-
Name
2,1,3-Benzothiadiazole-5-carboxylic acid
- EINECS
- CAS No. 16405-98-4
- Article Data6
- CAS DataBase
- Density 1.609 g/cm3
- Solubility
- Melting Point 229 °C
- Formula C7H4N2O2S
- Boiling Point 368.748 °C at 760 mmHg
- Molecular Weight 180.187
- Flash Point 176.813 °C
- Transport Information
- Appearance
- Safety 26-36/37/39-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Benzo[c][1,2,5]thiadiazole-5-carboxylicacid;
- PSA 91.32000
- LogP 1.38950
Synthetic route

-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: methyl benzo-2,1,3-thiadiazole-5-carboxylate With sodium hydroxide; ethanol; water at 20℃; for 3h; Stage #2: With hydrogenchloride In water pH=2; | 92% |
| With pyridine; lithium iodide Heating; |
-
-
71605-72-6
benzo[c][1,2,5]thiadiazole-5-carbaldehyde

-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide; silver nitrate |
-
-
222851-56-1
N-phenylsulfinylamine

-
-
619-05-6
3,4-diaminobenzoic acid

-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

| Conditions | Yield |
|---|---|
| With xylene |
-
-
1457-93-8
5-methylbenzo[c][1,2,5]thiadiazole

-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: N-bromo-succinimide 3: aqueous acetic acid; hexamethylenetetramine 4: AgNO3; aqueous NaOH View Scheme |
-
-
65858-50-6
5-bromomethyl-benzo[1,2,5]thiadiazole

-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 2: aqueous acetic acid; hexamethylenetetramine 3: AgNO3; aqueous NaOH View Scheme |

-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aqueous acetic acid; hexamethylenetetramine 2: AgNO3; aqueous NaOH View Scheme |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
1117-97-1
N,0-dimethylhydroxylamine

-
-
937279-22-6
benzo[1,2,5]thiadiazole-5-carboxylic acid methoxy-methyl-amide

| Conditions | Yield |
|---|---|
| With triethylamine; HATU In dichloromethane at 20℃; for 3h; Inert atmosphere; | 95% |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
198554-72-2
4-(4-methoxybenzyl)-6-piperidin-4-ylpyrimidin-2-amine

-
-
1085916-69-3
4-[1-(2,1,3-benzothiadiazol-5-ylcarbonyl)piperidin-4-yl]-6-(4-methoxybenzyl)pyrimidin-2-amine

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In chloroform at 20℃; | 84% |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
89795-51-7
2,1,3-benzothiadiazol-5-ylmethanol

| Conditions | Yield |
|---|---|
| Stage #1: benzo[1,2,5]thiadiazole-5-carboxylic acid With triethylamine; isobutyl chloroformate In tetrahydrofuran at 0℃; for 0.5h; Stage #2: With sodium tetrahydroborate; water In tetrahydrofuran at 0℃; for 0.5h; | 81% |
| Multi-step reaction with 2 steps 1: DIPEA / tetrahydrofuran / 1.5 h / 0 °C 2: 1.45 g / aq. NaBH4 / tetrahydrofuran / 2 h / 20 °C View Scheme |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
98946-18-0
tert-Butyl 2,2,2-trichloroacetimidate

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In tetrahydrofuran; cyclohexane at 20℃; for 0.5h; | 76% |
-
-
509101-25-1
6,6-difluoro-5-methyl-5-hexenyl methanesulfonate

-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In DMF (N,N-dimethyl-formamide) at 100℃; for 3h; | 64% |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
99620-31-2
benzo[1,2,5]thiadiazole-5-carboxylic acid amide

| Conditions | Yield |
|---|---|
| With thionyl chloride anschliessend Umsetzen mit NH3; |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
541-41-3
chloroformic acid ethyl ester

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0℃; for 1.5h; |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid


| Conditions | Yield |
|---|---|
| With O-(1H-benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 25 - 37℃; for 16h; |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: DIPEA / tetrahydrofuran / 1.5 h / 0 °C 2.1: 1.45 g / aq. NaBH4 / tetrahydrofuran / 2 h / 20 °C 3.1: MeSO2Cl; DIPEA / CH2Cl2 / 0.5 h / 20 °C 3.2: tetrahydrofuran / 1 h / 20 °C View Scheme |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: DIPEA / tetrahydrofuran / 1.5 h / 0 °C 2.1: 1.45 g / aq. NaBH4 / tetrahydrofuran / 2 h / 20 °C 3.1: dimethylformamide; tetrahydrofuran / 2 h / 20 °C 3.2: 16.5 percent / dimethylformamide; tetrahydrofuran View Scheme |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
6638-79-5
N,O-dimethylhydroxylamine*hydrochloride

