 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route
-
-
7664-41-7
ammonia

-
-
80937-33-3
oxygen

-
A
-
7727-37-9
nitrogen

-
B
-
10102-43-9
nitrogen(II) oxide

-
C
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With oxygen In neat (no solvent) Fe-ZSM-5 catalyst prepared by ion exchange and heat-treated at 400, 425or 450 °C, 100 % NH3 conversion, 100 % N2 selectivity, 1000 ppm NH3 in 2 % O2-contg. He; | A 100% B 0% C 0% |
| With catalyst:Fe-mordenite In neat (no solvent) Fe-mordenite catalyst prepared by ion exchange and heat-treated at 425 °C, 92 % NH3 conversion, 99 % N2 selectivity, 1000 ppm NH3 in 2 %O2-contg. He; | A 92% B n/a C 0% |
| With catalyst:Fe-ZSM-5 In neat (no solvent) Fe-ZSM-5 catalyst prepared by ion exchange and heat-treated at 375 °C, 90 % NH3 conversion, 99 % N2 selectivity, 1000 ppm NH3 in 2 % O2-contg. He; | A 90% B n/a C 0% |


-
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With cobaltocene In dichloromethane at 20℃; for 0.0166667h; Kinetics; Time; Temperature; Concentration; | 100% |
-
-
7664-41-7
ammonia

-
A
-
7727-37-9
nitrogen

-
B
-
10102-43-9
nitrogen(II) oxide

-
C
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With oxygen In neat (no solvent) Fe-mordenite catalyst prepared by ion exchange and heat-treated at 450 °C, 99 % NH3 conversion, 100 % N2 selectivity, 1000 ppm NH3 in 2 % O2-contg. He; | A 99% B 0% C 0% |
| With oxygen; platinum high excess O2,500 °C; | A <1 B n/a C n/a |
| With oxygen; platinum at 300 °C; | A n/a B <9 C n/a |

-
-
114581-86-1
[CuI(tris(2-pyridylmethyl)amine)(CH3CN)]2+

-
-
10102-43-9
nitrogen(II) oxide

-
A
-
133578-96-8
[Cu(tris(2-pyridylmethyl)amine)(H2O)](2+)

-
B
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In water room temp., pH = 7.0; reaction followed by gas chromy.; | A 99% B n/a |


-
A
-
7727-37-9
nitrogen

-
B
-
10102-43-9
nitrogen(II) oxide

-
C
-
10102-44-0
Nitrogen dioxide

-
D
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| at 220-260°C, nearly 98% N2O, 2% N2; troces of NO and NO2 (0.001%); | A 2% B n/a C n/a D 98% |

-
-
1112-04-5
trifluoromethyldifluorophosphine

-
-
10102-43-9
nitrogen(II) oxide

-
A
-
19162-94-8
Trifluormethylphosphonsaeuredifluorid

-
B
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| 20°C (7 d); oxydation; | A 98% B n/a |


-
-
10102-43-9
nitrogen(II) oxide

-
A
-
188637-84-5
[copper(I)(tris(2-pyridylmethyl)amine)](1+)

-
C
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; methanol for 8h; Solvent; Inert atmosphere; Schlenk technique; | A 96% B 85% C 98% |


-
B
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With P(C6H5)3 In not given byproducts: OP(C6H5)3; react. of 1 equiv. N(C4H9)4(NO2) with (Cu2(NO)N6OC36H39)(2+) in presence of P(C6H5)3 (1 equiv.);; elem. anal.; | A 60% B 97% |
| In not given byproducts: O2; react. of 1 equiv. N(C4H9)4(NO2) with (Cu2(NO)N6OC36H39)(2+);; elem. anal.; | A 60% B 79% |

-
-
90065-16-0
Cu2(I)(C6H3(O)(CH2N(C2H4-2-pyridyl)2)2)(1+)

-
-
10102-43-9
nitrogen(II) oxide

-
B
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In dichloromethane exposure of (Cu2N6OC36H39)(1+) to NO(g) in CH2Cl2 at -80 °C, warming;; | A 55% B 96% |


