 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route

| Conditions | Yield |
|---|---|
| With H nitrate, in dild. soln., is completely reacting with Pd, satd. with H, at 16 °C in 24 hours to Ag and HNO3;; | A 100% B 100% |
| With H |

-
-
3449-05-6
2-(salicylideneamino)thiophenol

-
B
-
240814-62-4
(OC6H4CHNC6H4S)Ce(NO3)2(H2O)

-
C
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| With ammonium hydroxide In ethanol; water addn. of hot soln. of Schiff base in EtOH to soln. of Ce salt in EtOH (ratio 1 : 1), addn. of H2O and EtOH (to 85% EtOH), pH adjusting to 5.0 - 6.0 (NH4OH), refluxing (2 h), concn. (vac.), crystn. on cooling (over night); filtration, washing (EtOH), recrystn. (hot DMF/EtOH), washing (EtOH, Et2O), drying (vac.); elem. anal.; | A n/a B 90% C n/a |


-
-
1333-84-2, 1344-28-1
aluminum oxide

-
-
7631-99-4
sodium nitrate

-
A
-
1302-42-7
sodium aluminate

-
B
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| heating; it attacks vessel of iron or platinum and glass or china; | A n/a B 89% |

-
-
119-65-3
isoquinoline

-
-
141-82-2
malonic acid

-
-
7722-84-1
dihydrogen peroxide

-
A
-
187455-26-1
UO(O2)(CH2(COO)2)(C9H7N)2

-
B
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| In ethanol; water addn. of org. acid in EtOH to aq. soln. of metal salt, cooling, addn. ofsoln. of isoquinoline in EtOH, addn. of aq. H2O2 (30 %), stirring (30 m in; pptn.); filtration, washing (water, EtOH), purification by TLC, drying (vac., over silica gel); elem. anal.; | A 79% B n/a |


| Conditions | Yield |
|---|---|
| In ethanol; water addn. of org. acid in EtOH to aq. soln. of metal salt, cooling, addn. ofsoln. of quinoline in EtOH, addn. of aq. H2O2 (30 %), stirring (30 min; pptn.); filtration, washing (water, EtOH), purification by TLC, drying (vac., over silica gel); elem. anal.; | A 76% B n/a |


| Conditions | Yield |
|---|---|
| In ethanol; water addn. of org. acid in EtOH to aq. soln. of metal salt, cooling, addn. ofsoln. of isoquinoline in EtOH, addn. of aq. H2O2 (30 %), stirring (30 m in; pptn.); filtration, washing (water, EtOH), purification by TLC, drying (vac., over silica gel); elem. anal.; | A 72% B n/a |

-
-
91-22-5
quinoline

-
-
141-82-2
malonic acid

-
-
7722-84-1
dihydrogen peroxide

-
A
-
187455-21-6
Zr(O2)(CH2(COO)2)(C9H7N)2

-
B
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| In ethanol; water addn. of org. acid in EtOH to aq. soln. of metal salt, cooling, addn. ofsoln. of quinoline in EtOH, addn. of aq. H2O2 (30 %), stirring (30 min; pptn.); filtration, washing (water, EtOH), purification by TLC, drying (vac., over silica gel); elem. anal.; | A 72% B n/a |

-
-
91-22-5
quinoline

-
-
141-82-2
malonic acid

-
-
7722-84-1
dihydrogen peroxide

-
A
-
187455-25-0
UO(O2)(CH2(COO)2)(C9H7N)2

-
B
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| In ethanol; water addn. of org. acid in EtOH to aq. soln. of metal salt, cooling, addn. ofsoln. of quinoline in EtOH, addn. of aq. H2O2 (30 %), stirring (30 min; pptn.); filtration, washing (water, EtOH), purification by TLC, drying (vac., over silica gel); elem. anal.; | A 70% B n/a |

-
-
119-65-3
isoquinoline

-
-
141-82-2
malonic acid

-
-
7722-84-1
dihydrogen peroxide

-
A
-
187455-22-7
Zr(O2)(CH2(COO)2)(C9H7N)2

-
B
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| In ethanol; water addn. of org. acid in EtOH to aq. soln. of metal salt, cooling, addn. ofsoln. of isoquinoline in EtOH, addn. of aq. H2O2 (30 %), stirring (30 m in; pptn.); filtration, washing (water, EtOH), purification by TLC, drying (vac., over silica gel); elem. anal.; | A 70% B n/a |


| Conditions | Yield |
|---|---|
| In ethanol; water addn. of org. acid in EtOH to aq. soln. of metal salt, cooling, addn. ofsoln. of isoquinoline in EtOH, addn. of aq. H2O2 (30 %), stirring (30 m in; pptn.); filtration, washing (water, EtOH), purification by TLC, drying (vac., over silica gel); elem. anal.; | A 68% B n/a |


| Conditions | Yield |
|---|---|
| In ethanol; water addn. of org. acid in EtOH to aq. soln. of metal salt, cooling, addn. ofsoln. of quinoline in EtOH, addn. of aq. H2O2 (30 %), stirring (30 min; pptn.); filtration, washing (water, EtOH), purification by TLC, drying (vac., over silica gel); elem. anal.; | A 67% B n/a |

