Henan Allgreen Chemical Co.,Ltd
T he company has advanced technology, as well as a large number of excellent R & D team, to provide customers from the grams to one hundred kilograms and tons of high-quality products, competitive prices and quality service Appearance:W
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Tianfu Chemical Co., Ltd.
Water Basic information Product Name: Water Synonyms: ALKALINE TRANSFER SOLUTION;Liquid soda;LYE;DOCTOR SOLUTION;DOCTORS SOLUTION;DOCTOR'S SOLUTION;Dihydrogen oxide;dihydrogenoxide
Cas:7732-18-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:7732-18-5
Min.Order:1 Gram
FOB Price: $6.0 / 9.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:7732-18-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
Water CAS:7732-18-5 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates, stero
Cas:7732-18-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Shandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Lonwin Chemical Group Limited
Water CAS: 7732-18-5 Specification Welcome to contact us to get complete COA. Shanghai Lonwin Chem is a leading manufacturer and supplier of chemicals in China.We develop,produce and distribute high quality pharmaceuticals, intermediates, s
Cas:7732-18-5
Min.Order:1 Kilogram
FOB Price: $100.0 / 150.0
Type:Other
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
HANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:colourless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Intermediat
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Jiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Henan Kanbei Chemical Co.,LTD
High quality, competitive price, fast delivery and first-class service we possesses have won the trust and praise of customers. Standard: BP/USP/EP The purity is equal or greater than 99%. As a supplier, we can provide high-quality products. Cle
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:7732-18-5
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryTAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:7732-18-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:7732-18-5
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Trading Company
inquiryHebei yanxi chemical co.,LTD.
搜索 复制 CHENGDU YANXI is a comprehensive manufacturer and an international distribution of products throughout the world. Specialized in Scrap metal, Chemical raw materials, Paper products and color industry. We aim to become leading posit
Cas:7732-18-5
Min.Order:1 Gram
FOB Price: $2.0 / 3.0
Type:Trading Company
inquiryGolden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Cas:7732-18-5
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Trading Company
inquiryHebei Mojin Biotechnology Co.,Ltd
We produce carbomer by our own factory,carbomer 940 is our best seller.It is used for cosmetic and medical.And we also become agent for lubrizol.Our carbomer 940 get high viscosity and good transparency.The price will depond on the market,surely supp
Hunan chemfish Pharmaceutical co.,Ltd
Hunan chemfish Pharmaceutical co.,Ltd.located in Lugu High-tech industral park ,Hunan province . with its own R&D center and more than 10000㎡manufacture plant . Chemfish owns 40 reactors from 1000L to 8000L. With complete auxiliary equipment as:
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Shanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:7732-18-5
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryWuxi TAA Chemical Industry Co.,LTD.
1.A strong technical force and advanced processing equipments. The quality of the products has been strictly inspected and all kinds of index have reached or exceeded domestic and international standards.2. Now we have established long-term stable re
Synthetic route

-
B
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| tetrabutyl ammonium fluoride In xylene at 152℃; Conversion of starting material; | A 100% B 100% |
| N-formyl Rosinamine In xylene at 154℃; Conversion of starting material; | A 92% B 92% |


| Conditions | Yield |
|---|---|
| platinum In neat (no solvent) reaction at room temperature;; | 100% |
| platinum In neat (no solvent) reaction at room temperature;; | 100% |
| alpha-alumina impergnated with patinum nitrate and tin (II) chloride calcinated at 500C (0.08 wtpercent Pt; 0.08 wtpercent Sn) at 300℃; under 9000.9 Torr; Conversion of starting material; Gas phase; |

| Conditions | Yield |
|---|---|
| In water pure SO2 is introduced into alkaline Na2S soln.; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| In water at 60°C, complete conversion;; pure Na2S2O3; | A n/a B 100% |
| In water mechanism discussed:; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) over H2SO4 in 7d;; | 100% |
| In neat (no solvent) loss of 3mol H2O in vac. over BaO;; | |
| In neat (no solvent) complete loss of H2O in vac. over H2SO4 within 8d at 0°C, in 2d at 22°C;; | >99 |
-
-
463-58-1
carbon oxide sulfide