-
-
7697-37-2
nitric acid

-
B
-
7732-18-5
water

-
C
-
10102-44-0
Nitrogen dioxide

-
D
-
2696-92-6
nitrosylchloride

-
E
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In nitric acid byproducts: Cl2; to Ru complex was added concd. HNO3; mixt. heated under reflux for 5 min; cooled to room temp.; ppt. filtered off; washed (water, alc., ether); dried (vac.); recrystd. (aq. HNO3); elem. anal.; | A 95% B n/a C n/a D n/a E n/a |

-
-
1194657-31-2
4-nitrosotetrahydro-2H-pyran-4-yl 2,2,2-trichloroacetate

-
A
-
29943-42-8
Tetrahydro-4H-pyran-4-one

-
B
-
10024-97-2
dinitrogen monoxide

-
C
-
76-03-9
trichloroacetic acid

| Conditions | Yield |
|---|---|
| With water In methanol; aq. phosphate buffer at 20℃; for 24h; pH=7.4; Kinetics; Reagent/catalyst; Sealed tube; | A n/a B 95% C n/a |


| Conditions | Yield |
|---|---|
| With oxygen In gaseous matrix byproducts: NO; 0.2 g of catalyst, 1000 ppm NH3, 2% O2, He as balance, GHSV=2.0E+5 h**-1, at 450°C; mass spect.; | A 94% B 0% |
| With oxygen In gaseous matrix byproducts: NO; 0.2 g of catalyst, 1000 ppm NH3, 2% O2, He as balance, GHSV=2.0E+5 h**-1, at 400°C; mass spect.; | A 93% B 0% |
| With oxygen In gaseous matrix byproducts: NO; 0.2 g of catalyst, 1000 ppm NH3, 2% O2, He as balance, GHSV=2.0E+5 h**-1, at 400°C; mass spect.; | A 92% B 0% |
-
-
1493-13-6
trifluorormethanesulfonic acid

-
B
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 1h; Schlenk technique; Inert atmosphere; | A n/a B 93% |


-
A
-
29943-42-8
Tetrahydro-4H-pyran-4-one

-
B
-
75-99-0
2,2-Dichloropropionic acid

-
C
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With water In methanol; aq. phosphate buffer at 20℃; for 24h; pH=7.4; Kinetics; Reagent/catalyst; Sealed tube; | A n/a B n/a C 92% |


-
C
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With cobaltocene In dichloromethane for 0.0833333h; Sealed tube; Inert atmosphere; | A n/a B n/a C 91% |

-
-
10102-43-9
nitrogen(II) oxide

-
A
-
7727-37-9
nitrogen

-
B
-
80937-33-3
oxygen

-
C
-
10102-44-0
Nitrogen dioxide

-
D
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| Irradiation (UV/VIS); irradn. with Al-glitter (wave length:1860,1930 and 1990 A); | A 90% B 90% C 10% D 10% |
| Irradiation (UV/VIS); irradn. with Al-glitter (wave length:1860,1930 and 1990 A); | A 90% B 90% C 10% D 10% |
| La0.7Ba0.3Mn0.8In0.1Cu0.1O3 In neat (no solvent) decomposition over catalyst at 1073 K; | A 69.4% B 42.3% C 27.1% D 0% |


| Conditions | Yield |
|---|---|
| manganese(II) oxide In neat (no solvent) catalytic oxidn. of NH3 (300 °C, O2-excess);; | 90% |
| byproducts: N2, NO; excess of O2; at 300 °C; catalyst MnO2/Fe2O3/(eventually) Bi2O3; yield depending on mole ratio O2:NH3 and flow rate;; | 90% |
| cobalt(II) oxide In neat (no solvent) oxidation at 300°C;; |
-
-
7664-41-7
ammonia

-
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With oxygen 300 °C,with air; | 90% |
| Electrolysis; NH3 soln. contg. CO2;anod contains Pt,Pd or Fe; | 14.6% |
| Electrolysis; |

