Chengdu Biopurify Phytochemicals Ltd.
Chengdu Biopurify Phytochemicals Ltd. is a leading company in the research, development, manufacture and marketing of High Quality Phytochemicals and Extracts(especially Active Ingredients from Traditional Chinese Medicine,Traditional Chinese Medic
Nanjing Spring & Autumn Biological Engineering Co., Ltd.
Nanjing Spring & Autumn Biological Engineering Co., Ltd. Which was founded at 2008, has an R & D team composed very experienced natural products chemists. The company is a high-tech enterprise engaged in functional health care products raw ma
Cas:30220-46-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryDayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:30220-46-3
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:30220-46-3
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryHebei yanxi chemical co.,LTD.
hebei yanxi chemical co., LTD who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and developm
Cas:30220-46-3
Min.Order:1 Metric Ton
FOB Price: $1.0 / 3.0
Type:Manufacturers
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:30220-46-3
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryShanghai Seasonsgreen Chemical Co.,Ltd
Shanghai Seasonsgreen Chemical is a high-tech research and development, production, sale and custom synthesis set in one high-tech chemical products enterprises. Our sales and marketing division is located in Shanghai, serving international pharmaceu
Cas:30220-46-3
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Tianfu Chemical Co., Ltd.
Product Name: INGENOL Synonyms: 1A,2,5,5A,6,9,10,10A OCTAHYDRO-5,5A,6-TRIHYDROXY4(HYDROXYMETHYL)1,1,7,9-TETRAMETHYL1H-2,8 AMETHANOCYCLOPENTA[A]CYCLOPROPA[E]CYCLODECEN-11 ONE;1 ALPHA,2,5,5 ALPHA,6,9,10,10 AL
Hebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Zhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:30220-46-3
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:30220-46-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At prese
Qingdao Beluga Import and Export Co., LTD
Ingenol CAS:30220-46-3 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates, st
Cas:30220-46-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:30220-46-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryLeader Biochemical Group
PRODUCT DETAILS
Cas:30220-46-3
Min.Order:500 Kilogram
FOB Price: $1.0 / 2.0
Type:Lab/Research institutions
inquiryTaizhou Crene Biotechnology co.ltd
Our company provides one-stop services of research - development - production for a variety of special prouducts. Not only do we make effective use of our strong technological strength, but also establish of cooperative relations with several well-
Cas:30220-46-3
Min.Order:100 Milligram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Chemsigma International Co.,Ltd.
bulk production superiority 1,high quality with competitive price in china market 2,fast and safe delivery,goods can be sent out immediately after payment. tracking number is available. 3,our products are with mature technical support. 4.
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:30220-46-3
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
1)quick response within 12 hours; 2)quality guarantee: all products are strictly tested by our qc, confirmed by qa and approved by third party lab in china, usa, canada, germany, uk, italy, france etc. 3) oem/odm available; 4) rea
Cas:30220-46-3
Min.Order:1 Kilogram
FOB Price: $4.0
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Henan Kanbei Chemical Co.,LTD
High quality, competitive price, fast delivery and first-class service we possesses have won the trust and praise of customers. Standard: BP/USP/EP The purity is equal or greater than 99%. As a supplier, we can provide high-quality products. Cle
Shanghai Minstar Chemical Co., Ltd
Ingenol Basic information Description Chemical Properties Ingenol mebutate Pharmacokinetics & metabolism Product Name: Ingenol Synonyms: 1A,2,5,5A,6,9,10,10A OCTAHYDRO-5,5A,6-TRIHYDROXY4(HYDROXYMETHYL)1,1,7,9-TETRAMETHYL1H-2,8 AMETHANOCYCLO
Hubei CuiRan Biotechnology Co., Ltd
Hubei CuiRan Biotechnology Co., Ltd is a leading company in the research, development, manufacture and marketing of High Quality Phytochemicals and Extracts(especially Active Ingredients from Traditional Chinese Medicine,Traditional Chinese Medicine)
Hangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
SAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in custom synthe
Cas:30220-46-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryshanghai Tauto Biotech Co., Ltd
The quality is guaranteed. If you find the product is wrong compared with COA, we promise 100% refund or change product. COA and HPLC will be shipped out with goods. You can also inform your analysis method and we will follow your analysi
Win-Win chemical Co.Ltd
Stock products, own laboratoryAppearance:White to off-white powder Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:30220-46-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
54706-99-9
(1aR,2S,5R,5aS,6S,8aS,9R,10aR)-5,5a,6-trihydroxy-1,1,4,7,9-pentamethyl-1a,2,5,5a,6,9,10,10a-octahydro-1H-2,8a-methanocyclopenta[a]cyclopropa[e][10]annulen-11-one

