Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:5578-73-4
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryshanghai Tauto Biotech Co., Ltd
17-year experience in Phytochemicals Each product has passed very strict tests (NMR\MS\HPLC) Agents in 13 countries Certified by ISO 9001:2008 quality management system Leader Supplier of Botanical Reference Materials in China
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:5578-73-4
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryShanghai Seasonsgreen Chemical Co.,Ltd
Shanghai Seasonsgreen Chemical is a high-tech research and development, production, sale and custom synthesis set in one high-tech chemical products enterprises. Our sales and marketing division is located in Shanghai, serving international pharmaceu
Cas:5578-73-4
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:5578-73-4
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:5578-73-4
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryChengdu Biopurify Phytochemicals Ltd.
Chengdu Biopurify Phytochemicals Ltd. is a leading company in the research, development, manufacture and marketing of High Quality Phytochemicals and Extracts(especially Active Ingredients from Traditional Chinese Medicine,Traditional Chinese Medic
Cas:5578-73-4
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryNanjing Spring & Autumn Biological Engineering Co., Ltd.
Nanjing Spring & Autumn Biological Engineering Co., Ltd. Which was founded at 2008, has an R & D team composed very experienced natural products chemists. The company is a high-tech enterprise engaged in functional health care products raw ma
Cas:5578-73-4
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:5578-73-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryShaanxi Cuicheng Biomedical Technology Co., Ltd.
Why Choose Us: 1. Factory direct sales, so we can provide the competitive price and high quality product base on 8 years of production and R&D experience. 2. It is available in stock for quick shipment.Products could be packaged according to cu
Henan Wentao Chemical Product Co., Ltd.
1)quick response within 12 hours; 2)quality guarantee: all products are strictly tested by our qc, confirmed by qa and approved by third party lab in china, usa, canada, germany, uk, italy, france etc. 3) oem/odm available; 4) rea
Cas:5578-73-4
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Hubei CuiRan Biotechnology Co., Ltd
Hubei CuiRan Biotechnology Co., Ltd is a leading company in the research, development, manufacture and marketing of High Quality Phytochemicals and Extracts(especially Active Ingredients from Traditional Chinese Medicine,Traditional Chinese Medicine)
Cas:5578-73-4
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:5578-73-4
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquirySinoway Industrial Co., Ltd.
Why is SINOWAY:1) Specialized in pharmaceutical and healthcare industrial since 19872) ISO 9001:2015 & SGS audited supplier . 3) Accept various payment terms : T.T 30-60 days.4) We have warehouse in USA with quickly shipment . Application:Herbal extr
Cas:5578-73-4
Min.Order:0
Negotiable
Type:Trading Company
inquiryShanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:5578-73-4
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Chengdu Lemeitian Pharmaceutical Technology Co,. Ltd
1.High quality and competetive price1)We are manufacturer with own lab and factory, can provide high quality products with factory price. 2)Products purity is test by our quality department strictly before sending, and you can also ask the third test
Cas:5578-73-4
Min.Order:0
Negotiable
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
best price high quality Storage:Room Temperature Application:For pharmacy chemicals Transportation:by sea, air
Antimex Chemical Limied
Our own factory produces direct sales with absolute price advantage Application:Pharmaceutical industry Transportation:By sea Port:Shanghai/tianjin
Cas:5578-73-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGuangdong Juda Chemical Industrial Co.,Limited
Appearance:solid or liquid Storage:sealed in cool and dry place Package:As customer's requested Application:Pharma Intermediate Transportation:by courier/air/sea Port:Any port in China
Cas:5578-73-4
Min.Order:0
Negotiable
Type:Trading Company
inquiryHangzhou Fandachem Co.,Ltd
Sanguinarium chloride cas 5578-73-4Appearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:intermediate Transportation:by air, by sea, by express
DB BIOTECH CO., LTD
SANGUINARINE CHLORIDEAppearance:fine powder Storage:cool, dry place and avoid strongl light Package:1kg/bag, 25kg/drum Application:health supplement Transportation:DHL,TNT,FEDEX,EMS, by air, by sea
Asure Biochem CO.,LTD.
*stable and better quality products*efficient and meticulous servicesAppearance:Powder Storage:Store in dry, cool and ventilated place Package:1kg/tin 5kg/tin 25kg/carton Application:Use in healthcare products. Use in food products. Use in Cosmetic p
BOC Sciences
BOC Sciences provides a wide range of research chemicals and biochemicals including inhibitors, building blocks, GMP Products, impurities and metabolites, APIs for Veterinary, Natural Compounds, ADCs, Stem Cell Molecule and chiral compounds.Appearanc
Shanghai Yuanye Bio-Technology Co., Ltd.
good quality, competitive price, thoughtful after sale serviceAppearance:white powder Storage:Keep it in dry,shady and cool place Package:20mg Application:Pharma;Industry;Agricultural;chemical reaserch Transportation:by express or by sea Port:Any por
ZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
LEAP CHEM Co., Ltd.
Best Seller, High Quality, Competitive Price, Fast Delivery, Quick ResponseAppearance:powder, or liquid Storage:Stored in room temperature, ventilated place Package:Bottle, barrel, cargo, container, etc. Application:Pharmaceuticals, intermediates, AP
HENAN SUNLAKE ENTERPRISE CORPORATION
HENAN SUNLAKE ENTERPRISE CORPORATION Our company advantages: 1、The highest quality with the competitive price. 2、Professional human services. 3、The fastest and safest delivery service. 4、The faster and safest delivery service. 5、The hig
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: oxysanguinarine With lithium aluminium tetrahydride In tetrahydrofuran at 20℃; for 0.333333h; Inert atmosphere; Stage #2: With hydrogenchloride In water at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydroxide; jones reagent In acetone at 0℃; for 0.5h; | 85.4% |
| With sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone In benzene | |
| With sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone In water; benzene for 1h; Ambient temperature; | 2.1 g |
| Stage #1: dihydrosanguinarine With 2,3-dicyano-5,6-dichloro-p-benzoquinone; sodium hydroxide In toluene at 20℃; for 2h; Stage #2: With hydrogenchloride | |
| Stage #1: dihydrosanguinarine With 2,3-dicyano-5,6-dichloro-p-benzoquinone; sodium hydroxide In water; toluene at 20℃; for 1h; Stage #2: With hydrogenchloride In chloroform; water at 0℃; |
-
-
22855-18-1, 107651-16-1
C20H17NO4

