ACHIEVER SYSTEAM BIOCHEM CO., LTD.
We are specialized in custom synthesis, chemical/pharmaceutical/ pesticides outsourcing and contract research. We are committed to provide excellence in researching, manufacturing and drug discovery process. Our research team of scientists co
Cas:163222-32-0
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryDayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:163222-32-0
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:163222-32-0
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:163222-32-0
Min.Order:1
Negotiable
Type:Other
inquiryHenan Allgreen Chemical Co.,Ltd
Good quality Good price Promptly delivery Appearance:white powder Storage:dry dondition Application:intermediate Port:shanghai
Cas:163222-32-0
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:163222-32-0
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:163222-32-0
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our Advantage Rich Experience Our products are sold all over Europe,North&South America, Sino-East, Asia and pacific area as well as Africa,we establish long term. Quality service Company cooperates with research institutes. We strictly con
Cas:163222-32-0
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:163222-32-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:163222-32-0
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:163222-32-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd. established since 2009 , has been always focusing on supplying products and services to our clients in the field of small molecule drug. Huarong Pharm has built platforms for the research, development and manufac
Cas:163222-32-0
Min.Order:1 Gram
FOB Price: $1.0 / 2.0
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
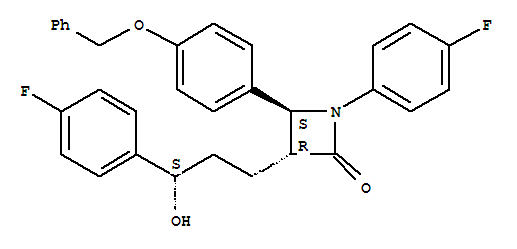
(3R,4S)-4-(4-(Benzyloxy)Phenyl)-1-(4-Fluorophenyl)-3-((S)-3-(4- Fluorophenyl)-3-Hydroxypropyl)Azetidin-2-One CAS:163222-32-0 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development,
Cas:163222-32-0
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:163222-32-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou JINLAN Pharm-Drugs Technology Co., Ltd
Ezetimibe intermediate CAS:163222-32-0 Specification:in-house Stock:Fresh Purity:98% min Application:163222-32-0 Port:FOB China
Cas:163222-32-0
Min.Order:10 Gram
FOB Price: $5.0
Type:Trading Company
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Cas:163222-32-0
Min.Order:0
Negotiable
Type:Manufacturers
inquiryTriumph International Development Limilted
Appearance:Off-White Solid Storage:Store in a cool,dry place and keep away from direct strong light Package:As customer request Application:Used for research and industrial manufacture. Transportation:Common products:Sea/Air/Cou
Cas:163222-32-0
Min.Order:100 Gram
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Cas:163222-32-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:163222-32-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHANWAYS CHEMPHARM CO.,LIMITED
We are concentrating on APIs and pharmaceutical intermediates. It is made obtainable from us at the most reasonable rate. Our Service: 1. Rich experience: Our company is a professional production leading factory in China in pharmaceutical are
Cas:163222-32-0
Min.Order:1 Kilogram
FOB Price: $4.8 / 5.0
Type:Other
inquiryChemlyte Solutions
Chemlyte Solutions believe that customers and suppliers deserve much more than what traditional distributors can offer. To grow in today s fast-paced and increasingly competitive market it is essential to be able to quickly adapt to market forces eff
Cas:163222-32-0
Min.Order:100 Gram
Negotiable
Type:Other
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:163222-32-0
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:163222-32-0
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:163222-32-0
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service and h
Cas:163222-32-0
Min.Order:0
Negotiable
Type:Other
inquirySuzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:Active Pharmaceutical I
Cas:163222-32-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGolden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Cas:163222-32-0
Min.Order:1 Kilogram
Negotiable
Type:Other
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white crystalline powder Storage:stored in a cool, dry and ventilated place to provent sun and rain Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical
Cas:163222-32-0
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Cas:163222-32-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryWuxi TAA Chemical Industry Co.,LTD.
1.A strong technical force and advanced processing equipments. The quality of the products has been strictly inspected and all kinds of index have reached or exceeded domestic and international standards.2. Now we have established long-term stable re
Cas:163222-32-0
Min.Order:0
Negotiable
Type:Other
inquirySynthetic route
-
-
190595-65-4
(3R,4S)-4-[4-(benzyloxy)phenyl]-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3-oxopropyl]azetidin-2-one

