-
Name
Tetrabenzyl pyrophosphate
- EINECS 628-817-3
- CAS No. 990-91-0
- Article Data28
- CAS DataBase
- Density 1.289 g/cm3
- Solubility Slightly soluble in water.
- Melting Point 63-66 °C(lit.)
- Formula C28H28O7P2
- Boiling Point 601.6 °C at 760 mmHg
- Molecular Weight 538.474
- Flash Point 330.4 °C
- Transport Information UN 3261 8/PG 2
- Appearance
- Safety 26-36/37/39-45
- Risk Codes 34
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms Benzylpyrophosphate ([(C7H7O)2PO]2O) (7CI);Diphosphoric acid, tetrakis(phenylmethyl)ester (9CI);Pyrophosphoric acid, tetrabenzyl ester (8CI);Tetrabenzyldiphosphate;Tetrabenzyl pyrophosphate;Tetrabenzylpyrophosphate;
- PSA 99.91000
- LogP 8.08640
Synthetic route
-
-
1623-08-1
phosphoric acid dibenzyl ester

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In Isopropyl acetate at 0 - 6℃; for 1.5h; Inert atmosphere; | 96% |
| With N,N-dicyclohexylurea In toluene at 20℃; for 5h; | 95% |
| With dicyclohexyl-carbodiimide In Isopropyl acetate at 3℃; for 0.916667 - 1.08333h; | 91% |
-
-
17176-77-1
Dibenzyl phosphite

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

| Conditions | Yield |
|---|---|
| With N,N,N,N,-tetramethylethylenediamine; copper(ll) bromide In tetrahydrofuran at 25℃; for 15h; | 90% |
| With tetrachloromethane; N-benzyl-N,N,N-triethylammonium chloride; potassium carbonate In toluene at 60℃; for 0.5h; | 88% |
| With N-chloro-succinimide; benzene anschliessendes Behandeln mit Phosphorsaeure-dibenzylester und Triaethylamin; |
-
-
18826-95-4, 107-88-0
1.3-butanediol

-
-
15205-57-9
tribenzyl phosphite

-
A
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
B
-
538-60-3
dibenzyl hydrogen phosphite

-
C
-
123417-01-6
3-(dibenzyl)phosphoryloxy-1-methyl-1-propanol

| Conditions | Yield |
|---|---|
| Stage #1: tribenzyl phosphite With iodine In dichloromethane at 0 - 20℃; for 0.416667h; Stage #2: 1.3-butanediol With pyridine In dichloromethane at 20℃; for 0.5h; | A n/a B n/a C 65% |

-
-
110-86-1
pyridine

-
-
1623-08-1
phosphoric acid dibenzyl ester

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
71-43-2
benzene

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
110-86-1
pyridine

-
-
1623-08-1
phosphoric acid dibenzyl ester

-
-
98-74-8
4-Nitrobenzenesulfonyl chloride

-
-
71-43-2
benzene

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
56-23-5
tetrachloromethane

-
-
17176-77-1
Dibenzyl phosphite

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
75-62-7
Bromotrichloromethane

-
-
17176-77-1
Dibenzyl phosphite

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

| Conditions | Yield |
|---|---|
| With potassium hydroxide |

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
1623-08-1
phosphoric acid dibenzyl ester

-
-
121-44-8
triethylamine

-
-
75-05-8
acetonitrile

-
-
407-25-0
trifluoroacetic anhydride

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
56-23-5
tetrachloromethane

-
-
17176-77-1
Dibenzyl phosphite

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
75-62-7
Bromotrichloromethane

-
-
17176-77-1
Dibenzyl phosphite

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: benzene; PCl3 / 5 °C / anschliessendes Behandeln mit Wasser 2: aqueous KOH View Scheme | |
| Multi-step reaction with 2 steps 1: benzene; PCl3 / 5 °C / anschliessendes Behandeln mit Wasser 2: aqueous KOH View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: benzene; PCl3 / 5 °C / anschliessendes Behandeln mit Wasser 2: aqueous KOH View Scheme | |
| Multi-step reaction with 2 steps 1: benzene; PCl3 / 5 °C / anschliessendes Behandeln mit Wasser 2: aqueous KOH View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: H3PO4, Et3N, trichloroacetonitrile 2: DCC View Scheme |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
134098-64-9
1-O-(tert-butyldimethylsilyl)-3-(2-hydroxy-4,6-dimethylphenyl)-3,3-dimethylpropan-1-ol

