Hebei Quanhe Biotechnology Co. LTD
1. Timely and efficient service to ensure communication with customers 2. Produce products of different specifications and sizes according to your requirements. 3. Quality procedures and standards recognized by SGS. Advanced plant equipment ensures
Hangzhou Dingyan Chem Co., Ltd
Items Standard Result Assay (Ursolic acid) 98%min 98.22% ----------------------------------------------------------------
Cas:7439-89-6
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Trading Company
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:7439-89-6
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryHebei yanxi chemical co.,LTD.
Hebei yanxi chemical co., LTD is a professional research, development and production lead diacetate trihydrate /Lead acetate trihydrateCAS:6080-56-4 2-phenylacetamide CAS:103-81-1 4-Aminophenyl-1-phenethylpiperidine CAS:21409-26-7 Citric acid monoh
Cas:7439-89-6
Min.Order:10 Metric Ton
FOB Price: $800.0 / 870.0
Type:Trading Company
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:7439-89-6
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Good quantity Iron Basic information Product Name: Iron Synonyms: 3ZhP;ANCOR EN 80/150;ancoren80/150;Armco iron;armcoiron;EFV 250/400;efv250/400;EO 5A CAS: 7439-8
Cas:7439-89-6
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufactu
Cas:7439-89-6
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:7439-89-6
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Shanghai Minstar Chemical Co., Ltd
Product Name: Iron Synonyms: 3ZhP;ANCOR EN 80/150;ancoren80/150;Armco iron;armcoiron;EFV 250/400;efv250/400;EO 5A CAS: 7439-89-6 MF: Fe MW: 55.85 EINECS: 231-096-4 Product Categories: IronEssential Chemicals;Chemical Synthesis;Me
Baoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present,
Cas:7439-89-6
Min.Order:1 Kilogram
FOB Price: $2.3
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
Iron CAS:7439-89-6 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates, steroi
Shandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Henan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Lonwin Chemical Group Limited
Iron CAS: 7439-89-6 Specification product name 4-tert-butylbenzoic acid (ptbba) cas number 98-73-7 assay ≥99% appearance white crystalline powder capacity 3000mt/year applicati
Changchun Artel lmport and Export trade company
Product Detail Minimum Order Qty. 10 Gram
Cas:7439-89-6
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Cas:7439-89-6
Min.Order:10 Kilogram
Negotiable
Type:Trading Company
inquiryAfine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:7439-89-6
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:grey powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Intermediates Tra
Hangzhou Huarong Pharm Co., Ltd.
Huarong Industrial Group Limited established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World.
Hebei Sankai Chemical Technology Co., Ltd
1. Product advantages High purity, all above 98.5%, no impurities after dissolution We will test each batch to ensure quality OEM and private brand services designed for free Various cap colors available We can also provide MT1 peptide powd
Jiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:7439-89-6
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Henan Kanbei Chemical Co.,LTD
High quality, competitive price, fast delivery and first-class service we possesses have won the trust and praise of customers. Standard: BP/USP/EP The purity is equal or greater than 99%. As a supplier, we can provide high-quality products. Cle
Cas:7439-89-6
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Lab/Research institutions
inquirySynthetic route

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| With hydrogen In neat (no solvent) passing H2 over Fe2O3 at 350°C over period of 36 h, at 440°C 12 h or at 500°C in fast reaction;; | 100% |
| With H2 In neat (no solvent) passing H2 over Fe2O3 at 350°C over period of 36 h, at 440°C 12 h or at 500°C in fast reaction;; | 100% |
| With hydrogen In neat (no solvent) reduction of Fe2O3 at 600°C leads to formation of powdered Fe, at 1000°C formed Fe hardly fragile;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Isothermal heat treatment for 2 h at 400°C.; | 100% |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| With 2,3,5,6-tetramethyl-1,4-bis(trimethylsilyl)-1,4-diaza-2,5-cyclo-hexadiene In tetrahydrofuran at 20℃; for 6h; Inert atmosphere; | 100% |
| With hydrogen In neat (no solvent) heated with H2 at approx. 350 °C; formation of pyrophoric Fe;; | |
| With aluminium trichloride; 1-ethyl-3-methyl-1H-imidazol-3-ium chloride In melt Electrochem. Process; (He or Ar); dissoln. of FeCl2 in AlCl3/1-ethyl-3-methyimidazoline chloride ionic liquid (room temp.), chronoamperometric electrodeposition (tungsten electrode substrate, -0.35 V vs. Fe/Fe(II), 120 s, room temp.); |

| Conditions | Yield |
|---|---|
| In gas collision induced dissocn. reaction (argon) in a mass spectrometer; energy range 17-78 eV; total pressure: 4E-6 Torr; not isolated; | A 100% B 100% |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| With water; lithium chloride In melt at 660℃; for 5h; Inert atmosphere; Electrolysis; | 98.4% |
-
-
13463-40-6
iron pentacarbonyl