-
-
7722-84-1
dihydrogen peroxide

-
A
-
124-38-9
carbon dioxide

-
B
-
7664-93-9
sulfuric acid

-
C
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| With potassium sulfate; potassium hydrogensulfate; potassium peroxomonosulfate In water Kinetics; oxidation of OCS studied in round-bottom Pyrex bulbs, acid-water mixtures introduced into bulbs and degassed, bulb reactors filled with with a gas mixture slightly above 1 atm total pressure with a typical mixing ratio of OCS:Ar:He=40:60:700 Torr; gas chromy. and mass spectroscopy applied for determination of product content; | A 100% B n/a C n/a |
| With sulfuric acid In water Kinetics; oxidation of OCS studied in round-bottom Pyrex bulbs, acid-water mixtures introduced into bulbs and degassed, bulb reactors filled with with a gas mixture slightly above 1 atm total pressure with a typical mixing ratio of OCS:Ar:He=40:60:700 Torr; gas chromy. and mass spectroscopy applied for determination of product content; | A 100% B n/a C n/a |


-
B
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) decomposition at 100 °C; | A 100% B n/a |

| Conditions | Yield |
|---|---|
| In water Kinetics; Reduction of (NH4)2S2O3 (c=0.4 mole/liter) by H2S in aq. soln. (50°C, pH=5, p(H2S)=0.08 MPa) in presence of Si-based catalyst.; Gravimetrical determination of S.; | A n/a B 99.7% |
| In water Kinetics; Reduction of (NH4)2S2O3 (c=1.0 mole/liter) by H2S in aq. soln. (50°C, pH=5, p(H2S)=0.08 MPa) in presence of Si-based catalyst.; Gravimetrical determination of S.; | A n/a B 99.87% |
| In water Kinetics; Reduction of (NH4)2S2O3 (c=1.0 mole/liter) by H2S in aq. soln. (50°C, pH=5, p(H2S)=0.08 MPa).; Gravimetrical determination of S.; | A n/a B 76.1% |
| In water Kinetics; Reduction of (NH4)2S2O3 (c=0.4 mole/liter) by H2S in aq. soln. (50°C, pH=5, p(H2S)=0.08 MPa).; Gravimetrical determination of S.; | A n/a B 71.5% |


| Conditions | Yield |
|---|---|
| In hydrogenchloride 20°C;satd. solns.; molar ratio 2 : 1; 15 % HCl soln., ,30 min;; S coagulated by addn. of gelatine or Al2(SO4)3; | A n/a B 99.7% |
| In hydrogenchloride 20°C;satd. solns.; molar ratio 2 : 1; 15 % HCl soln., ,30 min;; S coagulated by addn. of gelatine or Al2(SO4)3; | A n/a B 99.7% |
| In hydrogenchloride 20°C; satd. solns.; molar ratio 2 : 1; 3.5 % HCl soln.;; S coagulated by addn. of gelatine or Al2(SO4)3;; | A n/a B 93.5% |

-
-
67-56-1
methanol

-
A
-
34557-54-5
methane

-
B
-
124-38-9
carbon dioxide

-
C
-
7732-18-5
water

-
D
-
1333-74-0
hydrogen

| Conditions | Yield |
|---|---|
| Stage #1: methanol With dihydrogen peroxide In water Liquid phase; Stage #2: Cu/Zn/Al2O3 In water Conversion of starting material; Gas phase; | A n/a B n/a C n/a D 99% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) heated at 373 K for 1 h; XRD; | A 99% B n/a C n/a |

-
-
325744-49-8
1,1'-dicarboxylic cobalticinium chloride monohydrate

-
A
-
7647-01-0
hydrogenchloride

-
B
-
232598-14-0
Co(III)(η5-C5H4COOH)(η5-C5H4COO)

-
C
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| In neat (no solvent) heated at 440 K for 1 h at low pressure; XRD; | A n/a B 99% C n/a |


| Conditions | Yield |
|---|---|
| In neat (no solvent) heated at 373 K for 1 h; XRD; | A 99% B n/a C n/a |


| Conditions | Yield |
|---|---|
| Stage #1: LACTIC ACID at 180℃; for 5.5h; Heating / reflux; Stage #2: under 50 Torr; for 3.5h; Heating / reflux; | A n/a B 99% |

-
-
201230-82-2
carbon monoxide

-
-
1333-74-0
hydrogen

-
A
-
7783-06-4
hydrogen sulfide

-
B
-
7732-18-5
water

-
C
-
497-19-8
sodium carbonate

| Conditions | Yield |
|---|---|
| with molten Na2SO4*10H2O; heating at 927 to 983°C for 2 h; ratio of CO and H2 1:3; | A 98% B n/a C n/a |


