Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:361441-97-6
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:361441-97-6
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:361441-97-6
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
Cas:361441-97-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
(S) - N- Boc- adamantylglycine CAS:361441-97-6 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality o
Cas:361441-97-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:361441-97-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquirySHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:361441-97-6
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:361441-97-6
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:361441-97-6
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Cas:361441-97-6
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
Henan Tianfu Chemical Co., Ltd.
1.Our services:A.Supply sampleB.The packing also can be according the customers` requirmentC.Any inquiries will be replied within 24 hoursD.we provide Commerical Invoice, Packing List, Bill of loading, COA , Health certificate and Origin certificate.
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
BOC Sciences
We are committed to providing our customers with the best products and services at the most competitive prices.Appearance:white powder Storage:Room temperature with sealed well Package:according to the clients requirement Application:Use as primary a
Henan Kanbei Chemical Co.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Cas:361441-97-6
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryAsure Biochem CO.,LTD.
*stable and better quality products*efficient and meticulous servicesAppearance:Powder Storage:Store in dry, cool and ventilated place Package:1kg/tin 5kg/tin 25kg/carton Application:Pharmaceutical raw materials, making injection powder oral agent Tr
Shanghai Chinqesen Biotechnology Co., Ltd.
Good Quality Package:1kg/bag Application:Medical or chemical Transportation:Air/Train/Sea Port:Shenzhen
Chemsigma International Co.,Ltd.
bulk?production Application:Pharmaceutical intermediates
Cas:361441-97-6
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
LEAP CHEM Co., Ltd.
Best Seller, High Quality, Competitive Price, Fast Delivery, Quick ResponseAppearance:powder, or liquid Storage:Stored in room temperature, ventilated place Package:Bottle, barrel, cargo, container, etc. Application:Pharmaceuticals, intermediates, AP
Bide Pharmatech Ltd
Our mission is to provide high-quality and innovative products to our customers. By offering a broad range of products, custom synthesis and personalized services, Bide can help scientists speeding up their research in the chemical and pharmaceutical
Cas:361441-97-6
Min.Order:0
Negotiable
Type:Other
inquiryAmadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Cas:361441-97-6
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryChangzhou Extraordinary Pharmatech co.,LTD
Changzhou Extraordinary Pharmatech co., LTD. As a leading chemical manufacturer and supplier in China.DAS authentication is passed.We can provide the popular precursor chemicals, we have our own strong R & D team, have our own laboratories and fa
Cas:361441-97-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShaanxi Mingqi Chemical Co., Ltd
Known for its best quality and competitve price, this chemicals we offered is widely appreciated by our customers.Our advantages:1, High quality with competitive price:1) Standard:BP/USP/EP/Enterprise standard2) All Purity≥99%3) We are manufacturer a
Cas:361441-97-6
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryChangzhou Pesan Chemical Co.,Ltd
Strong R&D strength and team We have a R&D team with around 30 R&D personnel.All of our employees have chemical backgrounds and have worked in the chemical industry for many years.Our laboratory can synthesize target products with wei
Cas:361441-97-6
Min.Order:1 Kilogram
FOB Price: $1.0 / 9999.0
Type:Other
inquiryENAO Chemical Co, Limited
For quick quotation, please send us the inquiry include CAS#, Structure, Chemical Name, quantity, purity, as well as any additional specifications you require, we will try to get back to you within 24 hours. Our Services Besides manufacturing,
Cas:361441-97-6
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHuBei Ipure Biotech CO.,ltd
HuBei ipure is a diversified product production and operation enterprise, with API, pharma intermediates, and other fine chemicals as well as R&D and pigments development and sales as one of the large enterprises, with more than 130 acres of pl
Cas:361441-97-6
Min.Order:1 Gram
FOB Price: $1.0
Type:Trading Company
inquirySynthetic route
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
102502-64-7
(S)-(+)-(adamant-1-yl)aminoacetic acid hydrochloride

-
-
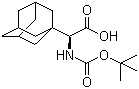
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In DMF (N,N-dimethyl-formamide) at 20℃; for 19h; | 92% |
| With triethylamine In methanol | 80% |
| Stage #1: (S)-(+)-(adamant-1-yl)aminoacetic acid hydrochloride With sodium hydroxide In water at 0 - 5℃; for 0.25h; Stage #2: di-tert-butyl dicarbonate In tetrahydrofuran; water at 0 - 5℃; | 80 g |
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 19h; Inert atmosphere; | 4.07 g |

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With sodium chlorite; potassium dihydrogenphosphate; 2-methyl-but-2-ene In tert-butyl alcohol at 0 - 10℃; for 19.5h; | 82% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
95853-35-3
(S)-(+)-β-amino-1-adamantaneacetic acid

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 19h; | 4.07 g |
-
-
361441-95-4
(S)-2-(adamantan-1-yl)-2-(((R)-2-hydroxy-1-phenylethyl)amino)acetonitrile