-
-
937279-22-6
benzo[1,2,5]thiadiazole-5-carboxylic acid methoxy-methyl-amide

| Conditions | Yield |
|---|---|
| With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane; N,N-dimethyl-formamide at 20℃; for 42h; Inert atmosphere; |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
1202376-82-6
5-(benzo[c][1,2,5]thiadiazol-5-yl)-2,2-dimethylfuran-3(2H)-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: triethylamine; HATU / dichloromethane / 3 h / 20 °C / Inert atmosphere 2.1: n-butyllithium / tetrahydrofuran; hexane / 0.67 h / -78 °C / Inert atmosphere 2.2: 3 h / -78 °C 3.1: toluene-4-sulfonic acid / dichloromethane / 2 h / 20 °C 4.1: diethylamine / ethanol / 20 °C View Scheme |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
1202376-83-7
5-(benzo[c][1,2,5]thiadiazol-5-yl)-4-bromo-2,2-dimethylfuran-3(2H)-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: triethylamine; HATU / dichloromethane / 3 h / 20 °C / Inert atmosphere 2.1: n-butyllithium / tetrahydrofuran; hexane / 0.67 h / -78 °C / Inert atmosphere 2.2: 3 h / -78 °C 3.1: toluene-4-sulfonic acid / dichloromethane / 2 h / 20 °C 4.1: diethylamine / ethanol / 20 °C 5.1: N-Bromosuccinimide / chloroform / 2 h / 20 °C View Scheme |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
1202376-81-5
1-(benzo[c][1,2,5]thiadiazol-5-yl)-4-hydroxy-4-methylpent-2-yn-1-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: triethylamine; HATU / dichloromethane / 3 h / 20 °C / Inert atmosphere 2.1: n-butyllithium / tetrahydrofuran; hexane / 0.67 h / -78 °C / Inert atmosphere 2.2: 3 h / -78 °C 3.1: toluene-4-sulfonic acid / dichloromethane / 2 h / 20 °C View Scheme |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
1202376-80-4
1-(benzo[c][1,2,5]thiadiazol-5-yl)-4-methyl-4-(trimethylsilyloxy)pent-2-yn-1-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: triethylamine; HATU / dichloromethane / 3 h / 20 °C / Inert atmosphere 2.1: n-butyllithium / tetrahydrofuran; hexane / 0.67 h / -78 °C / Inert atmosphere 2.2: 3 h / -78 °C View Scheme |
-
-
16405-98-4
benzo[1,2,5]thiadiazole-5-carboxylic acid

-
-
1202368-77-1
5-(benzo[c][1,2,5]thiadiazol-5-yl)-2,2-dimethyl-4-(4-(quinolin-2-ylmethoxy)phenyl)furan-3(2H)-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: triethylamine; HATU / dichloromethane / 3 h / 20 °C / Inert atmosphere 2.1: n-butyllithium / tetrahydrofuran; hexane / 0.67 h / -78 °C / Inert atmosphere 2.2: 3 h / -78 °C 3.1: toluene-4-sulfonic acid / dichloromethane / 2 h / 20 °C 4.1: diethylamine / ethanol / 20 °C 5.1: N-Bromosuccinimide / chloroform / 2 h / 20 °C 6.1: caesium carbonate / (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride / water; toluene / 12 h / Inert atmosphere; Reflux View Scheme |
2,1,3-Benzothiadiazole-5-carboxylic acid Chemical Properties
Molecular Structure of 2,1,3-Benzothiadiazole-5-carboxylic acid (CAS No.16405-98-4):
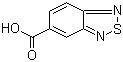
Molecular Formula: C7H4N2O2S
Molecular Weight: 180.1839
CAS No: 16405-98-4
H bond acceptors: 4
H bond donors: 1
Freely Rotating Bonds: 1
Polar Surface Area: 80.32 Å2
Index of Refraction: 1.749
Molar Refractivity: 45.59 cm3
Molar Volume: 112 cm3
Surface Tension: 89.6 dyne/cm
Density: 1.608 g/cm3
Flash Point: 176.8 °C
Enthalpy of Vaporization: 64.93 kJ/mol
Boiling Point: 368.7 °C at 760 mmHg
Vapour Pressure: 4.35E-06 mmHg at 25°C
InChI: InChI=1/C7H4N2O2S/c10-7(11)4-1-2-5-6(3-4)9-12-8-5/h1-3H,(H,10,11)
InChIKey: YHMXJZVGBCACMT-UHFFFAOYAM
Std. InChI: InChI=1S/C7H4N2O2S/c10-7(11)4-1-2-5-6(3-4)9-12-8-5/h1-3H,(H,10,11)
Std. InChIKey: YHMXJZVGBCACMT-UHFFFAOYSA-N
Product Categories: Heterocycles series;API intermediates
2,1,3-Benzothiadiazole-5-carboxylic acid Safety Profile
Hazard Codes: Xi
Xi
Risk Statements:36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements:26-36/37/39-24/25
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
S24/25:Avoid contact with skin and eyes.
Hazard Note:Irritant
HazardClass:IRRITANT
2,1,3-Benzothiadiazole-5-carboxylic acid Specification
2,1,3-Benzothiadiazole-5-carboxylic acid (CAS No.16405-98-4), its synonyms are Timtec-bb sbb007390 ; Rarechem al be 1094 ; 1,2,3-Benzothiadiazole-5-carboxylic acid ; Benzo-2,1,3-thiadiazole-5-carboxylic acid ; Benzo-1,2,3-thiadiazole-5-carboxylic acid ; Benzo[1,2,5]thiadiazole-5-carboxylic acid
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 16405-99-5
- 1640-60-4
- 164062-69-5
- 164071-35-6
- 164081-25-8
- 16408-62-1
- 1640-89-7
- 16409-29-3
- 16409-34-0
- 16409-43-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View