-
-
30220-46-3
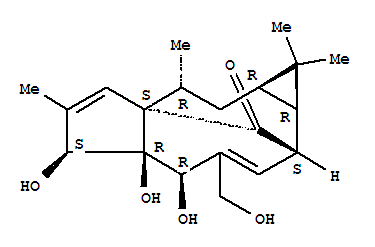
ingenol

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide; formic acid In 1,4-dioxane at 80℃; for 1.25h; | 76% |
| Stage #1: (1aR,2S,5R,5aS,6S,8aS,9R,10aR)-5,5a,6-trihydroxy-1,1,4,7,9-pentamethyl-1a,2,5,5a,6,9,10,10a-octahydro-1H-2,8a-methanocyclopenta[a]cyclopropa[e][10]annulen-11-one With selenium(IV) oxide; formic acid In 1,4-dioxane at 80℃; for 2h; Inert atmosphere; Stage #2: With sodium hydroxide In 1,4-dioxane; water for 0.5h; Inert atmosphere; | 76% |
| With selenium(IV) oxide In 1,4-dioxane; formic acid at 80℃; for 2h; | 70% |
| With selenium(IV) oxide; silica gel In tetrahydrofuran at 80℃; for 2h; | 40% |
-
-
64280-37-1
ingenol monoacetate

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol; water at 0 - 20℃; Temperature; Solvent; Inert atmosphere; | 74% |
-
-
30220-45-2
ingenol 3,5,20-triacetate

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol | 71% |
-
-
77573-43-4
(6S,6aR,7aR,9R,9aS,12S,12aR,12bR)-12,12a-dihydroxy-2,2,7,7,9,11-hexamethyl-4,6,6a,7,7a,8,9,12,12a,12b-decahydro-6,9a-methanocyclopenta[9,10]cyclopropa[5,6]cyclodeca[1,2-d][1,3]dioxin-13-one

-
A
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| With perchloric acid In methanol for 1h; | A 66% B n/a |
-
-
77573-44-5
ingenol-3,4:5,20-diacetonide

-
A
-
30220-46-3
ingenol

-
B
-
77573-43-4
(6S,6aR,7aR,9R,9aS,12S,12aR,12bR)-12,12a-dihydroxy-2,2,7,7,9,11-hexamethyl-4,6,6a,7,7a,8,9,12,12a,12b-decahydro-6,9a-methanocyclopenta[9,10]cyclopropa[5,6]cyclodeca[1,2-d][1,3]dioxin-13-one

| Conditions | Yield |
|---|---|
| With perchloric acid In methanol for 0.75h; Further byproducts given; | A 31% B 3% C 18% |

-
-
49620-09-9
C43H47N3O9

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol |
-
-
42483-56-7
ingenol 3-(2,4,6,8,10)-tetradecapentaenoate

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol |

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran for 0.5h; |

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol for 0.5h; Yield given; |

-
A
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol for 0.666667h; Ambient temperature; Yield given. Yields of byproduct given; |
-
-
91413-76-2
3,4-O-isopropylidene-20-deoxyingenol