-
-
153910-63-5
dibenzyl (2-(4-((tert-butyldimethylsilyl)oxy)-2-methylbutan-2-yl)-3,5-dimethylphenyl) phosphate

| Conditions | Yield |
|---|---|
| Stage #1: 1-O-(tert-butyldimethylsilyl)-3-(2-hydroxy-4,6-dimethylphenyl)-3,3-dimethylpropan-1-ol With potassium tert-butylate In tetrahydrofuran at 60℃; for 0.0833333h; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran at 60℃; for 1h; | 100% |
| With potassium tert-butylate In tetrahydrofuran at 60℃; for 0.75h; | 87% |
| Stage #1: 1-O-(tert-butyldimethylsilyl)-3-(2-hydroxy-4,6-dimethylphenyl)-3,3-dimethylpropan-1-ol With potassium tert-butylate In tetrahydrofuran at 60℃; for 0.0833333h; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran for 1h; Reflux; | 87.2% |
| With potassium tert-butylate In tetrahydrofuran at 60℃; | 43.8% |
| In N,N-dimethyl-formamide; mineral oil |
-
-
1032939-16-4
1-O-benzyl-2,3-(2,3-dimethoxybut-2,3-diyl)-α-D-xylopyranoside

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
1032939-17-5
1-O-benzyl-2,3-(2,3-dimethoxybut-2,3-diyl)-α-D-xylopyranoside-4-O-dibenzylphosphate

| Conditions | Yield |
|---|---|
| Stage #1: 1-O-benzyl-2,3-(2,3-dimethoxybut-2,3-diyl)-α-D-xylopyranoside With potassium tert-butylate In tetrahydrofuran at 20℃; for 0.333333h; Inert atmosphere; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran at -40 - 20℃; for 3.33333h; | 100% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
550346-02-6
benzyl 2,3-O-[(2R,3R)-2,3-dimethoxybutane-2,3-dioxy]-β-L-arabinopyranoside

-
-
1032939-14-2
1-O-benzyl-2,3-(2,3-dimethoxybut-2,3-diyl)-β-L-arabinopyranoside-4-O-dibenzylphosphate

| Conditions | Yield |
|---|---|
| Stage #1: benzyl 2,3-O-[(2R,3R)-2,3-dimethoxybutane-2,3-dioxy]-β-L-arabinopyranoside With potassium tert-butylate In tetrahydrofuran at 20℃; for 0.333333h; Inert atmosphere; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran at -40 - 20℃; for 3.33333h; | 100% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
81429-51-8
Glycerinaldehyd-diethylacetal-3-triphenylmethylether

-
-
1032939-18-6
2-dibenzylphosphoryl-3-triphenylmethylglyceraldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| Stage #1: Glycerinaldehyd-diethylacetal-3-triphenylmethylether With potassium tert-butylate In tetrahydrofuran at 20℃; for 0.333333h; Inert atmosphere; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran at -40 - 20℃; | 99% |

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
123-08-0
4-hydroxy-benzaldehyde

-
-
156745-33-4
dibenzyl (4-formylphenyl) phosphate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran | 98% |


-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
1092110-69-4
C32H46NO10PSi

| Conditions | Yield |
|---|---|
| Stage #1: 2-acetamido-3-O-acetyl-2-deoxy-4,6-O-(di-tert-butylsilandiyl)-D-glucopyranose With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.0833333h; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran at -78 - 20℃; stereoselective reaction; | 98% |
-
-
10210-17-0
3-(4-hydroxyphenyl)propan-1-ol

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

| Conditions | Yield |
|---|---|
| With tetrahexylammonium iodide; silver(l) oxide In dichloromethane at 20℃; for 20h; | 97% |
-
-
3360-41-6
4-phenyl-butan-1-ol

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
136025-58-6
4-phenylbutyl-dibenzylphosphate

| Conditions | Yield |
|---|---|
| With titanium(IV) tetrabutoxide; N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 16h; Inert atmosphere; | 97% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
1346041-88-0
(1,1-bis-(hydroxymethyl)propyl)carbamic acid benzyl ester

| Conditions | Yield |
|---|---|
| With titanium(IV) tetrabutoxide; N-ethyl-N,N-diisopropylamine In toluene at 20℃; for 16h; Time; Inert atmosphere; chemoselective reaction; | 97% |
| With titanium(IV) tetrabutoxide; N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 16h; Solvent; Inert atmosphere; | 96% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
156745-33-4
dibenzyl (4-formylphenyl) phosphate

| Conditions | Yield |
|---|---|
| Stage #1: 4-hydroxy-benzaldehyde With potassium tert-butylate In tetrahydrofuran at 20℃; for 0.166667h; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran at 70℃; for 1.16667h; | 96% |
-
-
1032939-22-2
1-O-benzyl-2,3-isopropylidene-β-L-ribopyranose