-
-
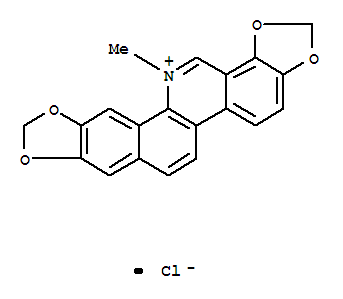
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; palladium on activated charcoal; maleic acid In water; acetic acid | 47% |

| Conditions | Yield |
|---|---|
| (methylation); | |
| Multi-step reaction with 2 steps 1.1: sodium tetrahydroborate / 40 °C 2.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone; sodium hydroxide / toluene; water / 1 h / 20 °C 2.2: 0 °C View Scheme |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water Heating; |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| With iron(III) chloride |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| With mercury(II) diacetate |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 9.8 g / pyridine / 6 h / Ambient temperature 2: HgO / acetic acid / 4 h / Heating 3: 2.1 g / 2,3-dichloro-5,6-dicyano-p-benzoquinone, 5percent NaOH / benzene; H2O / 1 h / Ambient temperature View Scheme |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: HgO / acetic acid / 4 h / Heating 2: 2.1 g / 2,3-dichloro-5,6-dicyano-p-benzoquinone, 5percent NaOH / benzene; H2O / 1 h / Ambient temperature View Scheme |
-
-
22855-17-0
13-methyl-6,7,13,14-tetrahydro-[1,3]dioxolo[4,5-i][1,3]dioxolo[4',5':4,5]benzo[1,2-c]phenanthridine