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 2: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 21 steps 1.1: 98 percent / KH / tetrahydrofuran / Heating 2.1: 76 percent / Hoveyda-Grubbs catalyst / toluene / 30 h / Heating 3.1: aq. HCl / tetrahydrofuran / 3 h / Heating 4.1: 1.9 g / NaBH4 / ethanol; tetrahydrofuran / 0.25 h / 0 °C 5.1: PPh3; imidazole; I2 / tetrahydrofuran / 3 h / 0 °C 5.2: 94 percent / t-BuOK / tetrahydrofuran; dimethylsulfoxide / 20 °C 6.1: 49 percent / SeO2; aq. AcOH; t-BuOOH / CH2Cl2; decane / 2 h / 20 °C 7.1: 74 percent / Dess-Martin periodinane / CH2Cl2 / 1.5 h / -40 - 10 °C 8.1: 74 percent / aq. RhCl3 / aq. ethanol / 0.58 h / 115 °C 9.1: 94 percent / P(OMe)3; O2; t-BuOK / tetrahydrofuran; 2-methyl-propan-2-ol / 0.75 h / -40 °C 10.1: 73 percent / VO(acac)2; t-BuOOH / benzene; decane / 10 h / 20 °C 11.1: 72 percent / Et3N / CH2Cl2 / 1 h / -10 - -5 °C 12.1: NaBH4 / methanol / 5 h / 20 °C 13.1: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 14.1: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 15.1: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 16.1: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 17.1: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 18.1: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 19.1: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 20.1: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 21.1: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-49-5
C23H32O5

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 2: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 3: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 4: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 5: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 6: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 7: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-41-7
C28H34O4

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 13 steps 1: 94 percent / P(OMe)3; O2; t-BuOK / tetrahydrofuran; 2-methyl-propan-2-ol / 0.75 h / -40 °C 2: 73 percent / VO(acac)2; t-BuOOH / benzene; decane / 10 h / 20 °C 3: 72 percent / Et3N / CH2Cl2 / 1 h / -10 - -5 °C 4: NaBH4 / methanol / 5 h / 20 °C 5: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 6: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 7: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 8: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 9: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 10: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 11: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 12: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 13: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 18 steps 1.1: 1.9 g / NaBH4 / ethanol; tetrahydrofuran / 0.25 h / 0 °C 2.1: PPh3; imidazole; I2 / tetrahydrofuran / 3 h / 0 °C 2.2: 94 percent / t-BuOK / tetrahydrofuran; dimethylsulfoxide / 20 °C 3.1: 49 percent / SeO2; aq. AcOH; t-BuOOH / CH2Cl2; decane / 2 h / 20 °C 4.1: 74 percent / Dess-Martin periodinane / CH2Cl2 / 1.5 h / -40 - 10 °C 5.1: 74 percent / aq. RhCl3 / aq. ethanol / 0.58 h / 115 °C 6.1: 94 percent / P(OMe)3; O2; t-BuOK / tetrahydrofuran; 2-methyl-propan-2-ol / 0.75 h / -40 °C 7.1: 73 percent / VO(acac)2; t-BuOOH / benzene; decane / 10 h / 20 °C 8.1: 72 percent / Et3N / CH2Cl2 / 1 h / -10 - -5 °C 9.1: NaBH4 / methanol / 5 h / 20 °C 10.1: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 11.1: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 12.1: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 13.1: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 14.1: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 15.1: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 16.1: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 17.1: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 18.1: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-50-8
C24H34O7S

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 2: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 3: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 4: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 5: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 6: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-39-3
C28H36O3

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 16 steps 1: 49 percent / SeO2; aq. AcOH; t-BuOOH / CH2Cl2; decane / 2 h / 20 °C 2: 74 percent / Dess-Martin periodinane / CH2Cl2 / 1.5 h / -40 - 10 °C 3: 74 percent / aq. RhCl3 / aq. ethanol / 0.58 h / 115 °C 4: 94 percent / P(OMe)3; O2; t-BuOK / tetrahydrofuran; 2-methyl-propan-2-ol / 0.75 h / -40 °C 5: 73 percent / VO(acac)2; t-BuOOH / benzene; decane / 10 h / 20 °C 6: 72 percent / Et3N / CH2Cl2 / 1 h / -10 - -5 °C 7: NaBH4 / methanol / 5 h / 20 °C 8: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 9: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 10: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 11: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 12: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 13: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 14: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 15: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 16: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-56-4
C28H38O4