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| With formic acid; [(S,S)-teth-TsDpen RuCl]; triethylamine In ethylbenzene at 35 - 40℃; for 24h; Inert atmosphere; | 99.8% |
| With methanesulfonic acid; dimethylsulfide borane complex; (3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole In tetrahydrofuran; methyl tertiarybutylether; toluene at -25 - -20℃; for 2.5 - 3.5h; Product distribution / selectivity; | 98.04% |
| With methanesulfonic acid; dimethylsulfide borane complex; (3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole In tetrahydrofuran; toluene at -25 - -20℃; for 2.5 - 3.5h; Product distribution / selectivity; | 98.6% |
-
-
100-39-0
benzyl bromide

-
-
163222-33-1
ezetemibe

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 20℃; for 3h; | 99% |
-
-
190595-65-4
(3R,4S)-4-[4-(benzyloxy)phenyl]-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3-oxopropyl]azetidin-2-one

-
A
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

-
B
-
163380-15-2
(3R,4S,3'R)-1-(4-fluorophenyl)-4-(4-hydroxyphenyl)-3-[3'-(4-fluorophenyl)-3'-hydroxy-propyl]-azetidin-2-one

| Conditions | Yield |
|---|---|
| With dimethylsulfide borane complex In tetrahydrofuran at 22℃; for 3h; | A 0.141 g B n/a |
| With dimethylsulfide borane complex In tetrahydrofuran at 22℃; for 3h; | |
| Stage #1: (3R,4S)-4-[4-(benzyloxy)phenyl]-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3-oxopropyl]azetidin-2-one With sodium tetrahydroborate; chloro-trimethyl-silane; (R)-α,α-diphenylprolinol In tetrahydrofuran at 19 - 22℃; for 2.78333h; Heating / reflux; Stage #2: With hydrogenchloride; water In tetrahydrofuran; toluene at 2℃; for 0.5h; Product distribution / selectivity; | A n/a B n/a |
| With 1,1,1,3',3',3'-hexafluoro-propanol; C32H39BrMnN2O2P; potassium tert-butylate; hydrogen In methanol at 20℃; under 22502.3 Torr; for 16h; Glovebox; Autoclave; Overall yield = 99 %; enantioselective reaction; | A n/a B n/a |
| With C32H39BrMnN2O2P; potassium tert-butylate; hydrogen In methanol at 20℃; under 22502.3 Torr; for 16h; Glovebox; Autoclave; Optical yield = 85 percent ee; enantioselective reaction; |
-
-
70627-52-0
4-benzyloxybenzylidene-N-(4-fluoro)phenylanisidine

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 64 percent / LDA; DMPU; LiCl / dimethylformamide; tetrahydrofuran / 18 h / -30 - -25 °C 2: 90 percent / NaIO4 / acetonitrile; H2O / 2 h / 20 °C 3: BF3 etherate / toluene / 0.08 h / -30 °C 4: 99 g / p-toluenesulfonic acid monohydrate; molecular sieves / toluene / 4 h / 40 - 50 °C 5: 71 percent / (PPh3)3RhCl; hydrogen / CH2Cl2 / 18 h / 3102.89 Torr 6: 70 percent / CBS; BH3-Me2S / toluene; CH2Cl2 / 2 h / -20 °C View Scheme | |
| Multi-step reaction with 4 steps 1: N-ethyl-N,N-diisopropylamine; titanium(IV) isopropylate; titanium tetrachloride / dichloromethane / 3 h / -5 °C 2: N,O-Bis(trimethylsilyl)acetamide; tetrabutyl ammonium fluoride / toluene / 48 h / Reflux 3: formic acid / dichloromethane / 12 h / Reflux 4: dimethyl sulfide borane / dichloromethane; toluene; tetrahydrofuran / 12 h / 15 °C View Scheme | |
| Multi-step reaction with 2 steps 1: N,O-bis-(trimethylsilyl)-acetamide; tetrabutyl ammonium fluoride / toluene / 60 °C 2: dimethylsulfide borane complex / dichloromethane; toluene / 3 h / -5 - 0 °C / Inert atmosphere View Scheme |
-
-
221349-58-2
(2S,3S)-2-(4-(benzyloxy)phenyl)-1-(4-fluorophenyl)-4-oxazetidine-3-carbaldehyde