-
B
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating up to 440°C; | A 98% B n/a |

-
B
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating up to 360°C; | A 98% B n/a |
-
-
75-91-2
tert.-butylhydroperoxide

-
-
947-42-2
diphenylsilanediol

-
-
603-36-1
triphenylantimony

-
B
-
7732-18-5
water

-
C
-
75-65-0
tert-butyl alcohol

| Conditions | Yield |
|---|---|
| In 1,4-dioxane dissoln., cooling (5°C), soln. of peroxo compd. addn. under stirring, stirring (2 h), volatile product sepn. in cooled trap (reduced pressure); elem. anal.; | A 98% B 89% C 96% |

-
-
80937-33-3
oxygen

-
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| With [Co2(OH)2(dipyridylethane naphthyridine)(μ-1,3-OC(NH)CH3)](PF6)3 In N,N-dimethyl-formamide; trifluoroacetic acid pH=7; Catalytic behavior; Reagent/catalyst; Solvent; pH-value; Electrochemical reaction; | 97% |
| LaCo5H3.4 In neat (no solvent) exposing LaCo5H3.4 to oxygen at 23.4°C; monitoring total pressure change; | |
| 20Cl(1-)*20Cl2C10H6N2*10Os(2+)*(C3H3N2CHCH2)11(CH(CONH2)CH2)77=(((Cl2C10H6N2)2ClOsN2C5H6)10CH(N2C3H3)CH2(C3H5NO)77)Cl10 In water Electrochem. Process; electroreduction of O2 under physiological conditions (pH 7.4, aq. NaCl,37.5°C); electrocatalyst: Os complex on carbon cloth; |
-
-
14989-30-1
hypochloric acid

-
-
13898-47-0
hypochloric acid

-
A
-
7647-01-0
hydrogenchloride

-
B
-
10049-04-4, 25052-55-5
chlorine dioxide

-
C
-
7732-18-5
water

-
D
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| In water reaction of HClO2 and HClO in weakly acidic or neutral soln. at ambient temp.;; removing of ClO2 with air;; | A n/a B 97% C n/a D n/a |
| In water reaction of HClO2 and HClO in aq. soln. at ambient temp.; influence of pH;; | |
| In water reaction of HClO2 and HClO in weakly acidic or neutral soln. at ambient temp.; acceleration on low concn. of ClO2(1-); no influence of ClO3(1-);; | |
| In water reaction of HClO2 and HClO in weakly acidic or neutral soln. at ambient temp.;; | |
| In water Kinetics; reaction of HClO2 and HClO in aq. soln. at ambient temp.;; |


-
B
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating up to 400°C; | A 97% B n/a |
-
-
1295-35-8
bis(1,5-cyclooctadiene)nickel (0)

-
-
107-18-6
allyl alcohol

-
A
-
187737-37-7
propene

-
B
-
79361-68-5
Ni(CH2CHCHO)(P(C6H5)3)2

-
C
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| With triphenylphosphine In tetrahydrofuran under N2 or argon or under vac., soln. of org. compd. (3.8 mol) added to mixt. of Ni(cod)2 and PPh3 (1.0/2.2 mol ratio), stirred at 30°Cfor 2 d; cooled to -78°C, solid filtered, washed with hexane, recrystd. from toluene-hexane, dried under vac.; detn. by IR and NMR; | A 97% B 93% C 92% |


| Conditions | Yield |
|---|---|
| aluminum oxide at 315.546℃; under 11103.3 Torr; | A 1.4% B 1.1% C 96.4% |


| Conditions | Yield |
|---|---|
| In not given equimolar amts. of HSO3(1-) and SO3(2-); | A n/a B 96% C 0% |
| In not given equimolar amts. of HSO3(1-) and SO3(2-); | A n/a B 96% C 0% |

-
-
64-17-5
ethanol

-
-
1795-62-6
ω-chlorocaprylic acid

-
A
-
105484-55-7
ethyl 8-chlorooctanoate

-
B
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| for 3h; Neat (no solvent); | A 95% B n/a |