-
A
-
7803-49-8
hydroxylamine

-
B
-
10024-97-2
dinitrogen monoxide

-
C
-
19467-31-3, 173728-04-6
trans-hyponitrous acid

| Conditions | Yield |
|---|---|
| In water | A n/a B 90% C n/a |

-
-
7782-77-6
cis-nitrous acid

-
A
-
7803-49-8
hydroxylamine

-
B
-
10024-97-2
dinitrogen monoxide

-
C
-
19467-31-3, 173728-04-6
trans-hyponitrous acid

| Conditions | Yield |
|---|---|
| In water | A n/a B 90% C n/a |


| Conditions | Yield |
|---|---|
| In neat (no solvent) decompn. at 225-230°C;; | A 2-6 B 3-5 C n/a D 90% |
| In neat (no solvent) decompn. at 225-230°C;; | A 2-6 B 3-5 C n/a D 90% |


| Conditions | Yield |
|---|---|
| With hypochloric acid In aq. phosphate buffer at 37℃; pH=7.4; | A n/a B 90% |
-
-
7758-09-0
potassium nitrite

-
A
-
7727-37-9
nitrogen

-
B
-
10024-97-2
dinitrogen monoxide

-
C
-
19467-31-3, 173728-04-6
trans-hyponitrous acid

| Conditions | Yield |
|---|---|
| In not given at room temp. or upon boiling from equimolar amounts in 3% solns.; no formation of H2N2O2 even in concd. solns. at low temps.; mole ratio of N2O:N2 depends on the mole ratio nitrite:hydrazonium salt;; | A 11% B 89% C 0% |


-
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With cobaltocene In dichloromethane for 0.0833333h; Inert atmosphere; | 89% |
| With silver; N,N,N,N-tetraethylammonium tetrafluoroborate In dichloromethane Inert atmosphere; Electrochemical reaction; | 89% |

| Conditions | Yield |
|---|---|
| With Hg2(NO3)2 In not given byproducts: Hg; addn. of 0.04 M NH2OH to 0.005 M Hg2(NO3)2 soln. buffered with acetate at 20°C, autocatalysis of reaction by Hg;; | A 13% B 87% |
| With Hg2(NO3)2; silver In not given byproducts: Hg; addn. of 0.04 M NH2OH to 0.005 M Hg2(NO3)2 soln. buffered with acetate at 20°C, autocatalysis of reaction by Hg, further catalysis by addn. of collodial Ag soln.;; | A 63% B 34% |
| With oxygen In not given byproducts: water; Electrolysis; N2O formed on cathode, N2 formed on anode; |

-
-
10102-43-9
nitrogen(II) oxide

-
B
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In methanol Inert atmosphere; Schlenk technique; | A 85% B n/a |

-
-
463-51-4
Ketene

-
-
10102-43-9
nitrogen(II) oxide

-
A
-
51060-05-0
fulminic acid

-
B
-
74-90-8
hydrogen cyanide

-
C
-
201230-82-2
carbon monoxide

-
D
-
7732-18-5
water

-
E
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| byproducts: H2CO; Irradiation (UV/VIS); photolysis of CH2CO/NO/Ar mixt. with 200 W high pressure Hg lamp (photolysis of CH2CO generates CH2 radicals) in Duran glass tube (20 cm long, 4cm i.d.) at room temp. and ambient pressure; | A 84% B 15% C n/a D n/a E n/a |


-
-
10024-97-2
dinitrogen monoxide

-
-
205107-24-0
(η(5)-C5Me5)2Zr(OCPh=CSiMe3)

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: N2; absence of air and moisture; 500 Torr N2O, room temp., 24 h; removal of gases; elem. anal.; | 100% |
| In toluene byproducts: N2; absence of air and moisture; N2O-atmosphere, warming from -78°C to room temp., stirring for 30 min; rsolvent removal, recrystn. (hexane/O(SiMe3)2); | 65% |
| In benzene absence of air and moisture; |

-
-
10024-97-2
dinitrogen monoxide

-
-
205107-25-1
(η(5)-C5Me5)2Zr(OC(c-C5H9)=CSiMe3)

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: N2; absence of air and moisture; 500 Torr N2O, room temp., 24 h; removal of gases; elem. anal.; | 100% |
| In benzene absence of air and moisture; |

-
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In benzene-d6 at 80℃; under 760.051 Torr; for 18h; Inert atmosphere; Schlenk technique; | 100% |
-
-
34557-54-5
methane