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 17 steps 1.1: PPh3; imidazole; I2 / tetrahydrofuran / 3 h / 0 °C 1.2: 94 percent / t-BuOK / tetrahydrofuran; dimethylsulfoxide / 20 °C 2.1: 49 percent / SeO2; aq. AcOH; t-BuOOH / CH2Cl2; decane / 2 h / 20 °C 3.1: 74 percent / Dess-Martin periodinane / CH2Cl2 / 1.5 h / -40 - 10 °C 4.1: 74 percent / aq. RhCl3 / aq. ethanol / 0.58 h / 115 °C 5.1: 94 percent / P(OMe)3; O2; t-BuOK / tetrahydrofuran; 2-methyl-propan-2-ol / 0.75 h / -40 °C 6.1: 73 percent / VO(acac)2; t-BuOOH / benzene; decane / 10 h / 20 °C 7.1: 72 percent / Et3N / CH2Cl2 / 1 h / -10 - -5 °C 8.1: NaBH4 / methanol / 5 h / 20 °C 9.1: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 10.1: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 11.1: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 12.1: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 13.1: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 14.1: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 15.1: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 16.1: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 17.1: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-42-8
C28H34O5

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 12 steps 1: 73 percent / VO(acac)2; t-BuOOH / benzene; decane / 10 h / 20 °C 2: 72 percent / Et3N / CH2Cl2 / 1 h / -10 - -5 °C 3: NaBH4 / methanol / 5 h / 20 °C 4: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 5: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 6: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 7: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 8: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 9: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 10: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 11: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 12: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-51-9
C29H36O4S

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 2: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 3: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 4: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 5: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-40-6
C28H36O4

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 15 steps 1: 74 percent / Dess-Martin periodinane / CH2Cl2 / 1.5 h / -40 - 10 °C 2: 74 percent / aq. RhCl3 / aq. ethanol / 0.58 h / 115 °C 3: 94 percent / P(OMe)3; O2; t-BuOK / tetrahydrofuran; 2-methyl-propan-2-ol / 0.75 h / -40 °C 4: 73 percent / VO(acac)2; t-BuOOH / benzene; decane / 10 h / 20 °C 5: 72 percent / Et3N / CH2Cl2 / 1 h / -10 - -5 °C 6: NaBH4 / methanol / 5 h / 20 °C 7: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 8: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 9: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 10: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 11: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 12: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 13: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 14: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 15: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-57-5
C28H34O4

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 14 steps 1: 74 percent / aq. RhCl3 / aq. ethanol / 0.58 h / 115 °C 2: 94 percent / P(OMe)3; O2; t-BuOK / tetrahydrofuran; 2-methyl-propan-2-ol / 0.75 h / -40 °C 3: 73 percent / VO(acac)2; t-BuOOH / benzene; decane / 10 h / 20 °C 4: 72 percent / Et3N / CH2Cl2 / 1 h / -10 - -5 °C 5: NaBH4 / methanol / 5 h / 20 °C 6: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 7: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 8: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 9: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 10: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 11: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 12: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 13: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 14: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 4 h / Heating 2: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 3: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 4: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-54-2
C29H36O6S

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 2: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 3: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-52-0
C29H36O6S

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 2: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 3: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 4: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-38-2
C30H40O5

