-
Name
CARYOPHYLLENE OXIDE
- EINECS 214-519-7
- CAS No. 1139-30-6
- Article Data29
- CAS DataBase
- Density 0.985 g/cm3
- Solubility
- Melting Point 62-63 °C(lit.)
- Formula C15H24O
- Boiling Point 279.682 °C at 760 mmHg
- Molecular Weight 220.355
- Flash Point 119.667 °C
- Transport Information
- Appearance white crystalline powder
- Safety 26
- Risk Codes 36/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Caryophyllene 4b,5a-oxide;trans-Caryophylleneoxide;b-Caryophyllene epoxide;6,7-Epoxy-3(15)-caryophyllene;(-)-Epoxydihydrocaryophyllene;(-)-b-Caryophyllene epoxide;(-)-b-Caryophyllene oxide;4b,5a-Epoxycaryophyllene;5-Oxatricyclo[8.2.0.04,6]dodecane,4,12,12-trimethyl-9-methylene-, [1R-(1R*,4R*,6R*,10S*)]-;Caryophyllene oxide(6CI);Caryophyllene, epoxide (7CI);(-)-Caryophyllene oxide;Caryophyllene 4b,5a-epoxide;
- PSA 12.53000
- LogP 3.93640
Synthetic route
-
-
1779-49-3
Methyltriphenylphosphonium bromide

-
-
10306-22-6, 10306-31-7, 24173-71-5, 34175-71-8
(1R,4R,6R,10S)-4,12,12-trimethyl-5-oxatricyclo[8.2.0.04,6]dodcan-9-one

-
-
1139-30-6
caryophyllene oxide

| Conditions | Yield |
|---|---|
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In tetrahydrofuran at -78 - 0℃; for 0.166667h; Stage #2: (1R,4R,6R,10S)-4,12,12-trimethyl-5-oxatricyclo[8.2.0.04,6]dodcan-9-one In tetrahydrofuran at -78 - 20℃; for 24h; | 74% |

| Conditions | Yield |
|---|---|
| With 4-methyloxetan-2-one; CpLIP2 Y179F (ipase/acyltransferase) from Candida parapsilosis; dihydrogen peroxide In aq. phosphate buffer; water at 20℃; for 24h; pH=6.5; Reagent/catalyst; Enzymatic reaction; | 67% |
| With sodium hydrogencarbonate; 3-chloro-benzenecarboperoxoic acid In dichloromethane at 20℃; for 2h; | 66% |
| With peracetic acid; sodium acetate In dichloromethane | |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0℃; for 0.5h; | 75 % Chromat. |

| Conditions | Yield |
|---|---|
| With chloroform |

| Conditions | Yield |
|---|---|
| With diethyl ether |
-
-
87-44-5
β-caryophyllene

-
A
-
118122-82-0
(1R,4R,5R,9S,11R)-4,5-epoxy-12-nor-8(14)-caryophyllene-11-ol

-
B
-
108525-13-9
(1R,4R,5R,9S,11R)-4,5-epoxy-caryophyll-8(14)-en-12-oic acid

-
C
-
73510-14-2
15-hydroxycaryophyllene oxide

-
D
-
1139-30-6
caryophyllene oxide

| Conditions | Yield |
|---|---|
| for 72h; Diplodia gossypina ATCC 10936; Further byproducts given; | A 220 mg B 900 mg C 300 mg D 65 mg |

-
-
87-44-5
β-caryophyllene

-
A
-
1139-30-6
caryophyllene oxide

-
B
-
103475-43-0
(5S,6S)-5,6-epoxy-5,6-dihydrocaryophyllene

| Conditions | Yield |
|---|---|
| With peracetic acid; sodium acetate In dichloromethane at 0℃; Yield given. Yields of byproduct given; | |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane for 2h; Title compound not separated from byproducts; | A 81 % Spectr. B 19 % Spectr. |
| With peracetic acid; sodium acetate In dichloromethane at 0℃; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |
-
-
87-44-5
β-caryophyllene

-
A
-
1139-30-6
caryophyllene oxide

-
B
-
60444-80-6
(2R,5R,6R)-2,12:5,6-diepoxycaryophyllane

-
C
-
60479-10-9
(1R,4R,6R,9S,10S)-4,12,12-trimethylspiro(5-oxatricyclo[8.2.0.04,6]dodecane-9,2'-oxirane)

