Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:7784-23-8
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:7784-23-8
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryLeader Biochemical Group
About Product Details ALUMINUM IODIDE Chemical Properties
Cas:7784-23-8
Min.Order:1 Kilogram
FOB Price: $3.0 / 10.0
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:7784-23-8
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:white or off-white powder Storage:Store in sealed containers
Cas:7784-23-8
Min.Order:100 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
Cas:7784-23-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Henan Kanbei Chemical Co.,LTD
High quality, competitive price, fast delivery and first-class service we possesses have won the trust and praise of customers. Standard: BP/USP/EP The purity is equal or greater than 99%. As a supplier, we can provide high-quality products. Cle
Cas:7784-23-8
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
ALUMINUM IODIDE Basic information Product Name: ALUMINUM IODIDE Synonyms: ALUMINIUM IODIDE;Aluminum Iodide, Ultra Dry (Metals Basis);uminum iodide;ALUMINUM TRIIODIDE;ALUMINUM IODIDE;AlI3;Aluminum iodide (AlI3);aluminumiodide(ali3) CAS: 7784
Win-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:7784-23-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Hubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
BOC Sciences
We are committed to providing our customers with the best products and services at the most competitive prices.Appearance:clear oily liquid Storage:Room temperature with sealed well Package:according to the clients requirement Application:Use as prim
Asure Biochem CO.,LTD.
*stable and better quality products*efficient and meticulous servicesAppearance:Powder Storage:Store in dry, cool and ventilated place Package:1kg/tin 5kg/tin 25kg/drum 180kg/drum Application:1. Chemical raw materials 2. Catalysts, additives, etc. Tr
Hunan Russell Chemicals Technology Co.,Ltd
low price and high purityAppearance:solid or liquid Storage:in sealed air resistant place Package:As customer require Application:Pharma;Industry;Agricultural Transportation:by sea or by airplane Port:any port in China
ZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
LEAP CHEM Co., Ltd.
Best Seller, High Quality, Competitive Price, Fast Delivery, Quick ResponseAppearance:powder, or liquid Storage:Stored in room temperature, ventilated place Package:Bottle, barrel, cargo, container, etc. Application:Pharmaceuticals, intermediates, AP
HENAN SUNLAKE ENTERPRISE CORPORATION
Tech Data Relevant 1.Certificate Of Analysis (COA) 2..Material Safety Data Sheet (MSDS) 3.Technical Data Sheet(TDS) 4.Route of synthesis (ROS) 5.Nuclear Magnetic Resonance(NMR) 6.Method of Analysis(MOA) Our Service 1. Before Shipmen
Cas:7784-23-8
Min.Order:100 Kilogram
Negotiable
Type:Trading Company
inquiryHangzhou KieRay Chem Co.,Ltd.
Hangzhou KieRaychem Co.,Ltd.is located in Yuhang District of Hangzhou City and specialized in the chemical product customization, development, sales, import and export. Current business is focused on fine chemicals, pharmaceutical materials and inter
Cas:7784-23-8
Min.Order:1 Gram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryMENGNA
The cas register number of Aluminium iodide is 7784-23-8. It also can be called as Aluminum triiodide and the Systematic name about this chemical is triiodoalumane. Physical properties about Aluminium iodide are: (1)#H bond acceptors: 0; (2)#H bond
Cas:7784-23-8
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Share Chemical Co., Ltd
At Share Chemical Company, we scrupulously abide by our policy of “Excellent Quality at a Reasonable Price”. We strive to satisfy all of our customers by providing the finest quality products supported by the finest in customer servi
SHAANXI TOP PHARM CHEMICAL CO.LTD
Appearance:white powder Storage:Ventilating and drying Package:25kg/drum Application:important raw matrtial Transportation:by air/by sea
Strem Chemicals, Inc.
1.High Quality Specialty Chemicals2.Expertise and Experience in Manufacturing High Purity, High Quality Chemicals3.Long Term Focus in Inorganics, Organometallics and Metals4.ISO 9001 CertifiedAppearance:tan to red-brown pwdr.
Synthetic route

| Conditions | Yield |
|---|---|
| In hexane thin foil of Al was suspended in degassed n-hexane under argon, 1.5 equiv. I2 was added, mixt. was boiled under reflux for 1-3 h; filtered into a heated receiver; elem. anal.; | 96% |
| In further solvent(s) sheet aluminium in I2/ethyl iodide soln.;; impurities of I2;; | |
| In neat (no solvent) addn. of Al to molten I2, ignition and melting of the metal;; |
-
-
7553-56-2
iodine