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 19 steps 1.1: aq. HCl / tetrahydrofuran / 3 h / Heating 2.1: 1.9 g / NaBH4 / ethanol; tetrahydrofuran / 0.25 h / 0 °C 3.1: PPh3; imidazole; I2 / tetrahydrofuran / 3 h / 0 °C 3.2: 94 percent / t-BuOK / tetrahydrofuran; dimethylsulfoxide / 20 °C 4.1: 49 percent / SeO2; aq. AcOH; t-BuOOH / CH2Cl2; decane / 2 h / 20 °C 5.1: 74 percent / Dess-Martin periodinane / CH2Cl2 / 1.5 h / -40 - 10 °C 6.1: 74 percent / aq. RhCl3 / aq. ethanol / 0.58 h / 115 °C 7.1: 94 percent / P(OMe)3; O2; t-BuOK / tetrahydrofuran; 2-methyl-propan-2-ol / 0.75 h / -40 °C 8.1: 73 percent / VO(acac)2; t-BuOOH / benzene; decane / 10 h / 20 °C 9.1: 72 percent / Et3N / CH2Cl2 / 1 h / -10 - -5 °C 10.1: NaBH4 / methanol / 5 h / 20 °C 11.1: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 12.1: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 13.1: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 14.1: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 15.1: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 16.1: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 17.1: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 18.1: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 19.1: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-46-2
C28H34O6

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1: 72 percent / Et3N / CH2Cl2 / 1 h / -10 - -5 °C 2: NaBH4 / methanol / 5 h / 20 °C 3: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 4: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 5: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 6: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 7: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 8: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 9: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 10: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 11: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |
-
-
827325-48-4
C31H40O6

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 2: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 3: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 4: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 5: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 6: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 7: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 8: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1: 39 mg / PPTS / CH2Cl2 / 0.5 h / Heating 2: 90 percent / 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / CH2Cl2 / 18 h / 20 °C 3: 96 percent / Et3N / CH2Cl2 / 0.75 h / -78 °C 4: 76 percent / Li2CO3 / dimethylformamide / 5 h / 55 °C 5: 97 percent / (NH4)6Mo7O24; aq. H2O2 / aq. ethanol / 6 h / 20 °C 6: 47 percent / 1,8-diazabicyclo[5.4.0]undec-7-ene / benzene / 5 h / Heating 7: 76 percent / Na2HPO4; Na(Hg) / methanol / 0.42 h / -20 - -10 °C 8: 92 percent / aq. HCl / tetrahydrofuran / 5 h / 20 °C 9: 40 percent / SeO2/SiO2 / tetrahydrofuran / 2 h / 80 °C View Scheme |

| Conditions | Yield |
|---|---|
| With pyridine Inert atmosphere; | 96% |
| In pyridine for 24h; | 89% |
| With dmap In pyridine for 16h; Etherification; | 86% |

| Conditions | Yield |
|---|---|
| In pyridine | 95% |
| With pyridine Ambient temperature; Yield given; |
-
-
30220-46-3
ingenol

-
-
18162-48-6
tert-butyldimethylsilyl chloride

| Conditions | Yield |
|---|---|
| With 2,6-dimethylpyridine In N,N-dimethyl-formamide at 20℃; for 1.5h; | 95% |
| In N,N-dimethyl-formamide for 0.75h; | 75% |

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 0 - 20℃; for 14h; Mitsunobu Displacement; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 0 - 20℃; Solvent; Temperature; Reagent/catalyst; Mitsunobu Displacement; Inert atmosphere; | 91% |

| Conditions | Yield |
|---|---|
| In pyridine | 90% |

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 1.5h; | A 87% B 3.62% |

-
-
67-56-1
methanol

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| With perchloric acid at 20℃; for 16h; Addition; rearrangement; | 84% |
-
-
30220-46-3
ingenol

-
-
67-64-1
acetone

-
-
77573-43-4
(6S,6aR,7aR,9R,9aS,12S,12aR,12bR)-12,12a-dihydroxy-2,2,7,7,9,11-hexamethyl-4,6,6a,7,7a,8,9,12,12a,12b-decahydro-6,9a-methanocyclopenta[9,10]cyclopropa[5,6]cyclodeca[1,2-d][1,3]dioxin-13-one