-
-
876938-53-3
Cyclohepta-1,3-diene

-
A
-
40674-86-0
tricarbonyl(η-3-cyclohepta-1,3-diene)iron

-
B
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In dibutyl ether cycloheptadiene stirred in n-Bu2O while N2 bubbled through mixt. for 15h, Fe(CO)5 added, heated with stirring at 150°C for 44 h, cooled; filtered through Celite, evapd. in vac.; | A 93% B n/a |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran FeCl3 in THF was added dropwise to stirring soln. of 12 ml LiBEt3H in THF (1.0 M), held at const. temp. under N2 atm.; forms of particles dependon dropping rate (1 drop/10s, 1/s, 2/s), stirring rate (200, 400, 1600 rpm) and temp.(0 - 60°C); vac. filtration, washed THF/EtOH (1:1), dried in vac.; | 90% |

| Conditions | Yield |
|---|---|
| In decane byproducts: CH2CH2, CH3CHCH2, CO; Ar atmosphere; decompn. (165°C, 13 h); further products; GLC, chromato-mass spectroscopy; | A n/a B n/a C n/a D 5% E 75% |

-
-
14243-23-3
(μ-dithio)bis(tricarbonyliron)

-
D
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In n-heptane N2 atmosphere; stirring (70°C, 40 min); filtn., evapn., chromy. (silica gel); | A n/a B 66% C 25% D n/a |


| Conditions | Yield |
|---|---|
| flash vac. pyrolysis in a Pyrex tube connected with a cold trap (E-5 Torr, 285-315°C); coating of the hot zone by Fe, rinsing of trap with acetone, GC; | A <1 B n/a C 63% |
| With ceric ammonium nitrate In acetone addn. of (NH4)2Ce(NO3)6 to a stirred soln. of Fe-compound in acetone (5 min, air), stirring for 2 h; monitored by TLC (hexane), filtn. (Celite), dilution of filtrate with ether, filtn. and evapn. to dryness, GLC of white crystals in acetone, TLC (CH2Cl2/hexane=1:1), (1)H NMR, IR, elem. anal.; | A 35.5% B 36% C 4.4% |

-
-
13463-40-6
iron pentacarbonyl

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In not given byproducts: CO; Sonication; soln. of Fe(CO)5 in diphenylmethane sonicated for 3 h under Ar at 30 °C to give iron nanoparticles; removed by centrifugation, washed with pentane, dried under vac., detd. by Moessbauer spectroscopy, XRD; | 53% |
| In further solvent(s) under Ar; thermolysis of Fe(CO)5 using heterogeneous nucleation technique (Proc. Phys. Soc. A 1949, 62, 562); diluted 2 times with octyl ether; Fe(CO)5 added (100°C); heated (260°C); cooled; EtOH (3:1 volume ratio to octyl ether); collected with magnet; | 40% |
| With cis-Octadecenoic acid In further solvent(s) under Ar; thermolysis of Fe(CO)5 using heterogeneous nucleation technique (Proc. Phys. Soc. A 1949, 62, 562); oleic acid:oleylamine 1:1 molar ratio; diluted 2 times with octyl ether; Fe(CO)5 added (100°C); heated (260°C); cooled; EtOH (3:1 volume ratio to octyl ether); collected with magnet; | 40% |

| Conditions | Yield |
|---|---|
| With n-BuLi In tetrahydrofuran; hexane under Ar; FeCl2 added at 0°C to stirred suspn. of (C5Me5)Li in THF; stirred (1 h); cooled to -78°C; suspn. prepared from n-BuLi inn-hexane and HSEt at 0°C added; 1 h at -78°C; warmed to r oom temp. with stirring overnight; evapd. to dryness; purified by column chromy. (neutral alumina, n-hexane); insol. solid washed with H2O (Fe); elem. anal.; | A 9% B 49% C 15% |

-
-
107339-80-0
(η4-1,3-butadiene)tris(triethylphosphine)iron(0)

-
A
-
103835-78-5
bis(η4-1,3-butadiene)(triethylphosphino)iron(II)

-
B
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In tetrahydrofuran under Ar at 5°C; | A 40% B 40% |
-
-
99611-53-7
iron(II) bromide dimethoxyethane adduct