-
-
56-81-5
glycerol

-
-
7429-90-5
aluminium

-
A
-
1333-74-0
hydrogen

-
B
-
7784-23-8
aluminium(III) iodide

| Conditions | Yield |
|---|---|
| In water in glycerine-soln. on heating;; |


| Conditions | Yield |
|---|---|
| In water byproducts: H2, Al(OH)3; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: H2; | |
| In neat (no solvent) byproducts: H2; |

| Conditions | Yield |
|---|---|
| heating till AgI softens, part of AlI3 is sublimated; | |
| heating till AgI softens, part of AlI3 is sublimated; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) mixt. of the starting materials heated in evacuated sealed quartz tubes to 500-900°C; decompn. of educts ocurrs, AlI3 sublimates off; | A 0% B n/a |


| Conditions | Yield |
|---|---|
| In neat (no solvent) mixt. of the starting materials heated in evacuated sealed quartz tubes to 500-900°C; decompn. of educts ocurrs, AlI3 sublimates off; | A 0% B n/a |


| Conditions | Yield |
|---|---|
| In neat (no solvent) formation at reaction of metallic Al with molten SnI4;; |

-
-
7784-23-8
aluminium(III) iodide

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 381°C;; |

| Conditions | Yield |
|---|---|
| With aluminium in stoichiometric ratio of educts, 325°C in closed evacuated tube; removal of AlI3 by sublimation at 250 °C; | |
| With Al in stoichiometric ratio of educts, 325°C in closed evacuated tube; removal of AlI3 by sublimation at 250 °C; |

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) thermal decompn. by heating at 385°C for 48 h in an evacuated sealed glass ampoule; AlI3 sublimates to the cold end of the ampoule; |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: C4H9I; iodide-ion was also formed; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: Al2S3; heating a mixture of molten AlI3 and Bi2S3 in an evacuated tube at 230°C for 48 hours;; sublimation in vacuum at 400°C;; | 100% |
| In neat (no solvent) byproducts: Al2S3; heating a mixture of molten AlI3 and Bi2S3 in an evacuated tube at 230°C for 48 hours;; sublimation in vacuum at 400°C;; | 100% |

| Conditions | Yield |
|---|---|
| In carbon disulfide exclusion of moisture; SbI3 and AlI3 (equimolar ratio) in CS2 are heated under reflux for 2 h; crystn. at room temp., complete at -20°C within 1 d; | 100% |

| Conditions | Yield |
|---|---|
| In benzene under N2, dropwise addn. of thiol soln. to a suspension of AlI3 in benzene, dissoln. of AlI3, stirred for 15 min at room temp.; evapn. of solvent, dried in vac. at room temp. for 3h, elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In benzene under N2, dropwise addn. of thiol soln. to a suspension of AlI3 in benzene, dissoln. of AlI3, stirred for 15 min at room temp.; evapn. of solvent, dried in vac. at room temp. for 3h, elem. anal.; | 100% |
-
-
3908-55-2
Trimethyl(methylthio)silane

-
-
7784-23-8
aluminium(III) iodide

-
-
112908-17-5
aluminium triiodide*trimethylsilyl methyl sulphide

| Conditions | Yield |
|---|---|
| In benzene a benzene soln. of Me3SiSMe was added dropwise under N2 to a suspn. of AlI3 in benzene, mixt. was stirred for 15 min at room temp.; solvent was evapd.; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In benzene under N2, dropwise addn. of thiol soln. to a suspension of AlI3 in benzene, dissoln. of AlI3, stirred for 15 min at room temp.; evapn. of solvent, dried in vac. at room temp. for 3h, elem. anal.; | 100% |
-
-
5573-62-6
(ethylthio)trimethylsilane

-
-
7784-23-8
aluminium(III) iodide

-
-
112908-18-6
aluminium triiodide*trimethylsilyl ethyl sulphide

| Conditions | Yield |
|---|---|
| In benzene a benzene soln. of Me3SiSEt was added dropwise under N2 to a suspn. of AlI3 in benzene, mixt. was stirred for 15 min at room temp.; solvent was evapd.; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Ar atm.; molar ratio Te:I2:AlI3 1:2:1, heating (150-200°C, several hours); | 100% |

-
-
260-94-6
acridine

-
-
194140-74-4
bis(2,2,6,6-tetramethylpiperidino)aluminium iodide

-
-
7784-23-8
aluminium(III) iodide

-
-
211381-97-4
(2,2,6,6-tetramethylpiperidino)2Al(acridine) tetraiodoaluminate

| Conditions | Yield |
|---|---|
| In dichloromethane inert atmosphere, addn. of soln. of acridine to soln. of piperidino compound at -78°C, -78°C (1 h), addn. of AlI3, slow warming toroom temp.; filtration, removement of volatiles (vacuum); elem. anal.; | 99% |

-
-
211381-78-1
(2,2,6,6-tetramethylpiperidino)2AlI(isoquinoline)