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0℃; for 0.5h; Product distribution; effect of stoichiometry, temperature, reaction time; | A 52 % Chromat. B 21 % Chromat. C 21 % Chromat. |

-
-
10577-25-0, 60479-11-0, 60479-16-5, 60479-18-7, 123808-25-3
5,6-epoxy-2-hydroxymethyl-6,10,10-trimethyl-bicyclo[7.2.0]undecan-2-ol

-
-
1139-30-6
caryophyllene oxide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: sodium periodate / water; methanol / 4.33 h / 20 °C / Inert atmosphere 2.1: n-butyllithium / tetrahydrofuran / 0.17 h / -78 - 0 °C 2.2: 24 h / -78 - 20 °C View Scheme |
-
-
1139-30-6
caryophyllene oxide

-
-
19888-34-7, 67737-67-1, 90820-79-4, 108101-44-6
1,5,5,8-tetramethyl-[1R-(1R*,3E,7E,11R*)]-12-oxabicyclo[9.1.0]dodeca-3,7-diene

| Conditions | Yield |
|---|---|
| With phenylsilane In benzene for 2h; | 100% |
| With (+/-)-trans-Co(III)-salen-Cl; phenylsilane In benzene for 2h; | 100% |
| With C32H46ClCoN2O2; phenylsilane In benzene at 22℃; for 5h; Inert atmosphere; | 95% |
-
-
1139-30-6
caryophyllene oxide

| Conditions | Yield |
|---|---|
| With (+/-)-trans-Co(III)-salen-Cl; phenylsilane In benzene for 2h; | 100% |
-
-
1139-30-6
caryophyllene oxide

-
-
10306-22-6, 10306-31-7, 24173-71-5, 34175-71-8
(1R,4R,6R,10S)-4,12,12-trimethyl-5-oxatricyclo[8.2.0.04,6]dodcan-9-one

| Conditions | Yield |
|---|---|
| Stage #1: caryophyllene oxide With ozone In dichloromethane at -78℃; Stage #2: With acetic acid; zinc In dichloromethane; water at 20℃; for 18h; Inert atmosphere; | 98% |
| With ruthenium trichloride; sodium periodate In water; ethyl acetate; acetonitrile for 6h; | 61% |
| With ruthenium trichloride; sodium periodate; water In ethyl acetate; acetonitrile for 6h; | 61% |
-
-
1361382-43-5
6-(trimethylsilyl)hex-5-yn-1-yl 2-cyano-2-diazoacetate

-
-
1139-30-6
caryophyllene oxide

| Conditions | Yield |
|---|---|
| With Rh2(esp)2 In dichloromethane at 22℃; for 4h; Inert atmosphere; diastereoselective reaction; | 94% |
-
-
1139-30-6
caryophyllene oxide

-
-
19431-79-9
1R,5S,9S-11,11-dimethyl-4,8-bismethylenebicyclo<7.2.0>undecan-5-ol

| Conditions | Yield |
|---|---|
| With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane Heating; | 90% |
| With n-butyllithium In diethyl ether | |
| With ethenetetracarbonitrile; lithium bromide In acetone | |
| With ethenetetracarbonitrile; lithium bromide In acetone |
-
-
1139-30-6
caryophyllene oxide

| Conditions | Yield |
|---|---|
| Stage #1: caryophyllene oxide With 9-bora-bicyclo[3.3.1]nonane In tetrahydrofuran for 8h; Inert atmosphere; Cooling with ice; Stage #2: With dihydrogen peroxide; sodium hydroxide In tetrahydrofuran at 20℃; for 2h; | 90% |

| Conditions | Yield |
|---|---|
| With silica gel regioselective reaction; | 89% |
-
-
1139-30-6
caryophyllene oxide

-
-
10577-25-0, 60479-11-0, 60479-16-5, 60479-18-7, 123808-25-3
5,6-epoxy-2-hydroxymethyl-6,10,10-trimethyl-bicyclo[7.2.0]undecan-2-ol

| Conditions | Yield |
|---|---|
| With osmium(VIII) oxide; 4-methylmorpholine N-oxide In water; tert-butyl alcohol at 20℃; for 70h; | 82% |

| Conditions | Yield |
|---|---|
| With formic acid; tributyl-amine; tris[2-phenylpyridinato-C2,N]iridium(III) In acetonitrile at 45℃; for 19h; Inert atmosphere; Irradiation; Sealed tube; diastereoselective reaction; | 71% |
-
-
1139-30-6
caryophyllene oxide