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 78 percent / aq. HCl; AcOH / 18 h / 80 °C 2: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 3: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 3 steps 1: hydrogenchloride; water / acetic acid / 80 °C 2: hydrogen; palladium 10% on activated carbon / acetic acid 3: triethylamine / methanol View Scheme | |
| Multi-step reaction with 3 steps 1.1: hydrogenchloride; acetic acid; water / 20 - 95 °C 2.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 3.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 3.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 3 steps 1: hydrogenchloride; water / acetic acid / 18 h / 80 °C 2: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 3: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 2: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 2 steps 1: hydrogen; palladium 10% on activated carbon / acetic acid 2: triethylamine / methanol View Scheme | |
| Multi-step reaction with 2 steps 1.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 2.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 2.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 2 steps 1: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 2: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |
-
-
770-71-8
1-adamantanemethanol

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 98 percent / oxalyl chloride; DMSO; Et3N / CH2Cl2 / 1 h / -78 °C 2: 65 percent / NaHSO3 / H2O; methanol / 16 h / Heating 3: 78 percent / aq. HCl; AcOH / 18 h / 80 °C 4: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 5: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 5 steps 1.1: potassium bromide; sodium hydrogencarbonate / dichloromethane / 0.25 h / 0 - 5 °C 1.2: 0 - 5 °C 2.1: sodium hydrogensulfite / methanol; water / 0 - 55 °C 3.1: hydrogenchloride; acetic acid; water / 20 - 95 °C 4.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 5.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 5.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: oxalyl dichloride; dimethyl sulfoxide / dichloromethane / 1 h / -78 °C / Inert atmosphere 1.2: 0.5 h / -78 °C / Inert atmosphere 2.1: sodium hydrogensulfite / methanol; water / 18 h / 0 - 20 °C / Reflux 3.1: hydrogenchloride; water / acetic acid / 18 h / 80 °C 4.1: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 5.1: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |
-
-
828-51-3
1-Adamantanecarboxylic acid

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 100 percent / diethyl ether; hexane; methanol / 3 h / 20 °C 2: 96 percent / LiAlH4 / tetrahydrofuran / 1.5 h / 0 - 20 °C 3: 98 percent / oxalyl chloride; DMSO; Et3N / CH2Cl2 / 1 h / -78 °C 4: 65 percent / NaHSO3 / H2O; methanol / 16 h / Heating 5: 78 percent / aq. HCl; AcOH / 18 h / 80 °C 6: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 7: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 7 steps 1.1: thionyl chloride / 3 h / 20 °C 2.1: lithium aluminium tetrahydride / tetrahydrofuran / 2 h / 0 - 20 °C / Inert atmosphere 3.1: potassium bromide; sodium hydrogencarbonate / dichloromethane / 0.25 h / 0 - 5 °C 3.2: 0 - 5 °C 4.1: sodium hydrogensulfite / methanol; water / 0 - 55 °C 5.1: hydrogenchloride; acetic acid; water / 20 - 95 °C 6.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 7.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 7.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: methanol; diethyl ether; hexane / 3 h / 20 °C 2.1: lithium aluminium tetrahydride / tetrahydrofuran / 1.5 h / 20 °C / Inert atmosphere 3.1: oxalyl dichloride; dimethyl sulfoxide / dichloromethane / 1 h / -78 °C / Inert atmosphere 3.2: 0.5 h / -78 °C / Inert atmosphere 4.1: sodium hydrogensulfite / methanol; water / 18 h / 0 - 20 °C / Reflux 5.1: hydrogenchloride; water / acetic acid / 18 h / 80 °C 6.1: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 7.1: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |
-
-
2094-74-8
1-Adamantanecarbaldehyde

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 65 percent / NaHSO3 / H2O; methanol / 16 h / Heating 2: 78 percent / aq. HCl; AcOH / 18 h / 80 °C 3: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 4: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 4 steps 1: sodium hydrogensulfite / 0 - 60 °C 2: hydrogenchloride; water / acetic acid / 80 °C 3: hydrogen; palladium 10% on activated carbon / acetic acid 4: triethylamine / methanol View Scheme | |
| Multi-step reaction with 4 steps 1.1: sodium hydrogensulfite / methanol; water / 0 - 55 °C 2.1: hydrogenchloride; acetic acid; water / 20 - 95 °C 3.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 4.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 4.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 4 steps 1: sodium hydrogensulfite / methanol; water / 18 h / 0 - 20 °C / Reflux 2: hydrogenchloride; water / acetic acid / 18 h / 80 °C 3: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 4: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |
-
-
711-01-3
methyl adamantane-1-carboxylate