-
-
1421320-35-5
potassium dimethylnopadienide

-
B
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; toluene at 20℃; for 4h; Inert atmosphere; | A 28% B n/a |


| Conditions | Yield |
|---|---|
| With H2 In decalin High Pressure; 71.5 atm H2 at room temp., heated to 200°C and stirred for 24 h; pressure realesed, detn. by GC and GC-MS; | A 27% B n/a |

| Conditions | Yield |
|---|---|
| In n-heptane; decalin Sonication; 0°C (Ar); products identified IR, UV, mass spect., chromy.; | A 9.8% B n/a |
| In octane Sonication; 0°C (Ar); products identified IR, UV, mass spect., chromy.; | A 6.9% B n/a |
| In decalin Sonication; 0°C (Ar); 0.1 M Fe(CO)5; products identified IR, UV, mass spect., chromy.; | A 4.7% B n/a |

| Conditions | Yield |
|---|---|
| In neat (no solvent, gas phase) mixt. of vapor of Fe(CO)5 and H2 decomposed by plasma-chemical decomposition on Al2O3; monitored by XRD; | A n/a B n/a C n/a D 1% E n/a |


| Conditions | Yield |
|---|---|
| Stage #1: ferric nitrate at 450℃; for 8h; Stage #2: With hydrogen at 500℃; for 16h; | |
| In neat (no solvent) 15 min at 900°C under atomic hydrogen atmosphere; | |
| With hydrogen In methanol Fe/MCM-41 prepd. by impregnation of MCM-41 with methanolic soln. of Fe(NO3)3 with stirring for 24 h under N2; catalyst filtered, washed with methanol, dried at 373 K and calcined at 773 K; catalyst reduced in H2 above 773 K; |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; methanol for 1h; Heating / reflux; |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| Heating / reflux; |

| Conditions | Yield |
|---|---|
| In melt at red heat; pression of KCN from iron sponge out; | |
| In melt byproducts: N2; at red heat; extraction of KCN with H2O; | |
| In melt at red heat; pression of KCN from iron sponge out; |


| Conditions | Yield |
|---|---|
| In water Electrolysis; electrolysis of aq. soln. of FeSO4*7H2O and NH3 leads to precipitation of light grey iron on cathode;; |

| Conditions | Yield |
|---|---|
| With H2 In neat (no solvent) sample heating in TG apparatus in 25% H2/He (50 ml/min) at 5 K/min up to800°C; TG; |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| With hydrogen In neat (no solvent) Kinetics; byproducts: H2O; 300°C, carrier gas N2 or Ar + CH4 or CH4 + Ar + CO or air; | |
| With hydrogen In gaseous matrix Kinetics; byproducts: H2O; sample redn. by 10% H2/Ar at 15 K/min up to 1000 K; | |
| With hydrogen; gold In gaseous matrix Kinetics; byproducts: H2O; Au/Fe2O3 mixt. heating at 15 K/min in 10% H2/Ar (24 ml/min) up to 1260 K; | |
| With hydrogen In gaseous matrix Kinetics; byproducts: H2O; sample heating at 15 K/min in 10% H2/Ar (24 ml/min) up to 1260 K; |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In sodium hydroxide aq. NaOH; Electrolysis; in 50 wt % NaOH/water electrolyte at 110°C using Pt cylinder counter electrode and steel/adsorbed hematite as cathode; at -1.2 V vs. Hg/HgO; | |
| With hydrogen hematite redn. at 500°C under hydrogen; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; reduced at 252.5-383°C under conditions without an external diffusion effect; TEM; |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In solid Electrolysis; electrolyzing molten FeSiO3 with addn. of either CaO or MgO leads to pptn. of Fe on cathode;; | |
| In neat (no solvent) no redn. of feO to Fe in a stream of H at 850°C;; | 0% |
| In neat (no solvent) no redn. of feO to Fe in a stream of H at 850°C;; | 0% |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In not given Electrolysis; electrolyzing soln. of NaFeP2O7 with bath potential 4 V;; contains Fe2O3 and pyrophosphorous acid;; |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In water Electrolysis; electrolyzing soln. FeCl2*4H2O and NaCl (addn. of little HCl to clear soln.) at 50 to 70°C with current efficiency of 95 %;; | |
| With ammonia In ethanol n-type Si(100) substrate coated with drop of soln. FeCl2*4H2O; dried; loaded on quartz boat into quartz tube reactor; heated under Ar; treated by NH3 with flow rate of 20 sccm for 1-10 min; | |
| With sodium hydroxide In further solvent(s) heating FeCl2*4H2O and NaOH in propylene glycol; X-ray diffraction; |
-
-
907994-35-8
4,4-dimethyl-3-(2-nitrobenzyl)-2-oxazolidinone