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Pd-C 2: DDQ, aq. NaOH / benzene View Scheme |

| Conditions | Yield |
|---|---|
| Acidic aq. solution; |
-
-
33842-29-4
methyl 5-iodobenzo[d][1,3]dioxole-4-carboxylate

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: bis-triphenylphosphine-palladium(II) chloride; triethylamine; zinc(II) chloride; zinc / tetrahydrofuran / 24 h / 60 °C / Inert atmosphere 2.1: potassium hydroxide / aacetone / 2 h / Reflux; Inert atmosphere 3.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 2 h / Inert atmosphere; Reflux 4.1: lithium aluminium tetrahydride / tetrahydrofuran / 0.33 h / 20 °C / Inert atmosphere 4.2: 20 °C View Scheme |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: 0.5 h / Inert atmosphere 2.1: bis-triphenylphosphine-palladium(II) chloride; triethylamine; zinc(II) chloride; zinc / tetrahydrofuran / 24 h / 60 °C / Inert atmosphere 3.1: potassium hydroxide / aacetone / 2 h / Reflux; Inert atmosphere 4.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 2 h / Inert atmosphere; Reflux 5.1: lithium aluminium tetrahydride / tetrahydrofuran / 0.33 h / 20 °C / Inert atmosphere 5.2: 20 °C View Scheme |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: potassium hydroxide / aacetone / 2 h / Reflux; Inert atmosphere 2.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 2 h / Inert atmosphere; Reflux 3.1: lithium aluminium tetrahydride / tetrahydrofuran / 0.33 h / 20 °C / Inert atmosphere 3.2: 20 °C View Scheme |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 2 h / Inert atmosphere; Reflux 2.1: lithium aluminium tetrahydride / tetrahydrofuran / 0.33 h / 20 °C / Inert atmosphere 2.2: 20 °C View Scheme |
-
-
1309123-33-8
5-iodobenzo[d][1,3]dioxole-4-carboxylic acid

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: oxalyl dichloride; N,N-dimethyl-formamide / dichloromethane / Inert atmosphere 2.1: 0.5 h / Inert atmosphere 3.1: bis-triphenylphosphine-palladium(II) chloride; triethylamine; zinc(II) chloride; zinc / tetrahydrofuran / 24 h / 60 °C / Inert atmosphere 4.1: potassium hydroxide / aacetone / 2 h / Reflux; Inert atmosphere 5.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 2 h / Inert atmosphere; Reflux 6.1: lithium aluminium tetrahydride / tetrahydrofuran / 0.33 h / 20 °C / Inert atmosphere 6.2: 20 °C View Scheme |
-
-
249636-72-4
5-iodobenzo[d][1,3]dioxole-4-carbaldehyde

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: dihydrogen peroxide; potassium hydroxide / methanol; water / 0.5 h / 65 °C / Inert atmosphere 2.1: oxalyl dichloride; N,N-dimethyl-formamide / dichloromethane / Inert atmosphere 3.1: 0.5 h / Inert atmosphere 4.1: bis-triphenylphosphine-palladium(II) chloride; triethylamine; zinc(II) chloride; zinc / tetrahydrofuran / 24 h / 60 °C / Inert atmosphere 5.1: potassium hydroxide / aacetone / 2 h / Reflux; Inert atmosphere 6.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 2 h / Inert atmosphere; Reflux 7.1: lithium aluminium tetrahydride / tetrahydrofuran / 0.33 h / 20 °C / Inert atmosphere 7.2: 20 °C View Scheme |
-
-
55438-13-6
sanguinarine acetate