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 96 percent / LiAlH4 / tetrahydrofuran / 1.5 h / 0 - 20 °C 2: 98 percent / oxalyl chloride; DMSO; Et3N / CH2Cl2 / 1 h / -78 °C 3: 65 percent / NaHSO3 / H2O; methanol / 16 h / Heating 4: 78 percent / aq. HCl; AcOH / 18 h / 80 °C 5: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 6: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 6 steps 1.1: lithium aluminium tetrahydride / tetrahydrofuran / 2 h / 0 - 20 °C / Inert atmosphere 2.1: potassium bromide; sodium hydrogencarbonate / dichloromethane / 0.25 h / 0 - 5 °C 2.2: 0 - 5 °C 3.1: sodium hydrogensulfite / methanol; water / 0 - 55 °C 4.1: hydrogenchloride; acetic acid; water / 20 - 95 °C 5.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 6.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 6.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: lithium aluminium tetrahydride / tetrahydrofuran / 1.5 h / 20 °C / Inert atmosphere 2.1: oxalyl dichloride; dimethyl sulfoxide / dichloromethane / 1 h / -78 °C / Inert atmosphere 2.2: 0.5 h / -78 °C / Inert atmosphere 3.1: sodium hydrogensulfite / methanol; water / 18 h / 0 - 20 °C / Reflux 4.1: hydrogenchloride; water / acetic acid / 18 h / 80 °C 5.1: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 6.1: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate In N,N-dimethyl-formamide at 0 - 20℃; | 93% |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

-
-
862784-24-5
[(S)-1-Adamantan-1-yl-2-((1S,3S,5S)-3-carbamoyl-2-aza-bicyclo[3.1.0]hex-2-yl)-2-oxo-ethyl]-carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With TEA; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 20℃; | 90% |
-
-
66025-93-2
(S)-1-(((R)-1-amino-5-(benzyloxy)-1,5-dioxopentan-2-yl)amino)-1-oxopropan-2-aminium 2,2,2-trifluoroacetate

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Stage #1: (alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 0℃; for 0.5h; Stage #2: (S)-1-(((R)-1-amino-5-(benzyloxy)-1,5-dioxopentan-2-yl)amino)-1-oxopropan-2-aminium 2,2,2-trifluoroacetate With triethylamine In 1,4-dioxane; dichloromethane at 0 - 20℃; for 49h; | 82% |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

-
A
-
361442-00-4
(αS)-α-[[(1,1-dimethylethoxy)carbonyl]-amino]-3-hydroxytricyclo[3.3.1.13,7]decane-1-acetic acid

-
B
-
681282-72-4
(S)-2-((tert-butoxycarbonyl)amino)-2-(3,5-dihydroxyadamantan-1-yl)acetic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide; potassium permanganate at 60 - 90℃; for 1.5h; | A 51% B 17% |
| With potassium hydroxide; potassium permanganate In water at 60 - 90℃; for 1.5h; | A 51% B 17% |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

-
-
361442-00-4
(αS)-α-[[(1,1-dimethylethoxy)carbonyl]-amino]-3-hydroxytricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With potassium permanganate; water; potassium hydroxide at 60 - 90℃; for 1.5h; | 51% |
| With potassium permanganate; potassium hydroxide at 90℃; | 49% |
| With potassium permanganate; tetrabutylammomium bromide; potassium hydroxide In water at 20 - 25℃; | 18.6 g |
-
-
1262389-74-1
C20-O-trityl-prostratin-ol

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 14h; | 24.8% |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid


| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane for 2.5h; |
-
-
10065-72-2
L-Alanine methyl ester

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

-
-
882034-92-6
(S)-2-((S)-2-Adamantan-1-yl-2-tert-butoxycarbonylamino-acetylamino)-propionic acid methyl ester

| Conditions | Yield |
|---|---|
| With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane |
-
-
24426-40-2
N-(cyanomethyl)ethylamine

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane |
-
-
54243-43-5
N-propene-2 aminoacetonitrile

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane |
-
-
16728-81-7
propylamino acetonitrile

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane |
-
-
13257-67-5
2-amino-2-methylpropionic acid methyl ester

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

-
-
882034-93-7
2-(2-adamantan-1-yl-2-tert-butoxycarbonylamino-acetylamino)-2-methyl-propionic acid methyl ester

| Conditions | Yield |
|---|---|
| With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane |
-
-
3010-05-7
benzylaminoacetonitrile

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane |
-
-
30858-68-5
(cyclopropylamino)acetonitrile

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane |
-
-
540-61-4
2-aminoacetonitrile

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane |
-
-
5616-32-0
(methylamino)acetonitrile

-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: TFA / CH2Cl2 View Scheme |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: TFA / CH2Cl2 View Scheme |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: aq. NaOH / methanol / 50 °C 3: NMO; i-BuOCOCl; NH3 / -15 °C 4: TFAA / CH2Cl2 5: TFA / CH2Cl2 View Scheme |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: aq. NaOH / methanol / 50 °C 3: NMO; i-BuOCOCl; NH3 / -15 °C 4: TFAA / CH2Cl2 5: TFA / CH2Cl2 View Scheme |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: TFA / CH2Cl2 View Scheme |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: TFA / CH2Cl2 View Scheme |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: TFA / CH2Cl2 View Scheme |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: TFA / CH2Cl2 View Scheme |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: TFA / CH2Cl2 View Scheme |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: aq. NaOH / methanol / 50 °C 3: NMO; i-BuOCOCl; NH3 / -15 °C 4: TFAA / CH2Cl2 View Scheme |
-
-
361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: EDAC; HOAt; Et3N / CH2Cl2 2: aq. NaOH / methanol / 50 °C 3: NMO; i-BuOCOCl; NH3 / -15 °C 4: TFAA / CH2Cl2 View Scheme |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View