3-(1H-indol-3-yl)-1-(octyloxy)-1-oxopropan-2-ylamino]carbonyl]-1-(pyridin-4-yl)pyrrolidine; N-ethyl-N,N-diisopropylamine; Trimethylacetic acid In chloroform at -20℃; for 0.25h; Catalytic behavior; Reagent/catalyst; | 87% |
-
-
98-88-4
benzoyl chloride

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With pyridine; dmap at 0 - 20℃; for 12h; | 87% |
-
-
77-76-9
2,2-dimethoxy-propane

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In N,N-dimethyl-formamide at 20℃; for 5h; | 83% |
-
-
1161-13-3
N-Cbz-L-Phe

-
-
29836-26-8
n-octyl β-D-glucopyranoside

-
-
1194234-22-4
octyl 4-O-((S)-2-benzyloxycarbonylamino-3-phenylpropanoyl)-β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| Stage #1: N-Cbz-L-Phe With pivaloyl chloride; N-ethyl-N,N-diisopropylamine In chloroform at 20℃; for 1h; Inert atmosphere; Stage #2: n-octyl β-D-glucopyranoside With 2,4,6-trimethyl-pyridine; (2S,5S)-2,5-bis[[(2S)-3-(1H-indol-3-yl)-1-(octyloxy)-1-oxopropan-2-ylamino]carbonyl]-1-(pyridin-4-yl)pyrrolidine In chloroform at -20℃; for 24h; Catalytic behavior; Reagent/catalyst; Inert atmosphere; regioselective reaction; | 82% |
-
-
97-72-3
2-Methylpropionic anhydride

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| Stage #1: n-octyl β-D-glucopyranoside With 2,4,6-trimethyl-pyridine; (2S,5S)-2,5-bis[[(2S)-3-(1H-indol-3-yl)-1-(octyloxy)-1-oxopropan-2-ylamino]carbonyl]-1-(pyridin-4-yl)pyrrolidine In chloroform; dimethyl sulfoxide at -20℃; Stage #2: 2-Methylpropionic anhydride In chloroform; dimethyl sulfoxide at -20℃; for 24h; Overall yield = 63 %; regioselective reaction; | A 78% B n/a C n/a D n/a |

-
-
97-72-3
2-Methylpropionic anhydride

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| Stage #1: n-octyl β-D-glucopyranoside With 2,4,6-trimethyl-pyridine; (2S,5S)-2,5-bis[[(2S)-3-(1H-indol-3-yl)-1-(octyloxy)-1-oxopropan-2-ylamino]carbonyl]-1-(pyridin-4-yl)pyrrolidine In chloroform; dimethyl sulfoxide at -20℃; Stage #2: 2-Methylpropionic anhydride In chloroform; dimethyl sulfoxide at -20℃; for 24h; Overall yield = 67 %; regioselective reaction; | A 78% B n/a C n/a |

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With 2,5-Diisopropylphenol; oxygen In acetonitrile at 60℃; under 760.051 Torr; chemoselective reaction; | 74% |
-
-
112289-38-0
1,2-anhydro-3,4,6-tri-O-benzyl-D-galactopyranose

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With 4-nitrophenylboronic acid In acetonitrile at -20℃; for 24h; Inert atmosphere; regioselective reaction; | 71% |
-
-
74372-90-0, 112289-38-0, 135202-48-1, 148888-66-8, 71696-32-7
1,2-anhydro-3,4,6-tri-O-benzyl-β-D-mannopyranose

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With 4-nitrophenylboronic acid; water In acetonitrile at 20℃; for 22h; Inert atmosphere; regioselective reaction; | A 1% B 70% |

-
-
108-24-7
acetic anhydride

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
29836-26-8
n-octyl β-D-glucopyranoside

-
-
1435740-69-4
octyl 2,3,4-tri-O-acetyl-6-O-tosyl-β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| Stage #1: p-toluenesulfonyl chloride; n-octyl β-D-glucopyranoside With pyridine at -5℃; Stage #2: acetic anhydride at 0 - 23℃; for 5h; | 68% |
| Stage #1: p-toluenesulfonyl chloride; n-octyl β-D-glucopyranoside With pyridine at -5℃; Stage #2: acetic anhydride at 0 - 20℃; for 5h; | 68% |


| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 20℃; for 4h; Inert atmosphere; | 67% |
-
-
60379-01-3
N-(benzyloxycarbonyl)-L-phenylalanine benzyl ester

-
-
29836-26-8
n-octyl β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With subtilisin-CLEC In acetonitrile at 40℃; for 144h; | 65% |
Octyl-beta-D-glucopyranoside Chemical Properties
Product Name: beta-D-Glucopyranoside, octyl (CAS NO.29836-26-8)
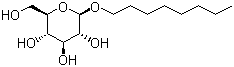
Molecular Formula: C14H28O6
Molecular Weight: 292.37g/mol
Mol File: 29836-26-8.mol
EINECS: 249-887-8
Appearance: white crystalline powder and chunks
Index of Refraction: 1.515
Molar Refractivity: 74.33 cm3
Molar Volume: 246.2 cm3
Surface Tension: 51.5 dyne/cm
Density: 1.18 g/cm3
Flash Point: 228.4 °C
Enthalpy of Vaporization: 82.32 kJ/mol
Boiling Point: 454.1 °C at 760 mmHg
Vapour Pressure: 3.72E-10 mmHg at 25°C
XLogP3-AA: 1.4
H-Bond Donor: 4
H-Bond Acceptor: 6
Structure Descriptors of beta-D-Glucopyranoside, octyl (CAS NO.29836-26-8):
IUPAC Name: (2R,3S,4S,5R,6R)-2-(hydroxymethyl)-6-octoxyoxane-3,4,5-triol
Canonical SMILES: CCCCCCCCOC1C(C(C(C(O1)CO)O)O)O
Isomeric SMILES: CCCCCCCCO[C@H]1[C@@H]([C@H]([C@@H]([C@H](O1)CO)O)O)O
InChI: InChI=1S/C14H28O6/c1-2-3-4-5-6-7-8-19-14-13(18)12(17)11(16)10(9-15)20-14/h10-18H,2-9H2,1H3/t10-,11-,12+,13-,14-/m1/s1
InChIKey: HEGSGKPQLMEBJL-RKQHYHRCSA-N
Product Categories: Biochemistry; Glucose; Glycosides; Sugars; Nonionic Surfactants (for Biochemistry); Surfactants (for Biochemistry)
Octyl-beta-D-glucopyranoside Safety Profile
Safety Information of beta-D-Glucopyranoside, octyl (CAS NO.29836-26-8):
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 24/25
S24/25:Avoid contact with skin and eyes.
WGK Germany: 3
Octyl-beta-D-glucopyranoside Specification
beta-D-Glucopyranoside, octyl , its CAS NO. is 29836-26-8, the synonyms are Octyl-beta-D-glucoside ; 1-O-Octyl-beta-D-glucopyranoside .
Related Products
- Octyl-beta-D-glucopyranoside
- 29838-67-3
- 29840-56-0
- 29840-65-1
- 29840-73-1
- 29841-69-8
- 29843-62-7
- 29843-90-1
- 29843-93-4
- 29844-80-2
- 2984-55-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View