-
-
75-91-2
tert.-butylhydroperoxide

-
-
2031-65-4
diethyldihydroxysilane

-
-
603-36-1
triphenylantimony

-
A
-
244109-21-5
Sb2O4Si2(C2H5)4(C6H5)6

-
B
-
7732-18-5
water

-
C
-
75-65-0
tert-butyl alcohol

| Conditions | Yield |
|---|---|
| In 1,4-dioxane dissoln., cooling (5°C), soln. of peroxo compd. addn. under stirring, stirring (2 h), volatile product sepn. in cooled trap (reduced pressure); elem. anal.; | A n/a B 89% C 95% |


-
-
7697-37-2
nitric acid

-
B
-
7732-18-5
water

-
C
-
10102-44-0
Nitrogen dioxide

-
D
-
2696-92-6
nitrosylchloride

-
E
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In nitric acid byproducts: Cl2; to Ru complex was added concd. HNO3; mixt. heated under reflux for 5 min; cooled to room temp.; ppt. filtered off; washed (water, alc., ether); dried (vac.); recrystd. (aq. HNO3); elem. anal.; | A 95% B n/a C n/a D n/a E n/a |


| Conditions | Yield |
|---|---|
| In dichloromethane stirring (room temp., 30 min), concg. (vac.), pptn. on hexane addn.; filtering, air-drying, recrystn. (CH2Cl2 / hexane); elem. anal.; | A 93% B n/a |

-
-
546104-93-2
3-(2-bromo-4,5-dimethoxyphenyl)pentan-2-one

-
-
107-21-1
ethylene glycol

-
A
-
546104-97-6
2-[1-(2-bromo-4,5-dimethoxyphenyl)propyl]-2-methyl-1,3-dioxolane

-
B
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene Heating / reflux; | A 92% B n/a |


| Conditions | Yield |
|---|---|
| With aluminium; sodium hydroxide at 21℃; under 758 Torr; Product distribution / selectivity; Sealed tube; | 100% |
| With Ce0896Y0.05Nb0054O2 at 1499.84℃; under 0.00750075 Torr; Reagent/catalyst; | 100% |
| With bis(pentamethylcyclopentadienyl)iron(II); Mn(bpy)2Br2 In acetonitrile for 22h; Catalytic behavior; Reagent/catalyst; Inert atmosphere; Sealed tube; | 100% |

| Conditions | Yield |
|---|---|
| In methanol; N,N-dimethyl-formamide | 100% |


-
-
7732-18-5
water

-
-
173206-12-7
4-chloro-5-methoxy-2-(tetrahydro-2H-pyran-2-yl)-3(2H)-pyridazinone

-
-
173206-14-9
5-methoxy-2-(tetrahydro-2H-pyran-2-yl)-3(2H)-pyridazinone

| Conditions | Yield |
|---|---|
| With triethylamine; Pd-C In ethanol; ethyl acetate | 100% |
| With triethylamine; Pd-C In ethanol; ethyl acetate | 100% |

-
-
149523-69-3
(3S,4S)-N-[(tert-butyloxy)carbonyl]-4-amino-3-(triisopropylsilyloxy)-5-phenylpentene

-
-
7732-18-5
water

-
-
149523-70-6
(2R,3S)-N-[(tert-Butyloxy)carbonyl]-3-amino-4-phenyl-2-(triisopropylsilyloxy) butan-1-ol

| Conditions | Yield |
|---|---|
| With sodium borohydrid In methanol; dichloromethane | 100% |
| With sodium borohydrid In methanol; dichloromethane | 100% |

-
-
15733-82-1
2-carboxy-4-chloroquinoline

-
-
75-09-2
dichloromethane

-
-
7732-18-5
water

-
-
114935-92-1
Methyl-4-chloroquinaldate

| Conditions | Yield |
|---|---|
| With NaH In N,N-dimethyl-formamide | 100% |


| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In acetonitrile; Petroleum ether | 100% |


-
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In diethyl ether | 100% |

-
-
7732-18-5
water

-
-
584-08-7
potassium carbonate

-
A
-
7567-87-5
6-formyl-2-naphthalenecarboxylic acid methyl ester

-
B
-
55343-77-6
methyl 6-hydroxymethyl-2-naphthalenecarboxylate

| Conditions | Yield |
|---|---|
| In methanol; ethyl acetate | A 100% B n/a |


| Conditions | Yield |
|---|---|
| at 80℃; | 100% |
| In nitric acid aq. HNO3; dissolving metal oxide in concd. HNO3, heating; evapn. on water bath, dissolving in water; | |
| In nitric acid aq. HNO3; by treating the metal oxide with dil. HNO3; the soln. was evapd. on a steam bath; the residue was dissolved in water, conced. to a viscous mass, cooled and kept in a desiccator after breaking up any lumps; |


| Conditions | Yield |
|---|---|
| at 80℃; | 100% |
| In nitric acid aq. HNO3; Eu2O3 treated with concd. HNO3; excess HNO3 evapd.; | |
| In nitric acid aq. HNO3; dissolving metal oxide in concd. HNO3, heating; evapn. on water bath, dissolving in water; |