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: BF3 etherate / toluene / 0.08 h / -30 °C 2: 99 g / p-toluenesulfonic acid monohydrate; molecular sieves / toluene / 4 h / 40 - 50 °C 3: 71 percent / (PPh3)3RhCl; hydrogen / CH2Cl2 / 18 h / 3102.89 Torr 4: 70 percent / CBS; BH3-Me2S / toluene; CH2Cl2 / 2 h / -20 °C View Scheme |
-
-
221349-56-0
(3S,4S)-4-(4-Benzyloxy-phenyl)-3-((S)-1,2-dihydroxy-ethyl)-1-(4-fluoro-phenyl)-azetidin-2-one

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 90 percent / NaIO4 / acetonitrile; H2O / 2 h / 20 °C 2: BF3 etherate / toluene / 0.08 h / -30 °C 3: 99 g / p-toluenesulfonic acid monohydrate; molecular sieves / toluene / 4 h / 40 - 50 °C 4: 71 percent / (PPh3)3RhCl; hydrogen / CH2Cl2 / 18 h / 3102.89 Torr 5: 70 percent / CBS; BH3-Me2S / toluene; CH2Cl2 / 2 h / -20 °C View Scheme |

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: BF3 etherate / toluene / 0.08 h / -30 °C 2: 99 g / p-toluenesulfonic acid monohydrate; molecular sieves / toluene / 4 h / 40 - 50 °C 3: 71 percent / (PPh3)3RhCl; hydrogen / CH2Cl2 / 18 h / 3102.89 Torr 4: 70 percent / CBS; BH3-Me2S / toluene; CH2Cl2 / 2 h / -20 °C View Scheme |
-
-
221349-60-6
(3R,4S)-4-(4-Benzyloxy-phenyl)-1-(4-fluoro-phenyl)-3-[(E)-3-(4-fluoro-phenyl)-3-oxo-propenyl]-azetidin-2-one

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 71 percent / (PPh3)3RhCl; hydrogen / CH2Cl2 / 18 h / 3102.89 Torr 2: 70 percent / CBS; BH3-Me2S / toluene; CH2Cl2 / 2 h / -20 °C View Scheme |
-
-
231301-00-1
(3S,4S)-4-(4-Benzyloxy-phenyl)-1-(4-fluoro-phenyl)-3-[3-(4-fluoro-phenyl)-1-hydroxy-3-oxo-propyl]-azetidin-2-one

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 99 g / p-toluenesulfonic acid monohydrate; molecular sieves / toluene / 4 h / 40 - 50 °C 2: 71 percent / (PPh3)3RhCl; hydrogen / CH2Cl2 / 18 h / 3102.89 Torr 3: 70 percent / CBS; BH3-Me2S / toluene; CH2Cl2 / 2 h / -20 °C View Scheme |
-
-
219653-96-0
(Z)-1-(4-(benzyloxy)phenyl)-N-(4-fluorophenyl)methaneimine

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: (nBu)3N / heptane; toluene / 80 - 90 °C 2: LiOH*H2O / methanol / 2 h 3: oxalyl chloride / CH2Cl2 / 16 h / 22 °C 4: (Ph3P)4Pd / tetrahydrofuran / 1 h / 10 °C 5: BH3*Me2S / tetrahydrofuran / 3 h / 22 °C View Scheme |
-
-
204589-82-2
(3R,4S)-1-(4-fluorophenyl)-3-(3-hydroxy-3-oxopropyl)-4-(4-benzyloxyphenyl)-2-azetidinone

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: oxalyl chloride / CH2Cl2 / 16 h / 22 °C 2: (Ph3P)4Pd / tetrahydrofuran / 1 h / 10 °C 3: BH3*Me2S / tetrahydrofuran / 3 h / 22 °C View Scheme |
-
-
204589-84-4
3-((3R,4S)-1-(4-fluorophenyl)-2-oxo-4-(4-(benzyloxy)phenyl)azetidin-3-yl)propionic acid chloride