-
A
-
122621-21-0
(1R,2S,5R,8S,9R)-1,4,4,8-tetramethyltricyclo[6.3.0.02,5]undecan-9-ol

-
B
-
122571-86-2
(1S,2S,5R,8R,9R)-1,4,4,8-tetramethyltricyclo[6.3.0.02,5]undecan-9-ol

| Conditions | Yield |
|---|---|
| With 2,4,6-trimethyl-pyridine; manganese; chloro-trimethyl-silane; bis(cyclopentadienyl)titanium dichloride In tetrahydrofuran at 20℃; for 0.666667h; | A 65% B 30% |
-
-
1139-30-6
caryophyllene oxide

-
-
75-66-1
2-methylpropan-2-thiol

-
A
-
2649-64-1
clovanediol

-
C
-
475285-25-7
4,10,10-trimethyl-7-methylene-1β,8α-bicyclo[6.2.0]decene-4-carboxaldehyde

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In dichloromethane Reflux; | A 6% B 63% C 7% |


| Conditions | Yield |
|---|---|
| With ethenetetracarbonitrile at 25℃; | 62% |
-
-
1139-30-6
caryophyllene oxide

-
-
103189-78-2
4,9,12,12-tetramethyl-5-oxatricyclo[8.2.0.04,6]dodecan-9-ol

| Conditions | Yield |
|---|---|
| With iron(III)-acetylacetonate; methyl 4-nitrobenzenesulfonate; phenylsilane; sodium hydrogencarbonate In methanol at 0 - 20℃; for 12h; Schlenk technique; Inert atmosphere; diastereoselective reaction; | 62% |
-
-
1139-30-6
caryophyllene oxide

-
-
92760-17-3
(1R,5S,8R,9R)-4,4,8-trimethylbicyclo<6.3.1.01,5>dodeca-2-en-9-ol

| Conditions | Yield |
|---|---|
| With bithmuth(III) triflate hydrate In dichloromethane for 0.166667h; Wagner-Meerwein rearrangement; Reflux; | 60% |
| With methanol; fluorosulfonylchloride; fluorosulphonic acid 1.) -100 deg C, 2.) diethyl ether; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In chloroform for 5h; Reflux; | A 21% B 25% C 51% |

-
-
1139-30-6
caryophyllene oxide

-
-
75-66-1
2-methylpropan-2-thiol

-
A
-
19431-76-6
caryophylla-2(12),6(13)-dien-5β-ol

-
B
-
2649-64-1
clovanediol

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In dichloromethane Reflux; | A 10% B 7% C 35% D 48% |


| Conditions | Yield |
|---|---|
| With tin(II) trifluoromethanesulfonate at 80℃; for 24h; | A 5% B 47% |

-
-
1139-30-6
caryophyllene oxide

-
A
-
19431-76-6
caryophylla-2(12),6(13)-dien-5β-ol

-
B
-
19431-79-9
1R,5S,9S-11,11-dimethyl-4,8-bismethylenebicyclo<7.2.0>undecan-5-ol

| Conditions | Yield |
|---|---|
| With ethenetetracarbonitrile In dimethyl sulfoxide for 96h; Ambient temperature; | A 46% B 19% C 2% |


| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In chloroform at 20℃; for 2h; | A 46% B 13% C 26% |

-
-
1139-30-6
caryophyllene oxide

| Conditions | Yield |
|---|---|
| at 550℃; under 0.5 Torr; | 45% |
| at 550℃; under 0.1 - 2 Torr; for 0.000277778h; Product distribution; Thermodynamic data; flash vacuum thermolysis; further stereoisomers; variation of temperature; activation energy; | 43% |
-
-
1139-30-6
caryophyllene oxide

-
-
100-53-8
phenylmethanethiol

-
A
-
68263-68-3
4α,11,11-trimethyl-8-methylene-1β,9α-bicyclo[7.2.0]undecan-5-one

-
B
-
2649-64-1
clovanediol

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In chloroform for 5h; Reflux; | A 12% B 26% C 10% D 45% |

-
-
1139-30-6
caryophyllene oxide

-
-
75-66-1
2-methylpropan-2-thiol

-
A
-
68263-68-3
4α,11,11-trimethyl-8-methylene-1β,9α-bicyclo[7.2.0]undecan-5-one

-
B
-
2649-64-1
clovanediol

-
D
-
475285-25-7
4,10,10-trimethyl-7-methylene-1β,8α-bicyclo[6.2.0]decene-4-carboxaldehyde