-
-
7446-09-5
sulfur dioxide

-
-
7732-18-5
water

-
-
7553-56-2
iodine

-
A
-
7664-93-9
sulfuric acid

-
B
-
10034-85-2
hydrogen iodide

| Conditions | Yield |
|---|---|
| 0 - 25 °C; part of a Mg-S-I water splitting cycle; | A 100% B 100% |

-
-
7732-18-5
water

-
-
80937-33-3
oxygen

| Conditions | Yield |
|---|---|
| With sodium periodate; [(1,2,3,4,5-pentamethylcyclopentadienyl)Ir{P(O)(OH)2}3]Na at 25℃; Catalytic behavior; Reagent/catalyst; Electrochemical reaction; | 100% |
| With sodium sulfate pH=5.8; Reagent/catalyst; Electrochemical reaction; Irradiation; | 100% |
| With ammonium cerium (IV) nitrate; [(1,2,3,4,5-pentamethylcyclopentadienyl)IrCl{(3-methylimidazol-2-yliden-1-yl)2CHCOO}]; nitric acid at 26.84℃; pH=1; Kinetics; Reagent/catalyst; pH-value; Schlenk technique; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| at 170-180°C; in very dilute soln. complete decompn. in 2 h, incomplete decompn. in concd. solns.; | A n/a B 100% |
| byproducts: H2S4O6; | |
| sodium thiosulfate In water 100°C; |


| Conditions | Yield |
|---|---|
| at 80℃; | 100% |
| In water react. metal oxide with 6N HNO3; evapn. at 100°C; | |
| In nitric acid aq. HNO3; by treating the metal oxide with dil. HNO3; the soln. was evapd. on a steam bath; the residue was dissolved in water, conced. to a viscous mass, cooled and kept in a desiccator after breaking up any lumps; | |
| In nitric acid aq. HNO3; dissolving of Y2O3 in excess amt. of aq. nitric acid; |


| Conditions | Yield |
|---|---|
| In water soln. of Fe in concd. HCl was refluxed; ppt. filtered off, washed with Et2O, dried in vac.; | 100% |
| In hydrogenchloride evapn. a soln. of iron filings in dild. aq. HCl over iron filings until the hot soln. starts foaming; crystn. on cooling;; filtn.; crystn.; drying in a stream of dry air at 30-40°C;; | |
| In hydrogenchloride evapn. a soln. of iron filings in dild. aq. HCl over iron filings until the hot soln. starts foaming; crystn. on cooling;; filtn.; crystn.; drying in a stream of dry air at 30-40°C;; | |
| In water slight excess of 0.1 M hydrochloric acid added to iron powder, heated to dissolution; evapd.; | |
| In hydrogenchloride iron powder and aq. HCl; |


| Conditions | Yield |
|---|---|
| byproducts: In2O3; at 473°K and then at 673-773°K more; | 100% |

| Conditions | Yield |
|---|---|
| TiCl4 was added to deionized H2O, mixt. was stirred for 16 h at room temp., mixt was dialyzed in deionized H2O to pH 2.0; | 100% |
| In hydrogenchloride other Radiation; soln. of TiCl4 in cold 2 M HCl, hydrolysed, TiOCl2 formed, soln. transferred to Teflon container, treated in microwave oven (2.45 GHz) at temp. of 120°C for 120 min, or at 140°C for 60 or 120 min, or at160°C for 5 or 30 min; washed (deionized H2O, ethanol, repeatedly), dried in oven at 60°C for 16 h, detd. by XRD, mixt. of rutile and anatase obtained; | 99% |
| In hydrogenchloride other Radiation; soln. of TiCl4 in cold 2 M HCl, hydrolysed, TiOCl2 formed, soln. transferred to Teflon container, treated in microwave oven (2.45 GHz) at temp. of 160°C for 60 min; washed (deionized H2O, ethanol, repeatedly), dried in oven at 60°C for 16 h, detd. by XRD, mixt. of rutile and anatase obtained; | 98% |