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: (Ph3P)4Pd / tetrahydrofuran / 1 h / 10 °C 2: BH3*Me2S / tetrahydrofuran / 3 h / 22 °C View Scheme |
-
-
204589-80-0
methyl 3-((2S,3R)-2-(4-(benzyloxy)phenyl)-1-(4-fluorophenyl)-4-oxoazetidin-3-yl)propanoate

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: LiOH*H2O / methanol / 2 h 2: oxalyl chloride / CH2Cl2 / 16 h / 22 °C 3: (Ph3P)4Pd / tetrahydrofuran / 1 h / 10 °C 4: BH3*Me2S / tetrahydrofuran / 3 h / 22 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: diethyl aluminiumcholoride / toluene / -12 - 20 °C / Inert atmosphere 2.1: magnesium / tetrahydrofuran / 1 h / 40 - 50 °C / Inert atmosphere 2.2: 1 h / -5 - 0 °C 3.1: [(S,S)-teth-TsDpen RuCl]; triethylamine; formic acid / ethylbenzene / 24 h / 35 - 40 °C / Inert atmosphere View Scheme |
-
-
191330-56-0
[14C]-Sch 57871

-
-
112022-83-0
(3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Stage #1: [14C]-Sch 57871; (3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole In tetrahydrofuran at -20℃; for 0.0833333h; Stage #2: With dimethylsulfide borane complex In tetrahydrofuran at -20℃; for 1.5h; |
-
-
163222-31-9
1-(4-fluorophenyl)-4(S)-(4-hydroxyphenyl)-3(R)-(3-oxo-3-phenylpropyl)-2-azetidinone

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| With dimethylsulfide borane complex; (3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole In tetrahydrofuran at -20℃; for 1.5h; |
-
-
67-56-1
methanol

-
-
190595-65-4
(3R,4S)-4-[4-(benzyloxy)phenyl]-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3-oxopropyl]azetidin-2-one

-
-
112022-83-0
(3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| With hydrogenchloride In tetrahydrofuran |

-
-
460-00-4
1-Bromo-4-fluorobenzene

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: magnesium; Bis<2-(N,N-dimethylamino)aethyl>aether / 0.5 h / 0 °C 2.1: (R)-1,1'-Bi-2-naphthol / titanium(IV) isopropylate / diethyl ether; tetrahydrofuran / 1.5 h / 0 - 25 °C 2.2: -20 - 25 °C 2.3: 0 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: magnesium / tetrahydrofuran / 1 h / 40 - 50 °C / Inert atmosphere 1.2: 1 h / -5 - 0 °C 2.1: [(S,S)-teth-TsDpen RuCl]; triethylamine; formic acid / ethylbenzene / 24 h / 35 - 40 °C / Inert atmosphere View Scheme |
-
-
1412967-16-8
3-[(2S,3R)-2-(4-(benzyloxy)phenyl)-1-(4-fluorophenyl)-4-oxoazetidin-3-yl]propanal

-
-
352-13-6
4-flourophenylmagnesium bromide

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Stage #1: 4-flourophenylmagnesium bromide With (R)-1,1'-Bi-2-naphthol; titanium(IV) isopropylate In tetrahydrofuran; diethyl ether at 0 - 25℃; for 1.5h; Stage #2: 3-[(2S,3R)-2-(4-(benzyloxy)phenyl)-1-(4-fluorophenyl)-4-oxoazetidin-3-yl]propanal In tetrahydrofuran; diethyl ether at -20 - 25℃; Stage #3: With hydrogenchloride; water In tetrahydrofuran; diethyl ether at 0℃; |
-
-
1412967-14-6
(3R,4S)-4-(4-(benzyloxy)phenyl)-1-(4-fluorophenyl)-3-(3-hydroxypropyl)azetidin-2-one

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: sodium hydrogencarbonate; sodium hypochlorite; potassium bromide; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical / Difluoromethane / 25 °C / Cooling with ice 2.1: (R)-1,1'-Bi-2-naphthol / titanium(IV) isopropylate / diethyl ether; tetrahydrofuran / 1.5 h / 0 - 25 °C 2.2: -20 - 25 °C 2.3: 0 °C View Scheme |
-
-
1416263-33-6
(1'R,6R)-3-[(4-benzyloxyphenyl)-(4-fluorophenylamino)-methyl]-6-(4-fluorophenyl)-5,6-dihydro-2H-pyran-2-one