-
-
7439-89-6
iron

-
-
907993-76-4
4,4-dimethyl-3-(2-aminobenzyl)-2-oxazolidinone

| Conditions | Yield |
|---|---|
| With ammonium chloride In ethanol; water | 100% |
-
-
907994-36-9
5-methyl-3-(2-nitrobenzyl)-2-oxazolidinethione

-
-
7439-89-6
iron

-
-
907993-79-7
5-methyl-3-(2-aminobenzyl)-2-oxazolidinethione

| Conditions | Yield |
|---|---|
| With ammonium chloride In ethanol; water | 100% |
-
-
907994-37-0
5-ethyl-3-(2-nitrobenzyl)-2-oxazolidinone

-
-
7439-89-6
iron

-
-
907993-80-0
5-ethyl-3-(2-aminobenzyl)-2-oxazolidinone

| Conditions | Yield |
|---|---|
| With ammonium chloride In ethanol; water | 100% |
-
-
907994-43-8
5,5-dimethyl-3-(2-nitrobenzyl)thiazolidine-2-one

-
-
7439-89-6
iron

-
-
907994-20-1
3-(2-aminobenzyl)-5,5-dimethylthiazolidine-2-one

| Conditions | Yield |
|---|---|
| With ammonium chloride In ethanol; water | 100% |
-
-
280759-64-0
3-hexyl-6-methyl-6-(3-nitrophenyl)-3-azabicyclo[3.1.0]hexan-2-one

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| With calcium chloride In ethanol; water | 100% |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| With calcium chloride In ethanol; dichloromethane; water | 100% |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| With calcium chloride In ethanol; dichloromethane; water | 100% |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| With calcium chloride In ethanol; dichloromethane; water | 100% |

| Conditions | Yield |
|---|---|
| In water soln. of Fe in concd. HCl was refluxed; ppt. filtered off, washed with Et2O, dried in vac.; | 100% |
| In hydrogenchloride evapn. a soln. of iron filings in dild. aq. HCl over iron filings until the hot soln. starts foaming; crystn. on cooling;; filtn.; crystn.; drying in a stream of dry air at 30-40°C;; | |
| In hydrogenchloride evapn. a soln. of iron filings in dild. aq. HCl over iron filings until the hot soln. starts foaming; crystn. on cooling;; filtn.; crystn.; drying in a stream of dry air at 30-40°C;; | |
| In water slight excess of 0.1 M hydrochloric acid added to iron powder, heated to dissolution; evapd.; | |
| In hydrogenchloride iron powder and aq. HCl; |


| Conditions | Yield |
|---|---|
| at 250℃; for 16h; Inert atmosphere; | 100% |
| In not given HCOOH was neutralized with Fe at 70-80°C; filtered, concd., cooled to room temp., recrystd. from water, dried in air; | |
| Inert atmosphere; | |
| Inert atmosphere; | |
| In neat (no solvent) at 80℃; |

-
-
917-23-7
5,10,15,20-tetraphenyl-21H,23H-porphine

-
-
7439-89-6
iron

-
-
16591-56-3
5,10,15,20-tetraphenyl porphyrin iron

| Conditions | Yield |
|---|---|
| In toluene byproducts: H2; cocondensation of iron and toluene vapor at liq. nitrogen temp.; heating to -94.6°C; dropwise addn. of porphine soln. under nitrogen to the slurry (molar ratio Fe:porphine 4:1); gradually warming to 0°C, 1h; filtration; evapn. of filtrate; | 100% |
| With Ag(111) In neat (no solvent) deposition of porphyrin deriv. onto silver by evapn. at 638 K, removing excess of porphyrin at 550 K, deposition of stoich. amount of iron; Buchner F., Schwald V., Cmanici K., Steinrueck H.-P., Marbach H. ChemPhysChem 2007, 8, 241-243; XPS and UPS; |

| Conditions | Yield |
|---|---|
| With HClO4 In water iron fillings dissolve in dilute HClO4, added aq. NaOH, resulting soln. added to aq. picrolonic acid at room temp.; ppt. filtered, washed with water, ethanol, ether, dried in vac. till constant weight, elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Li3N fused in pure iron vessel; sealed under 300 kPa of N2; heated at 850-1050°C for 12 h; thermally quenched; detd. by X-ray powder diffraction; | 100% |


| Conditions | Yield |
|---|---|
| In melt (Ar glovebox) U, Fe and Se were placed in fused-silica ampoule, evacuated to about 1E-4 Torr, sealed, heated to 1173 K in 30 h, maintained at 1173 K for 2 days, cooled to 773 K in 6 days, maintained at 773 K for 2 days, cooled to 298 K over 6 h; washed with water and dried with acetone, XRD; | 100% |