-
-
7784-23-8
aluminium(III) iodide

-
-
211381-95-2
(2,2,6,6-tetramethylpiperidino)2Al(isoquinoline) tetraiodoaluminate

| Conditions | Yield |
|---|---|
| In toluene inert atmosphere, addn. of soln. of AlI3 to soln. of piperidino compoundat 5-10°C, stirring (1.5 h); filtration, concn. of filtrate to dryness (vacuum); elem. anal.; | 99% |
-
-
55664-33-0
bis(tricyclohexylphosphine)platinum(0)

-
-
7784-23-8
aluminium(III) iodide

-
-
960294-96-6
Pt(P(C6H11)3)2AlI3

| Conditions | Yield |
|---|---|
| In benzene AlBr3 was added to a soln. of Pt-complex in benzene, the soln. was kept at room temp. for 24 h; | 99% |

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) (Ar); solid state reaction; equimolar mixt. of TlI, and AlI3 sealed under vac. in glass ampoules; heated with 10°C/h to 150°C for 48 h; cooled to 25°C with 2.degre.C/h; detn. by XRD; | 99% |
-
-
10034-85-2
hydrogen iodide

-
-
7784-23-8
aluminium(III) iodide

-
-
158875-67-3
1,1,3,3-tetraphenyltrisilan

-
-
192330-84-0
tetraiodotrisilane

| Conditions | Yield |
|---|---|
| In benzene byproducts: C6H6; N2-atmosphere; passing hydrogen halide through soln. of silane contg. little Al-halide (gentle heating or cooling when necessary) to end of hydrogen halide consumption; evapn. (vac.), heptane addn., decantation from Al-halide, evapn.; elem. anal.; | 98% |

-
-
176763-62-5
(1R,2R)-(-)-N,N'-bis(3,5-di-tert-butylsalicydene)-1,2-cyclohexanediaminocobalt(II)

-
-
7784-23-8
aluminium(III) iodide

| Conditions | Yield |
|---|---|
| In dichloromethane; water addn. of Co(C6H10(NCHC6H2O(t-Bu)2)2) (1 equiv.) to soln. of AlI3 (1 equiv.) in CH2Cl2 and H2O; stirring at room temp. for 1 h; concn. under reduced pressure, treatment with H2O and CH2Cl2; | 98% |
-
-
18816-18-7
1,1,2,2,3,3-hexaphenyltrisilane

-
-
10034-85-2
hydrogen iodide

-
-
7784-23-8
aluminium(III) iodide

-
-
192330-89-5
I2HSiSiI2SiHI2

| Conditions | Yield |
|---|---|
| In benzene byproducts: C6H6; N2-atmosphere; passing hydrogen halide through soln. of silane contg. little Al-halide (gentle heating or cooling when necessary) to end of hydrogen halide consumption; evapn. (vac.), heptane addn., decantation from Al-halide, evapn.; elem. anal.; | 97% |

-
-
3978-81-2
4-tert-butylpyridine

-
-
194140-74-4
bis(2,2,6,6-tetramethylpiperidino)aluminium iodide

-
-
7784-23-8
aluminium(III) iodide

-
-
211381-99-6
AlI3(4-tert-butylpyridine)

| Conditions | Yield |
|---|---|
| In hexane inert atmosphere, addn. of tert-butylpyridine to soln. of piperidino compound, addn. of AlI3 at room temp.; crystn. (overnight), filtration, washing (pentane); elem. anal.; | 96% |

-
-
78129-68-7
2,2-dimethylpropylidynephosphine

-
-
7784-23-8
aluminium(III) iodide

| Conditions | Yield |
|---|---|
| In dichloromethane (argon); -78°C to room temp., stirring (2 h); removal of solvent (25°C/E-3 mbar); | 95% |
-
-
281654-50-0
CH3NC(CH3)CHC(CH3)N(CH3)AlH2

-
-
7784-23-8
aluminium(III) iodide

-
-
420087-18-9
C6H5NC(CH3)CHC(CH3)NC6H5AlI2

| Conditions | Yield |
|---|---|
| In dichloromethane under inert gas, 2 equiv. of Me3NHI was added to CH2Cl2 soln. of Al-complex at -20 °C., stirring for 3 h, warming up to room temp., stirring at room temp. for 8 h; solvent was removed in vac., residue was three times extd. with toluene,solvent was removed, solid was recrystd. from toluene/pentane at -30 .d egree.C, elem. anal.; | 94% |
-
-
10034-85-2
hydrogen iodide

-
-
7784-23-8
aluminium(III) iodide

-
-
185988-25-4
1,1,2,2-Tetraphenyl-trisilane

-
-
192330-83-9
I2HSiSiI2SiH3

| Conditions | Yield |
|---|---|
| In benzene byproducts: C6H6; N2-atmosphere; passing hydrogen halide through soln. of silane contg. little Al-halide (gentle heating or cooling when necessary) to end of hydrogen halide consumption; evapn. (vac.), heptane addn., decantation from Al-halide, evapn.; elem. anal.; | 94% |