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: lithium hydroxide / tetrahydrofuran; water / 18 h 2: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 3: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme |
-
-
1416263-36-9
(1'S,3R,6S)-3-[(4-benzyloxyphenyl)-(4-fluorophenylamino)-methyl]-6-(4-fluorophenyl)-tetrahydro-2H-pyran-2-one

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| With tert-butylmagnesium chloride In diethyl ether at 0℃; for 2h; |
-
-
1416263-37-0
(3S,3aS,6S,7aS)-3-(4-(benzyloxy)phenyl)-2,6-bis(4-fluorophenyl)tetrahydro-2H-pyrano[3,4-d]isoxazol-4(6H)-one

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 2: Burgess Reagent / toluene / 18 h / 90 °C 3: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 4: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme |

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: cerium(III) chloride heptahydrate; sodium tetrahydroborate / dichloromethane; methanol / 0.5 h / -20 °C 2: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 3: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 4: toluene / 72 h / Reflux 5: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 6: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 7: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 8: Burgess Reagent / toluene / 18 h / 90 °C 9: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 10: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 10 steps 1: cerium(III) chloride heptahydrate; sodium tetrahydroborate / dichloromethane; methanol / 0.5 h / -20 °C 2: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 3: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 4: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 5: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 6: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 7: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 8: Burgess Reagent / toluene / 18 h / 90 °C 9: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 10: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 9 steps 1.1: cerium(III) chloride heptahydrate; sodium tetrahydroborate / dichloromethane; methanol / 0.5 h / -20 °C 2.1: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 3.1: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 4.1: toluene / 72 h / Reflux 5.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 6.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 6.2: 0.25 h / 20 °C 7.1: lithium hydroxide / tetrahydrofuran; water / 18 h 8.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 9.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 9 steps 1.1: cerium(III) chloride heptahydrate; sodium tetrahydroborate / dichloromethane; methanol / 0.5 h / -20 °C 2.1: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 3.1: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 4.1: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 5.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 6.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 6.2: 0.25 h / 20 °C 7.1: lithium hydroxide / tetrahydrofuran; water / 18 h 8.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 9.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme |

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Burgess Reagent / toluene / 18 h / 90 °C 2: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 3: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme |
-
-
1416263-38-1
(6S)-3-((R)-(4-(benzyloxy)phenyl)(4-fluorophenylamino)-methyl)-6-(4-fluorophenyl)-5,6-dihydro-2H-pyran-2-one

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 2: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme |
-
-
459-57-4
4-fluorobenzaldehyde

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: [(R,R)-salen-Cr]BF4 / tert-butyl methyl ether / 24 h / -30 °C / Molecular sieve 1.2: 5 h / 20 °C 2.1: cerium(III) chloride heptahydrate; sodium tetrahydroborate / dichloromethane; methanol / 0.5 h / -20 °C 3.1: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 4.1: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 5.1: toluene / 72 h / Reflux 6.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 7.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 7.2: 0.25 h / 20 °C 8.1: lithium hydroxide / tetrahydrofuran; water / 18 h 9.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 10.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 10 steps 1.1: [(R,R)-salen-Cr]BF4 / tert-butyl methyl ether / 24 h / -30 °C / Molecular sieve 1.2: 5 h / 20 °C 2.1: cerium(III) chloride heptahydrate; sodium tetrahydroborate / dichloromethane; methanol / 0.5 h / -20 °C 3.1: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 4.1: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 5.1: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 6.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 7.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 7.2: 0.25 h / 20 °C 8.1: lithium hydroxide / tetrahydrofuran; water / 18 h 9.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 10.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 11 steps 1.1: [(R,R)-salen-Cr]BF4 / tert-butyl methyl ether / 24 h / -30 °C / Molecular sieve 1.2: 5 h / 20 °C 2.1: cerium(III) chloride heptahydrate; sodium tetrahydroborate / dichloromethane; methanol / 0.5 h / -20 °C 3.1: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 4.1: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 5.1: toluene / 72 h / Reflux 6.1: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 7.1: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 8.1: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 9.1: Burgess Reagent / toluene / 18 h / 90 °C 10.1: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 11.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 11 steps 1.1: [(R,R)-salen-Cr]BF4 / tert-butyl methyl ether / 24 h / -30 °C / Molecular sieve 1.2: 5 h / 20 °C 2.1: cerium(III) chloride heptahydrate; sodium tetrahydroborate / dichloromethane; methanol / 0.5 h / -20 °C 3.1: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 4.1: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 5.1: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 6.1: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 7.1: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 8.1: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 9.1: Burgess Reagent / toluene / 18 h / 90 °C 10.1: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 11.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme |
-
-
1416263-27-8
(2R)-2-(4-fluorophenyl)-3,4-dihydro-2H-pyran-4-ol