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Stage #1: sanguinarine acetate With potassium hydroxide In water Stage #2: With hydrogenchloride In water |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: potassium osmate(VI) dihydrate; 4-methylmorpholine N-oxide / tetrahydrofuran; water; tert-butyl alcohol / 18 h 2: IO4(1-)*Na(1+)*O2Si / dichloromethane / 1 h / Inert atmosphere 3: zinc(II) chloride / diethyl ether; 1,2-dichloro-ethane / 36 h / 55 °C / Inert atmosphere; Microwave irradiation 4: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 5: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 5 steps 1: potassium osmate(VI) dihydrate; 4-methylmorpholine N-oxide / tetrahydrofuran; water; tert-butyl alcohol / 18 h 2: IO4(1-)*Na(1+)*O2Si / dichloromethane / 1 h / Inert atmosphere 3: zinc(II) chloride / dichloromethane; diethyl ether / 18 h / 55 °C / Inert atmosphere; Microwave irradiation 4: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 5: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: cerium(III) chloride / tetrahydrofuran / 0.08 h / -78 °C / Inert atmosphere 1.2: -78 - 20 °C / Inert atmosphere 2.1: toluene-4-sulfonic acid / dichloromethane / 0.5 h / Inert atmosphere 3.1: potassium osmate(VI) dihydrate; 4-methylmorpholine N-oxide / tetrahydrofuran; water; tert-butyl alcohol / 18 h 4.1: IO4(1-)*Na(1+)*O2Si / dichloromethane / 1 h / Inert atmosphere 5.1: zinc(II) chloride / dichloromethane; diethyl ether / 18 h / 55 °C / Inert atmosphere; Microwave irradiation 6.1: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 7.1: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: cerium(III) chloride / tetrahydrofuran / 0.08 h / -78 °C / Inert atmosphere 1.2: -78 - 20 °C / Inert atmosphere 2.1: toluene-4-sulfonic acid / dichloromethane / 0.5 h / Inert atmosphere 3.1: potassium osmate(VI) dihydrate; 4-methylmorpholine N-oxide / tetrahydrofuran; water; tert-butyl alcohol / 18 h 4.1: IO4(1-)*Na(1+)*O2Si / dichloromethane / 1 h / Inert atmosphere 5.1: zinc(II) chloride / diethyl ether; 1,2-dichloro-ethane / 36 h / 55 °C / Inert atmosphere; Microwave irradiation 6.1: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 7.1: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: zinc(II) chloride / dichloromethane; diethyl ether / 18 h / 55 °C / Inert atmosphere; Microwave irradiation 2: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 3: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: zinc(II) chloride / diethyl ether; 1,2-dichloro-ethane / 36 h / 55 °C / Inert atmosphere; Microwave irradiation 2: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 3: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme |
-
-
72744-54-8
5-bromo-benzo[1,3]dioxole-4-carbaldehyde

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: methanol / 3 h / 20 °C / Inert atmosphere 2: sodium tetrahydroborate / methanol / 1 h / 0 - 20 °C 3: zinc(II) chloride / dichloromethane; diethyl ether / 18 h / 55 °C / Inert atmosphere; Microwave irradiation 4: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 5: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 5 steps 1: methanol / 3 h / 20 °C / Inert atmosphere 2: sodium tetrahydroborate / methanol / 1 h / 0 - 20 °C 3: zinc(II) chloride / diethyl ether; 1,2-dichloro-ethane / 36 h / 55 °C / Inert atmosphere; Microwave irradiation 4: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 5: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: sodium tetrahydroborate; methanol / 0.5 h / 0 °C 2.1: pyridine; phosphorus tribromide / dichloromethane / 1 h / 0 - 20 °C 3.1: potassium carbonate / N,N-dimethyl-formamide / 110 °C 4.1: tri-n-butyl-tin hydride; 2,2'-azobis(isobutyronitrile) / toluene / 16 h / 90 - 110 °C / Inert atmosphere 5.1: sodium tetrahydroborate / 40 °C 6.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone; sodium hydroxide / toluene; water / 1 h / 20 °C 6.2: 0 °C View Scheme |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: zinc(II) chloride / dichloromethane; diethyl ether / 18 h / 55 °C / Inert atmosphere; Microwave irradiation 2: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 3: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: zinc(II) chloride / diethyl ether; 1,2-dichloro-ethane / 36 h / 55 °C / Inert atmosphere; Microwave irradiation 2: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 3: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme |
-
-
1207666-53-2
C9H8BrNO2