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 20℃; for 1.5h; Product distribution / selectivity; | 81% |
| With toluene-4-sulfonic acid for 0.133333h; | 80% |
| With methanesulfonic acid In tetrahydrofuran at 45℃; | 80% |
-
-
30220-46-3
ingenol

-
-
67-64-1
acetone

-
A
-
77573-43-4
(6S,6aR,7aR,9R,9aS,12S,12aR,12bR)-12,12a-dihydroxy-2,2,7,7,9,11-hexamethyl-4,6,6a,7,7a,8,9,12,12a,12b-decahydro-6,9a-methanocyclopenta[9,10]cyclopropa[5,6]cyclodeca[1,2-d][1,3]dioxin-13-one

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 20℃; for 0.5h; Overall yield = 73 %; | A 73% B 4.5% |


| Conditions | Yield |
|---|---|
| With PS-triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 0℃; for 2h; Substitution; | 67% |
-
-
30220-46-3
ingenol

-
-
103-71-9
phenyl isocyanate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 2h; Acylation; | 51% |
| In dichloromethane; N,N-dimethyl-formamide at 20℃; for 5.33h; | 45.4% |

| Conditions | Yield |
|---|---|
| Stage #1: ingenol; methyl 2-(benzo[d]thiazol-2-ylsulfonyl)acetate With triphenylphosphine In benzene at 0℃; for 0.0833333h; Stage #2: With di-isopropyl azodicarboxylate In benzene at 0 - 20℃; for 12.5h; | 51% |
-
-
30220-46-3
ingenol

-
-
85272-31-7
di-tert-butylsilyl bis(trifluoromethanesulfonate)

-
-
1356187-16-0
ingenol-5,20-(di(tert-butyl)silylene) ether

| Conditions | Yield |
|---|---|
| With 2,6-dimethylpyridine In N,N-dimethyl-formamide at 0 - 20℃; | 50% |
-
-
94487-74-8
(Z)-2-methyl-2-butenoic anhydride

-
-
30220-46-3
ingenol

-
-
1356187-34-2
ingenol 3,20-di-angelate

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 20℃; for 1h; | 48% |
-
-
30220-46-3
ingenol

-
-
18162-48-6
tert-butyldimethylsilyl chloride

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide for 22h; | 47% |
-
-
30220-46-3
ingenol

-
-
111-36-4
n-butyl isocyanide

| Conditions | Yield |
|---|---|
| Stage #1: ingenol; n-butyl isocyanide In dichloromethane; N,N-dimethyl-formamide at 20℃; for 3.66h; Stage #2: With triethylamine In dichloromethane; N,N-dimethyl-formamide at 20℃; for 18h; | 46% |

| Conditions | Yield |
|---|---|
| With dmap; N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In dichloromethane at -18℃; for 72h; Acylation; | A 42% B 18% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In dichloromethane; N,N-dimethyl-formamide at 0 - 20℃; | A 42% B 19% |

-
-
30220-46-3
ingenol

| Conditions | Yield |
|---|---|
| Stage #1: ingenol With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; tetrabutylammomium bromide In dichloromethane at 20℃; for 0.0833333h; pH=8.6; Inert atmosphere; Stage #2: With N-chloro-succinimide In dichloromethane at 20℃; for 12h; pH=8.6; Reagent/catalyst; Solvent; pH-value; Inert atmosphere; | 35% |
-
-
30220-46-3
ingenol

-
-
55239-75-3
N-(4-chlorophenyl)-N-methylcarbamyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: ingenol; N-(4-chlorophenyl)-N-methylcarbamyl chloride In N,N-dimethyl-formamide at 0℃; for 0.25h; Stage #2: With potassium carbonate In N,N-dimethyl-formamide at 0 - 20℃; for 48h; | 19.98% |
-
-
30220-46-3
ingenol

-
-
69034-12-4
2-chloro-5-(trifluoromethyl)pyrimidine

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 20℃; for 39.5h; | 5% |
-
-
30220-46-3
ingenol

-
-
3173-56-6
benzyl isothiocyanate

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 20℃; | 4.7% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

 Xi
Xi