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1.1: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 2.1: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 3.1: toluene / 72 h / Reflux 4.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 5.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 5.2: 0.25 h / 20 °C 6.1: lithium hydroxide / tetrahydrofuran; water / 18 h 7.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 8.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 8 steps 1.1: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 2.1: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 3.1: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 4.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 5.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 5.2: 0.25 h / 20 °C 6.1: lithium hydroxide / tetrahydrofuran; water / 18 h 7.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 8.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 9 steps 1: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 2: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 3: toluene / 72 h / Reflux 4: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 5: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 6: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 7: Burgess Reagent / toluene / 18 h / 90 °C 8: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 9: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 9 steps 1: dihydrogen peroxide; molybdenum(VI) oxide / acetonitrile / 24 h 2: pyridine; acetic anhydride / dichloromethane / 2 h / 20 °C 3: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 4: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 5: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 6: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 7: Burgess Reagent / toluene / 18 h / 90 °C 8: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 9: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme |
-
-
123-08-0
4-hydroxy-benzaldehyde

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1.1: potassium carbonate; potassium iodide / tetrahydrofuran / 20 h / Reflux 2.1: methanesulfonic acid / acetone / 2 h / 20 °C 3.1: toluene / 72 h / Reflux 4.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 5.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 5.2: 0.25 h / 20 °C 6.1: lithium hydroxide / tetrahydrofuran; water / 18 h 7.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 8.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 8 steps 1.1: potassium carbonate; potassium iodide / tetrahydrofuran / 20 h / Reflux 2.1: methanesulfonic acid / acetone / 2 h / 20 °C 3.1: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 4.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 5.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 5.2: 0.25 h / 20 °C 6.1: lithium hydroxide / tetrahydrofuran; water / 18 h 7.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 8.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 9 steps 1: potassium carbonate; potassium iodide / tetrahydrofuran / 20 h / Reflux 2: methanesulfonic acid / acetone / 2 h / 20 °C 3: toluene / 72 h / Reflux 4: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 5: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 6: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 7: Burgess Reagent / toluene / 18 h / 90 °C 8: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 9: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 9 steps 1: potassium carbonate; potassium iodide / tetrahydrofuran / 20 h / Reflux 2: methanesulfonic acid / acetone / 2 h / 20 °C 3: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 4: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 5: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 6: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 7: Burgess Reagent / toluene / 18 h / 90 °C 8: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 9: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: potassium carbonate / 12 h / 20 °C 1.2: 4 h / Reflux 2.1: N-ethyl-N,N-diisopropylamine; titanium(IV) isopropylate; titanium tetrachloride / dichloromethane / 3 h / -5 °C 3.1: N,O-Bis(trimethylsilyl)acetamide; tetrabutyl ammonium fluoride / toluene / 48 h / Reflux 4.1: formic acid / dichloromethane / 12 h / Reflux 5.1: dimethyl sulfide borane / dichloromethane; toluene; tetrahydrofuran / 12 h / 15 °C View Scheme |
-
-
100-44-7
benzyl chloride