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: sodium tetrahydroborate / methanol / 1 h / 0 - 20 °C 2: zinc(II) chloride / dichloromethane; diethyl ether / 18 h / 55 °C / Inert atmosphere; Microwave irradiation 3: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 4: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1: sodium tetrahydroborate / methanol / 1 h / 0 - 20 °C 2: zinc(II) chloride / diethyl ether; 1,2-dichloro-ethane / 36 h / 55 °C / Inert atmosphere; Microwave irradiation 3: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 4: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 2: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: toluene-4-sulfonic acid / dichloromethane / 0.5 h / Inert atmosphere 2: potassium osmate(VI) dihydrate; 4-methylmorpholine N-oxide / tetrahydrofuran; water; tert-butyl alcohol / 18 h 3: IO4(1-)*Na(1+)*O2Si / dichloromethane / 1 h / Inert atmosphere 4: zinc(II) chloride / diethyl ether; 1,2-dichloro-ethane / 36 h / 55 °C / Inert atmosphere; Microwave irradiation 5: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 6: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 6 steps 1: toluene-4-sulfonic acid / dichloromethane / 0.5 h / Inert atmosphere 2: potassium osmate(VI) dihydrate; 4-methylmorpholine N-oxide / tetrahydrofuran; water; tert-butyl alcohol / 18 h 3: IO4(1-)*Na(1+)*O2Si / dichloromethane / 1 h / Inert atmosphere 4: zinc(II) chloride / dichloromethane; diethyl ether / 18 h / 55 °C / Inert atmosphere; Microwave irradiation 5: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 6: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme |

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: IO4(1-)*Na(1+)*O2Si / dichloromethane / 1 h / Inert atmosphere 2: zinc(II) chloride / dichloromethane; diethyl ether / 18 h / 55 °C / Inert atmosphere; Microwave irradiation 3: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 4: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1: IO4(1-)*Na(1+)*O2Si / dichloromethane / 1 h / Inert atmosphere 2: zinc(II) chloride / diethyl ether; 1,2-dichloro-ethane / 36 h / 55 °C / Inert atmosphere; Microwave irradiation 3: 2.9-dimethyl-1,10-phenanthroline; potassium tert-butylate / benzene / 5 h / 100 °C / Inert atmosphere; Schlenk technique 4: sodium hydroxide; 2,3-dicyano-5,6-dichloro-p-benzoquinone / toluene / 2 h / 20 °C View Scheme |
-
-
142886-75-7
2,3-methylenedioxy-5-nitronaphthalene

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: palladium 10% on activated carbon; hydrogen / methanol / 2 h / 20 °C / 760.05 Torr 2.1: potassium carbonate / N,N-dimethyl-formamide / 110 °C 3.1: tri-n-butyl-tin hydride; 2,2'-azobis(isobutyronitrile) / toluene / 16 h / 90 - 110 °C / Inert atmosphere 4.1: sodium tetrahydroborate / 40 °C 5.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone; sodium hydroxide / toluene; water / 1 h / 20 °C 5.2: 0 °C View Scheme |
-
-
53811-49-7
naphtho[2,3-d][1,3]dioxol-5-amine

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 110 °C 2.1: tri-n-butyl-tin hydride; 2,2'-azobis(isobutyronitrile) / toluene / 16 h / 90 - 110 °C / Inert atmosphere 3.1: sodium tetrahydroborate / 40 °C 4.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone; sodium hydroxide / toluene; water / 1 h / 20 °C 4.2: 0 °C View Scheme |
-
-
93666-99-0
N-(6'-bromo-2',3'-methylenedioxybenzyl)-6,7-methylenedioxy-1-naphthylamine