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
1032939-23-3
1-O-benzyl-2,3-isopropylidene-β-L-ribopyranoside-4-O-dibenzylphosphate

| Conditions | Yield |
|---|---|
| Stage #1: 1-O-benzyl-2,3-isopropylidene-β-L-ribopyranose With potassium tert-butylate In tetrahydrofuran at 20℃; for 0.333333h; Inert atmosphere; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran at -40 - 20℃; for 5.66667h; | 96% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

| Conditions | Yield |
|---|---|
| With sodium hydride In dichloromethane at 0 - 20℃; for 0.25h; Schlenk technique; | 96% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
129365-65-7
(1S,2S,3R,4S,5S,6R)-3-Benzyloxy-5-(1-ethoxy-ethoxy)-6-fluoro-cyclohexane-1,2,4-triol

-
-
129365-66-8
Phosphoric acid dibenzyl ester (1S,2S,3R,4S,5S,6S)-2-benzyloxy-3,4-bis-(bis-benzyloxy-phosphoryloxy)-6-(1-ethoxy-ethoxy)-5-fluoro-cyclohexyl ester

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0℃; for 8h; | 95% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
4132-28-9, 4291-69-4, 6386-24-9, 6564-72-3, 59531-24-7, 69257-52-9, 78184-89-1, 78609-16-2, 78609-17-3, 78609-18-4, 96553-53-6, 104111-61-7, 131347-08-5
2,3,4,6-tetra-O-benzyl-D-mannopyranose

-
-
90357-98-5
dibenzyl 2,3,4,6-tetra-O-benzyl-α-D-mannopyranosyl phosphate

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran; hexane; ethylbenzene at -78℃; for 3h; | 95% |
-
-
691409-52-6
N6-methyl-2-(2-phenylethynyl)-2'-deoxyadenosine

-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
691409-55-9
N6-methyl-2-(2-phenylethynyl)-2'-deoxyadenosine-3',5'-bis(dibenzylphosphate)

| Conditions | Yield |
|---|---|
| Stage #1: N6-methyl-2-(2-phenylethynyl)-2'-deoxyadenosine With potassium tert-butylate In tetrahydrofuran at -40℃; for 0.0833333h; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran at -40℃; for 0.5h; | 95% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
1256037-41-8
2-(3-fluorophenyl)-5-hydroxy-6-methoxyquinolin-4-one

-
-
1256037-56-5
2-(3-fluorophenyl)-6-methoxyquinoline-4,5-diyl bis(dibenzylphosphate)

| Conditions | Yield |
|---|---|
| Stage #1: 2-(3-fluorophenyl)-5-hydroxy-6-methoxyquinolin-4-one With sodium hydride In tetrahydrofuran at 0℃; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran for 0.416667h; | 95% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
1334530-12-9
(2-azido-4,6-di-O-benzyl-2,3-dideoxy-3-fluoro-α-D-glucopyranosyl)-1-dibenzylphosphate

| Conditions | Yield |
|---|---|
| Stage #1: 2-azido-4,6-di-O-benzyl-2,3-dideoxy-3-fluoro-α/β-D-glucopyranose With ethylbenzene; lithium diisopropyl amide In tetrahydrofuran; n-heptane at -78℃; for 0.25h; Inert atmosphere; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran; n-heptane at -78 - 0℃; for 3h; Inert atmosphere; | 95% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
1256037-41-8
2-(3-fluorophenyl)-5-hydroxy-6-methoxyquinolin-4-one

-
-
1256037-56-5
2-(3-fluorophenyl)-6-methoxyquinoline-4,5-diyl bis(dibenzylphosphate)

| Conditions | Yield |
|---|---|
| Stage #1: 2-(3-fluorophenyl)-5-hydroxy-6-methoxyquinolin-4-one With sodium hydride In tetrahydrofuran at -1 - 1℃; for 1h; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran for 0.416667h; Product distribution / selectivity; | 95% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
207564-31-6
1,5-anhydro-3,4-di-O-benzyl-2-deoxy-5a-carba-D-arabino-hex-1-enitol