-
-
10034-85-2
hydrogen iodide

-
-
7784-23-8
aluminium(III) iodide

-
-
192330-96-4
1,1,2,2,3-Pentaphenyl-trisilane

-
-
192330-86-2
I2HSiSiI2SiH2I

| Conditions | Yield |
|---|---|
| In benzene byproducts: C6H6; N2-atmosphere; passing hydrogen halide through soln. of silane contg. little Al-halide (gentle heating or cooling when necessary) to end of hydrogen halide consumption; evapn. (vac.), heptane addn., decantation from Al-halide, evapn.; elem. anal.; | 94% |


| Conditions | Yield |
|---|---|
| In benzene under N2, CH3SH introduced in a suspension of AlI3 for 7 min, dissoln. of AlI3, stirred for 15 min at room temp.; evapn., pptn., filtered, washed (hexane), dried in vac., elem. anal.; | 93% |

| Conditions | Yield |
|---|---|
| In benzene lead dithiolate was added to a suspn. of AlI3 in benzene under N2, mixt. was heated under reflux for 2 h; filtered, filtrate was concd., ppt. collected by filtration, washed with pentane, dried under high vac.; elem. anal.; | 93% |
-
-
7784-23-8
aluminium(III) iodide

-
-
13455-01-1
phosphorous triiodide

-
-
85887-16-7
P2I5(1+)*AlI4(1-) = [P2I5][AlI4]

| Conditions | Yield |
|---|---|
| In carbon disulfide (N2); stirring (room temp., 24 h); solvent removal (dynamic vac.); | 92% |

| Conditions | Yield |
|---|---|
| In diethyl ether conventional Schlenk technique under dried Ar: addn. (dropwise) of 22mmol acetone in 10ml diethylether to a stirred, recently prepared, soln. of 10mmol AlI3 in 100ml diethylether, further stirring of mixture for 5min;; filtration, washing and drying in vacuo; elem. anal.;; | 91% |
| In carbon disulfide conventional Schlenk technique under dried Ar: addn. of acetone to soln. of aluminium (III) iodide in carbondisulfide;; elem. anal.; 1H-NMR;; | >90 |
-
-
28420-40-8, 32812-83-2, 40597-30-6
lead(II) ethyl sulfide

-
-
7784-23-8
aluminium(III) iodide

| Conditions | Yield |
|---|---|
| In benzene lead dithiolate was added to a suspn. of AlI3 in benzene under N2, mixt. was heated under reflux for 2 h; filtered, filtrate was concd., ppt. collected by filtration, washed with pentane, dried under high vac.; elem. anal.; | 89% |
-
-
23897-15-6
tris(2,4,6-trimethylphenyl)phosphine

-
-
7553-56-2
iodine

-
-
7784-23-8
aluminium(III) iodide

-
-
1334609-05-0
[Mes3PI][AlI4]

| Conditions | Yield |
|---|---|
| In toluene under N2; stirred for 30 min; solvent removed (vac.), solid stirred in hexanes for 10 min, filtered onglass frit, washed with hexanes, dried; | 89% |

-
-
13455-00-0
diphosphorus tetraiodide

-
-
7553-56-2
iodine

-
-
7784-23-8
aluminium(III) iodide

-
-
85887-16-7
P2I5(1+)*AlI4(1-) = [P2I5][AlI4]

| Conditions | Yield |
|---|---|
| In carbon disulfide (N2); stirring (room temp., 24 h); solvent removal (dynamic vac.); | 88% |

-
-
211381-81-6
(2,2,6,6-tetramethylpiperidino)2AlI(pyridine)

-
-
7784-23-8
aluminium(III) iodide

-
-
211382-02-4
(2,2,6,6-tetramethylpiperidino)AlI2(pyridine)

| Conditions | Yield |
|---|---|
| In toluene inert atmosphere, addn. of AlI3 to soln. of piperidino compound at 0°C, stirring (overnight); filtration, concn. to dryness (vacuum), crystn. (temperatures up to 35°C (several days, storage (1 week)), washing (pentane); elem. anal.; | 88% |
-
-
124-38-9
carbon dioxide

-
-
23897-15-6
tris(2,4,6-trimethylphenyl)phosphine

-
-
7784-23-8
aluminium(III) iodide

| Conditions | Yield |
|---|---|
| In further solvent(s) in bromobenzene; for 5 min; pptd. using hexanes, filtered, dried on a frit; | 87% |

Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
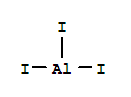
 C
C