-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: tri-n-butyl-tin hydride; 2,2'-azobis(isobutyronitrile) / toluene / 16 h / 90 - 110 °C / Inert atmosphere 2.1: sodium tetrahydroborate / 40 °C 3.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone; sodium hydroxide / toluene; water / 1 h / 20 °C 3.2: 0 °C View Scheme |

| Conditions | Yield |
|---|---|
| With copper(II) chloride dihydrate; oxygen at 80℃; for 10h; | 99% |
| With sodium carbonate In water |
-
-
71-23-8
propan-1-ol

-
-
5578-73-4
sanguinarine chloride

-
-
49702-42-3
13-methyl-14-propoxy-13,14-dihydro-[1,3]dioxolo[4,5-i][1,3]dioxolo[4',5':4,5]benzo[1,2-c]phenanthridine

| Conditions | Yield |
|---|---|
| With copper(II) chloride dihydrate; oxygen at 80℃; for 10h; | 99% |
-
-
5578-73-4
sanguinarine chloride

-
-
67-63-0
isopropyl alcohol

-
-
1292824-30-6, 1333222-84-6, 1333222-85-7
C23H21NO5

| Conditions | Yield |
|---|---|
| With copper(II) chloride dihydrate; oxygen at 80℃; for 10h; | 98% |

| Conditions | Yield |
|---|---|
| With copper(II) chloride dihydrate; oxygen at 80℃; for 10h; | 97% |
-
-
67-56-1
methanol

-
-
5578-73-4
sanguinarine chloride

-
-
72401-54-8, 88544-84-7
6-methoxy-5,6-dihydrosanguinarine

| Conditions | Yield |
|---|---|
| With copper(II) chloride dihydrate; oxygen at 80℃; for 10h; | 96% |

| Conditions | Yield |
|---|---|
| In water at 60℃; for 0.5h; | 83% |

| Conditions | Yield |
|---|---|
| With cesium fluoride In acetonitrile at 20℃; | 59% |

| Conditions | Yield |
|---|---|
| With methanol; sodium tetrahydroborate at 20℃; for 0.5h; Darkness; | 55% |
| With sodium tetrahydroborate In methanol at 20℃; for 0.5h; | 36% |
| With 1,4-dihydronicotinamide adenine dinucleotide In water at 25℃; Rate constant; Kinetics; Thermodynamic data; other coenzyme, var. pH, ΔH(excit.), ΔS(excit.); | |
| With sodium tetrahydroborate In methanol |
-
-
124-41-4
sodium methylate

-
-
5578-73-4
sanguinarine chloride

-
-
72401-54-8, 88544-84-7
6-methoxy-5,6-dihydrosanguinarine

| Conditions | Yield |
|---|---|
| In methanol for 6h; pH=10; Reflux; | 51% |
-
-
5578-73-4
sanguinarine chloride

-
-
86-74-8
9H-carbazole

-
-
1452540-22-5
6-(carbazol-1-yl)-5,6-dihydrosanguinarine

| Conditions | Yield |
|---|---|
| Stage #1: 9H-carbazole With sodium hydride In tetrahydrofuran Inert atmosphere; Sonication; Stage #2: sanguinarine chloride In tetrahydrofuran at 20℃; | 45% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; potassium hydroxide; potassium hexacyanoferrate(III) In water at 90℃; for 3h; | 43% |
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0 - 20℃; for 10h; | 29% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile for 10h; Reflux; | 17.4% |

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| With sodium hydroxide | |
| With potassium hydroxide |
-
-
5578-73-4
sanguinarine chloride

-
-
67-64-1
acetone

-
-
37687-34-6, 88588-10-7
(+/-)-6-acetonyldihydrosanguinarine

| Conditions | Yield |
|---|---|
| With sodium carbonate Heating; |

| Conditions | Yield |
|---|---|
| at 207℃; for 0.116667h; |
-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| With sodium carbonate In water |
-
-
5578-73-4
sanguinarine chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 0.12 h / 207 °C 2: 1.3 g / NaBH4 / hexamethylphosphoric acid triamide / 2 h / 50 °C View Scheme |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View