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; | 95% |
-
-
990-91-0
dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate

-
-
262863-48-9
2-chloro-9-[(diethanolamino)carboxymethyl]-6-methylamino-purine

-
-
262863-49-0
9-bis(2-dibenzylphosphatoethylamino)-acetyl-2-chloro-6-methylamino-purine

| Conditions | Yield |
|---|---|
| Stage #1: 2-chloro-9-[(diethanolamino)carboxymethyl]-6-methylamino-purine With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.25h; deprotonation; Stage #2: dibenzyl [[bis(benzyloxy)phosphoryl]oxy]phosphonate In tetrahydrofuran at 0 - 25℃; for 12h; phosphorylation; Further stages.; | 94% |
Tetrabenzyl pyrophosphate Specification
The CAS register number of Tetrabenzyl pyrophosphate is 990-91-0. It also can be called as Pyrophosphoric acid tetrabenzyl ester and the IUPAC name about this chemical is dibenzyl bis(phenylmethoxy)phosphoryl phosphate. It belongs to the following product categories, such as Biochemistry, Nucleosides, Nucleotides & Related Reagents, Phosphorylating and Phosphorothioating Agents, Phosphorylation, Protecting Agents, Phosphorylating Agents & Condensing Agents, Synthetic Organic Chemistry and so on.
Physical properties about Tetrabenzyl pyrophosphate are: (1)ACD/LogP: 6.01; (2)# of Rule of 5 Violations: 2; (3)ACD/LogD (pH 5.5): 6.01; (4)ACD/LogD (pH 7.4): 6.01; (5)ACD/BCF (pH 5.5): 21756.89; (6)ACD/BCF (pH 7.4): 21756.89; (7)ACD/KOC (pH 5.5): 44303.46; (8)ACD/KOC (pH 7.4): 44303.46; (9)#H bond acceptors: 7; (10)#Freely Rotating Bonds: 14; (11)Polar Surface Area: 99.91Å2; (12)Index of Refraction: 1.59; (13)Molar Refractivity: 141 cm3; (14)Molar Volume: 417.4 cm3; (15)Polarizability: 55.89x10-24cm3; (16)Surface Tension: 52.2 dyne/cm; (17)Enthalpy of Vaporization: 86.23 kJ/mol; (18)Boiling Point: 601.6 °C at 760 mmHg; (19)Vapour Pressure: 8.62E-14 mmHg at 25°C.
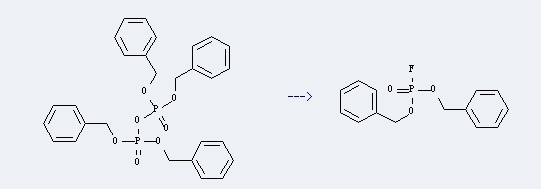
Uses of Tetrabenzyl pyrophosphate: it can be used to produce dibenzyl phosphorofluoridate at Ambient temperature. This reaction will need reagent CsF and solvent acetonitrile. The yield is about 80%.
When you are using this chemical, please be cautious about it as the following:
This chemical can causes burns. When you are using it, wear suitable protective clothing, gloves and eye/face protection. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice and in case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
You can still convert the following datas into molecular structure:
(1)SMILES: O=P(OCc1ccccc1)(OCc2ccccc2)OP(=O)(OCc3ccccc3)OCc4ccccc4
(2)InChI: InChI=1/C28H28O7P2/c29-36(31-21-25-13-5-1-6-14-25,32-22-26-15-7-2-8-16-26)35-37(30,33-23-27-17-9-3-10-18-27)34-24-28-19-11-4-12-20-28/h1-20H,21-24H2
(3)InChIKey: NSBNXCZCLRBQTA-UHFFFAOYAE
(4)Std. InChI: InChI=1S/C28H28O7P2/c29-36(31-21-25-13-5-1-6-14-25,32-22-26-15-7-2-8-16-26)35-37(30,33-23-27-17-9-3-10-18-27)34-24-28-19-11-4-12-20-28/h1-20H,21-24H2
(5)Std. InChIKey: NSBNXCZCLRBQTA-UHFFFAOYSA-N
Related Products
- Tetrabenzyl pyrophosphate
- Tetrabenzylthiuramdisulfide
- Tetrabenzyl-voglibose
- 99-09-2
- 99092-02-1
- 99092-88-3
- 99102-04-2
- 99102-25-7
- 99-10-5
- 99105-77-8
- 99-11-6
- 99-12-7
- 99129-21-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View