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In dichloromethane at 20℃; Catalytic behavior; | A 8% B 21% C 44% D 7% |


| Conditions | Yield |
|---|---|
| With ethenetetracarbonitrile at 25℃; | 43% |
-
-
67-56-1
methanol

-
-
1139-30-6
caryophyllene oxide

-
A
-
19431-76-6
caryophylla-2(12),6(13)-dien-5β-ol

-
B
-
127156-29-0
9α-hydroxy-2β-methoxyclovane

| Conditions | Yield |
|---|---|
| With ethenetetracarbonitrile for 24h; Ambient temperature; Further byproducts given; | A 20% B 42% C 11% D 6% |

-
-
67-56-1
methanol

-
-
1139-30-6
caryophyllene oxide

-
A
-
19431-76-6
caryophylla-2(12),6(13)-dien-5β-ol

-
B
-
127156-29-0
9α-hydroxy-2β-methoxyclovane

| Conditions | Yield |
|---|---|
| With ethenetetracarbonitrile for 24h; Ambient temperature; Further byproducts given; | A 20% B 42% C 3% D 6% |

-
-
1139-30-6
caryophyllene oxide

-
A
-
19431-76-6
caryophylla-2(12),6(13)-dien-5β-ol

-
B
-
127156-29-0
9α-hydroxy-2β-methoxyclovane

| Conditions | Yield |
|---|---|
| With ethenetetracarbonitrile In methanol for 24h; Ambient temperature; Further byproducts given; | A 20% B 42% C 11% D 6% |


| Conditions | Yield |
|---|---|
| With tin(II) trifluoromethanesulfonate at 80℃; for 24h; | A 2% B 42% |


| Conditions | Yield |
|---|---|
| With tin(II) trifluoromethanesulfonate at 80℃; for 24h; | 39% |

b-Caryophyllene oxide Chemical Properties
IUPAC Name: (-)-beta-Caryophyllene epoxide
The MF of (-)-beta-Caryophyllene epoxide (1139-30-6) is C15H24O.
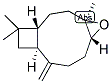
The MW of (-)-beta-Caryophyllene epoxide (1139-30-6) is 220.35.
Synonyms of (-)-beta-Caryophyllene epoxide (1139-30-6): (-)-b-Caryophyllene epoxide ; (-)-beta-Caryophyllene epoxide ; (-)-Epoxydihydrocaryophyllene ; (1R,4R,6R,10S)-4,12,12-Trimethyl-9-methylen-5-oxatricyclo[8.2.0.0~4,6~]dodecan ; 5-Oxatricyclo(8.2.0.0(4,6))dodecane, 4,12,12-trimethyl-9-methylene-, (1R,4R,6R,10S)-
Product Categories: Alphabetical Listings;C-D;Flavors and Fragrances
Form: white crystalline powder
Index of Refraction: 1.507
Storage temp: 2-8 °C
EINECS: 214-519-7
Density: 0.98 g/ml
Flash Point: 119.7 °C
Boiling Point: 279.7 °C
Melting Point: 62-63 °C
FEMA: 4085
b-Caryophyllene oxide Toxicity Data With Reference
| 1. | skn-rbt 500 mg/24H MOD | FCTOD7 Food and Chemical Toxicology. 21 (1983),661. | ||
| 2. | orl-rat LD50:>5 g/kg | FCTOD7 Food and Chemical Toxicology. 21 (1983),661. | ||
| 3. | skn-rbt LD50:>2 g/kg | FCTOD7 Food and Chemical Toxicology. 21 (1983),661. |
b-Caryophyllene oxide Consensus Reports
Reported in EPA TSCA Inventory.
b-Caryophyllene oxide Safety Profile
Low toxicity by ingestion and skin contact. A skin irritant. When heated to decomposition it emits acrid smoke and irritating fumes.Safety information of (-)-beta-Caryophyllene epoxide (1139-30-6):
Hazard Codes ![]() Xi
Xi
Risk Statements
36/38 Irritating to eyes and skin
Safety Statements
26 In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
WGK Germany 2
RTECS RP5530000
F 1-10
Related Products
- b-Caryophyllene oxide
- 113942-30-6
- 113944-46-0
- 1139453-98-7
- 113949-10-3
- 1139-52-2
- 113952-21-9
- 113963-68-1
- 113967-71-8
- 113967-74-1
- 113969-43-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View