-
-
10024-97-2
dinitrogen monoxide

-
A
-
67-56-1
methanol

-
B
-
50-00-0
formaldehyd

-
C
-
124-38-9
carbon dioxide

-
D
-
201230-82-2
carbon monoxide

-
E
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; Oxidation of CH4 by N2O in presence of catalyst (773 K): deposited Cu(2+) on carbon;; | A 0.2% B 0.3% C 99.5% D 0% E n/a |
| In neat (no solvent) Kinetics; byproducts: C2H5OH (small quantity); oxidation of CH4 by N2O in presence of catalyst (773 K): deposited Ti(4+) on carbon;; | A 13.8% B 0% C 86.2% D 0% E n/a |
| In neat (no solvent) Kinetics; Oxidation of CH4 by N2O in presence of catalyst (773 K): deposited Co(2+) on carbon;; | A 0% B 0% C 75% D 25% E n/a |


| Conditions | Yield |
|---|---|
| byproducts: KOH, NH3; 270-280 °C in rotating furnace; the white layer of KOH and KN3 dissolved in water, the soln. evaporated, KN3 crystd.; | 99% |
| byproducts: KOH, NH3; 270-280 °C in rotating furnace; the white layer of KOH and KN3 dissolved in water, the soln. evaporated, KN3 crystd.; | 99% |
| In ammonia byproducts: KOH, NH3; -70°C; | |
| In ammonia byproducts: KOH, NH3; NH3 (liquid); -70°C; |
-
-
7783-54-2
nitrogen trifluoride

-
-
7783-70-2
antimony pentafluoride

-
-
10024-97-2
dinitrogen monoxide

-
-
12528-55-1
NF2O(1+)*Sb2F11(1-) =(NF2O)(Sb2F11)

| Conditions | Yield |
|---|---|
| byproducts: N2; 150°C; | 99% |
| 100°C; | 0% |

-
-
1039748-93-0
N[2-P(CHMe3)2-4-methylphenyl]2V(CH2tBu)2

-
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In benzene V-complex reacted with N2O in benzene at 90°C; | 99% |

-
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| at 25℃; for 216h; | 99% |
-
-
328396-60-7
bis[2,6-bis(1-naphthyl)phenyl]germylene

-
-
10024-97-2
dinitrogen monoxide

-
-
327179-04-4
bis[2,6-bis(1-naphthyl)phenyl]germanone

| Conditions | Yield |
|---|---|
| In toluene left standing under N2 for 2 d; solvent evpd. in vacuo; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran soln. of B compd. stirred at room temp. under N2O atm. until gas evolution stopped (ca. 5 min); evapd., elem. anal.; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran soln. of B compd. stirred at room temp. under N2O atm. until gas evolution stopped (ca. 5 min); evapd., elem. anal.; | 98% |

-
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 0.5h; Inert atmosphere; Schlenk technique; | 98% |

| Conditions | Yield |
|---|---|
| In benzene-d6 at 20℃; under 760.051 Torr; for 24h; | 98% |
-
-
1033858-47-7
ethylenebis(1,3-di-tert-butylcyclopentadienyl)titanium

-
-
10024-97-2
dinitrogen monoxide

-
A
-
827030-28-4
μ-oxotetrakis(1,3-di-tert-butylcyclopentadienyl)dititanium

-
B
-
1033858-56-8
di-μ-oxotetrakis(1,3-di-tert-butylcyclopentadienyl)dititanium

| Conditions | Yield |
|---|---|
| In pentane Ti complex dissolved in pentane; exposed to N2O (1 atm); solvent removed under vac.; extd. into hot toluene; cooled to room temp.and then to -20°C; crystals isolated; elem. anal.; | A 0% B 97% |

-
-
382150-90-5
(HC(CMeNC-2,6-i-Pr2C6H3)2)-germanium(II) hydride

-
-
10024-97-2
dinitrogen monoxide

-
-
692776-46-8
(HC(CMeNC-2,6-i-Pr2C6H3)2)-germanium(II) hydroxide

| Conditions | Yield |
|---|---|
| In toluene under N2 or Ar; dry N2O bubbled into soln. of Ge compd. in toluene at room temp. for 30 min; volatiles removed in vac.; treated with n-hexane; filtered; dried in vac.; | 95% |

| Conditions | Yield |
|---|---|
| In toluene at 20℃; under 760.051 Torr; for 24h; Cooling with liquid nitrogen; Inert atmosphere; | 94% |

-
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In acetonitrile Inert atmosphere; | 94% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View




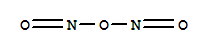


 O
O