-
-
1342835-29-3
1-(4-(7-(2-fluoro-4-nitrophenoxy)thieno[3,2-b]pyridin-2-yl)benzyl)pyrrolidin-2-one

-
-
7439-89-6
iron

-
-
1342835-31-7
1-(4-(7-(4-amino-2-fluorophenoxy)thieno[3,2-b]pyridin-2-yl)benzyl)pyrrolidin-2-one

| Conditions | Yield |
|---|---|
| With ammonium chloride In water | 100% |

| Conditions | Yield |
|---|---|
| In water at 60℃; for 47h; | 100% |
-
-
557-34-6
zinc diacetate

-
-
373-02-4, 14998-37-9, 33025-09-1, 94044-50-5, 98268-31-6
nickel diacetate

-
-
144-62-7
oxalic acid

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| In acetic acid heating of iron powder in a twofold excess of 1.5-2.0 M acetic acid under a N2 atmosphere; stirring; addn. of a soln. of Zn(2+) and Ni(2+) acetate; boilig; addn. of 3-5% excess 1 M oxalic acid; boiling for 1 h; filtration;; washing and drying at 100°C;; | 99.8% |
| In acetic acid heating of iron powder in a twofold excess of 1.5-2.0 M acetic acid under a N2 atmosphere; stirring; addn. of a soln. of Zn(2+) and Ni(2+) acetate; boilig; addn. of 3-5% excess 1 M oxalic acid; boiling for 1 h;; frozen ppt. with liquid N2 (with total mother soln.) and then freeze dried;; |


| Conditions | Yield |
|---|---|
| With bromine; iodine In tetrachloromethane; (2S)-N-methyl-1-phenylpropan-2-amine hydrate | 99.5% |

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| With calcium chloride In methanol; ethanol; dichloromethane; water | 99% |
-
-
98027-81-7
2,6-dibromo-4-nitro-pyridine 1-oxide

-
-
7439-89-6
iron

-
-
39771-34-1
4-amino-2,6-dibromopyridine

| Conditions | Yield |
|---|---|
| With acetic acid | 99% |

-
-
131341-02-1
2,2,4-trimethyl-4-[5-nitro-3-(3-phenylprop-2-ynyloxy)phenyl]-1,3-dioxolane

-
-
7439-89-6
iron

| Conditions | Yield |
|---|---|
| With hydrogenchloride; triethylamine In methanol; water | 99% |


-
-
875774-88-2
4-nitro-1-(4-trifluoromethoxyphenoxy)-2-trifluoromethylbenzene

-
-
7439-89-6
iron

-
-
875774-55-3
4-(4-trifluoromethoxyphenoxy)-3-trifluoromethyl-aniline

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; water; ethyl acetate | 99% |


| Conditions | Yield |
|---|---|
| With oxygen In dimethyl sulfoxide metal. Fe under O2 atm. treated with DMSO and triflic acid (3 equiv.) in3 portions, heated at 100°C for 24 h; | 99% |


| Conditions | Yield |
|---|---|
| In further solvent(s) under Ar atm. using Schlenk techniques; metal powder, thiourea (excess),4-methylpyridine refluxed overnight; soln. refluxed for 4 ds; soln. coo led to room temp.; soln. layered (hexane); crystn.; | 99% |


| Conditions | Yield |
|---|---|
| In water stoich., Fe powder dissolved in aq. soln. of CF3SO3H by heating; ppt. filtered, dried (air), elem. anal.; | 99% |
| In water metal compd. dissolved in aq. soln. of triflic acid; filtered, cocd., crystd., dried at 200°C for several h; | |
| In water mixed, warmed in water; |

| Conditions | Yield |
|---|---|
| In acetonitrile Electrolysis; iron anode, 0.1 M tetraethylammonium bromide base electrolyte, inert atmosphere, 40°C, current density 5 mA/cm*2; pptn. on water diln., collection (filtn.), dissoln. (acetone), repptn. on hexane addn., recrystn. (acetone); | 99% |
| In dimethyl sulfoxide Electrolysis; iron anode, sodium bromide base electrolyte, inert atmosphere, 40°C, current density 5 mA/cm*2; pptn. on water diln., collection (filtn.), dissoln. (acetone), repptn. on hexane addn., recrystn. (acetone); | <80 |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
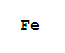
















 F,
F,  Xi
Xi