| Conditions | Yield |
|---|---|
| With diethylenetriamine In water High Pressure; addn. of diethylenetriamine (DETA) to aq. soln. of Ce(NH4)2(NO3)6 with molar ratio of Ce to DETA =1:3; stirring for 10 min; transferring of gel to autoclave bomb; heating in hot air oven at 200°C for 24 h; crystn., filtration, drying in hot air oven at 100°C for 5 h; | 100% |
| With melamine In water High Pressure; addn. of melamine to aq. soln. of Ce(NH4)2(NO3)6 with molar ratio of Ce to melamine = 1:2; stirring for 10 min; transferring of gel to autoclavebomb; heating in hot air oven at 200°C for 24 h; crystn., filtration, drying in hot air oven at 100°C for 5 h; | 100% |
| In neat (no solvent) soln. hydrolyzed at 150 °C for 48 h, ppt. washed, dried, heated at 300, 500, 700 °C for 5 h in each step in air; powder XRD; |

| Conditions | Yield |
|---|---|
| With boric acid In water 1.5 equiv. of acid added to a soln. of Ti compd., aged for 18 h at 85°C; disperesed (water), centrifuged, washed (water); HR-TEM, ATR-FTIR, XRD; | 100% |

| Conditions | Yield |
|---|---|
| With cetyltrimethylammonium bromide (CTABr); NaOH In water Sonication; CTABr was dissolved in a NaOH soln. which was irradiated with ultrasoundfor 5 min at 25-35°C; addn. of TEOS (0.12 CTABr:0.35 NaOH:1.0 TE OS:922 H2O); sonication was further continued for 1 h; heating at 200°C for 48 h in an autoclave; identified by XRD studies; | 100% |
| With polyvinyl pyrrolidine; NaOH; cetyltrimethylammonium bromide (CTABr) In water High Pressure; polyvinyl pyrrolidine and NaOH were dissolved in H2O with stirring; cetyltrimethylammonium bromide and Si-contg. compd. were added with stirring; stirring for 24 h at 25°; mixt. was sealed in Teflon-lined autoclave, heated at 80°C for 48 h; identified by XRD studies; | 100% |
| With sodium dodecyl sulphonate (SDS); cetyltrimethylammonium chloride (CTACl) In water Sonication; SDS aq. soln. was sonicated for 5 min; CTACl was added; soln. was sonicated for 5 min; TEOS was added and soln. was sonicated for another 2 min at 25°C; heating at 180°C for 24 h in a Teflon-lined stainless autoclave; the compd. was recovered by filtration, washed with distd. water and dried under vac. at room temp.; then it was calcined in air to remove the templates; identified by XRD studies; | 100% |

| Conditions | Yield |
|---|---|
| In water; ethylene glycol 1:8:25:15 mixt., sealed, heated at 170°C for 4 ds; | 100% |


| Conditions | Yield |
|---|---|
| With CaCO3 In water High Pressure; mixt. of UO3, Sr(NO3)2, CaCO3 and H2O heated in Teflon-lined Parr vesselat 493 K for 30 d; crystals filtered, washed with boiling H2O; | 100% |

-
-
12354-84-6, 12354-85-7
bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]

-
-
7732-18-5
water

-
-
254734-81-1
[Ir(III)(η5-pentamethylcyclopentadienyl)(H2O)3](SO4)

| Conditions | Yield |
|---|---|
| at 20℃; for 3h; Inert atmosphere; | 100% |
| In water byproducts: AgCl; Ar-atmosphere; stoich. amts.; stirring (pH = 2.3, room temp., 12 h); AgCl removal (filtration), filtrate evapn., drying (vac.); elem. anal.; | 98% |
| at 20℃; for 12h; | 97% |

Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

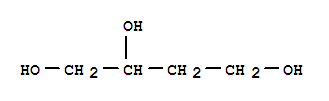




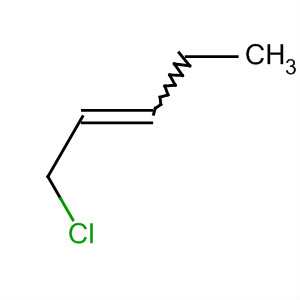

 C
C