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1.1: potassium carbonate; potassium iodide / tetrahydrofuran / 20 h / Reflux 2.1: methanesulfonic acid / acetone / 2 h / 20 °C 3.1: toluene / 72 h / Reflux 4.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 5.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 5.2: 0.25 h / 20 °C 6.1: lithium hydroxide / tetrahydrofuran; water / 18 h 7.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 8.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 8 steps 1.1: potassium carbonate; potassium iodide / tetrahydrofuran / 20 h / Reflux 2.1: methanesulfonic acid / acetone / 2 h / 20 °C 3.1: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 4.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 5.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 5.2: 0.25 h / 20 °C 6.1: lithium hydroxide / tetrahydrofuran; water / 18 h 7.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 8.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 9 steps 1: potassium carbonate; potassium iodide / tetrahydrofuran / 20 h / Reflux 2: methanesulfonic acid / acetone / 2 h / 20 °C 3: toluene / 72 h / Reflux 4: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 5: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 6: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 7: Burgess Reagent / toluene / 18 h / 90 °C 8: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 9: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 9 steps 1: potassium carbonate; potassium iodide / tetrahydrofuran / 20 h / Reflux 2: methanesulfonic acid / acetone / 2 h / 20 °C 3: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 4: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 5: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 6: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 7: Burgess Reagent / toluene / 18 h / 90 °C 8: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 9: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme |
-
-
4397-53-9
p-benzyloxybenzaldehyde

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: methanesulfonic acid / acetone / 2 h / 20 °C 2.1: toluene / 72 h / Reflux 3.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 4.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 4.2: 0.25 h / 20 °C 5.1: lithium hydroxide / tetrahydrofuran; water / 18 h 6.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 7.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: methanesulfonic acid / acetone / 2 h / 20 °C 2.1: scandium tris(trifluoromethanesulfonate) / toluene / 72 h / 30 °C / Molecular sieve 3.1: potassium iodide; chloro-trimethyl-silane / acetonitrile / 0.58 h / 20 °C 4.1: chloro-trimethyl-silane; 1H-imidazole / dichloromethane / 0.5 h / 20 °C 4.2: 0.25 h / 20 °C 5.1: lithium hydroxide / tetrahydrofuran; water / 18 h 6.1: triphenylphosphine; di-isopropyl azodicarboxylate / 3 h / 0 °C 7.1: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme | |
| Multi-step reaction with 8 steps 1: methanesulfonic acid / acetone / 2 h / 20 °C 2: toluene / 72 h / Reflux 3: lithium hydroxide monohydrate / tetrahydrofuran; water / 18 h 4: triphenylphosphine; di-isopropyl azodicarboxylate / tetrahydrofuran / 3 h / 0 °C 5: potassium iodide / acetonitrile; water / 0.58 h / 20 °C 6: Burgess Reagent / toluene / 18 h / 90 °C 7: platinum(IV) oxide; hydrogen / toluene / 20 °C / 750.08 Torr 8: tert-butylmagnesium chloride / diethyl ether / 2 h / 0 °C View Scheme |
-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

-
-
163222-33-1
ezetemibe

| Conditions | Yield |
|---|---|
| With 10 wt% Pd(OH)2 on carbon; hydrogen In methanol; cyclohexane at 70℃; under 760.051 Torr; for 3h; | 99% |
| With 5%-palladium/activated carbon; hydrogen In tetrahydrofuran; ethanol at 50 - 55℃; under 37.5038 - 75.0075 Torr; | 90.3% |
| With palladium 10% on activated carbon; ammonium formate; acetic acid for 6h; Reflux; | 90% |
-
-
15761-38-3
L-N-Boc-Ala

-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane Inert atmosphere; | 69% |
-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: H2 / 10percent Pd/C / ethanol / 16 h / 3102.9 Torr 2: p-TsOH*H2O / toluene / 6 h / 80 °C View Scheme |
-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

-
-
163380-20-9
4(S)-(4-acetyloxyphenyl)-3(R)-(3(S)-acetyloxy-3-(4-fluorophenyl)propyl)-1-(4-fluorophenyl)-2-azetidinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: H2 / 10percent Pd/C / ethanol / 16 h / 3102.9 Torr 2: 0.260 g / pyridine / CH2Cl2 / 1 h / 22 °C View Scheme |
-
-
163222-32-0
(3R,4S)-1-(4-fluorophenyl)-3-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4-(4-benzyloxyphenyl)-2-azetidinone

-
A
-
1197811-72-5
EZT-FAM

-
B
-
163222-33-1
ezetemibe

| Conditions | Yield |
|---|---|
| With formic acid; ammonium formate; 5%-palladium/activated carbon In methanol at 20℃; Product distribution / selectivity; Inert atmosphere; |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View