-
-
326-91-0
1,1,1-trifluoro-4-(2-thienyl)butane-2,4-dione

-
-
23978-55-4
1,4,10,13-tetraoxa-7,16-diazacyclooctadecane

-
B
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| In water; toluene dropwise addn. of soln. of Eu-salt in H2O to soln. of macrocycle and diketone (stirring), sepd. of org. phase after 30 min, pptn. on addn. of cyclohexane to org. phase; collection, recrystn. (toluene/cyclohexane 1/1), drying (over P2O5, vac., 5 h); elem. anal.; | A 62% B n/a |


| Conditions | Yield |
|---|---|
| With oxygen NH3-air mixture,at gently red heat; | 60% |
| With oxygen; Sr(2+)*PbO3(2-)*Sr(2+)*MnO4(2-)=SrPbO3*SrMnO4 byproducts: nitrogen oxides; above 500 °C,NH3-air mixture,overall yield:from 90%; | |
| With oxygen; Ba(2+)*PbO3(2-)*Ba(2+)*MnO4(2-)=BaPbO3*BaMnO4 byproducts: nitrogen oxides; above 500 °C,NH3-air mixture,overall yield:from 90%; |

| Conditions | Yield |
|---|---|
| at 4 - 20℃; pH=2.5; | A 56% B n/a C n/a |

-
-
7647-01-0
hydrogenchloride

-
-
10102-03-1
dinitrogen pentoxide

-
A
-
7697-37-2
nitric acid

-
B
-
7782-50-5
chlorine

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 213 K; purified from N2O5 and nitric acid by distillation at 213 K; | A n/a B 2% C n/a |


| Conditions | Yield |
|---|---|
| at 0℃; Rate constant; in festen und fluessigen H2SO4-H2O-Gemischen <84.5-86.4%ig>.Hydrolysis; |
-
-
53360-90-0
nitro-tetrahydropyrimidin-2-ylidene-amine

-
-
7664-93-9
sulfuric acid

-
-
64-19-7
acetic acid

-
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| at 25℃; Rate constant; |


| Conditions | Yield |
|---|---|
| at 25℃; Rate constant; |

-
-
7732-18-5
water

-
-
100130-44-7
(4-methyl-4,5-dihydro-1(3)H-imidazol-2-yl)-nitro-amine

-
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| at 25℃; Rate constant; Hydrolysis; |


| Conditions | Yield |
|---|---|
| at 25℃; Rate constant; Hydrolysis; |

-
-
7732-18-5
water

-
-
100130-44-7
(4-methyl-4,5-dihydro-1(3)H-imidazol-2-yl)-nitro-amine

-
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| at 0℃; Rate constant; in festen und fluessigen Gemischen <84.5%ig>.Hydrolysis; |


| Conditions | Yield |
|---|---|
| at 25℃; Rate constant; |


| Conditions | Yield |
|---|---|
| With sodium nitrite |

-
A
-
151201-39-7
methyl-imidazolidinetrione-5-oxime

-
B
-
7697-37-2
nitric acid

| Conditions | Yield |
|---|---|
| With sodium nitrite |
-
-
7732-18-5
water

-
-
88-89-1
2,4,6-Trinitrophenol

-
A
-
74-90-8
hydrogen cyanide

-
B
-
124-38-9
carbon dioxide

-
C
-
7697-37-2
nitric acid


| Conditions | Yield |
|---|---|
| at 70 - 80℃; es erfolgt Oxidation; |

-
-
871932-89-7
5-chloro-4-[cyclohexyl-(2,2,2-trifluoroacetyl)amino]-2-methoxybenzoic acid methyl ester

-
-
7697-37-2
nitric acid

-
-
871932-90-0
5-chloro-4-[cyclohexyl-(2,2,2-trifluoroacetyl)amino]-2-methoxy-3-nitrobenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: 5-chloro-4-[cyclohexyl-(2,2,2-trifluoroacetyl)amino]-2-methoxybenzoic acid methyl ester; nitric acid at -40 - -20℃; for 2h; Stage #2: With sodium hydroxide In water | 100% |

| Conditions | Yield |
|---|---|
| at 80℃; | 100% |
| In nitric acid aq. HNO3; dissolving metal oxide in concd. HNO3, heating; evapn. on water bath, dissolving in water; | |
| In nitric acid aq. HNO3; by treating the metal oxide with dil. HNO3; the soln. was evapd. on a steam bath; the residue was dissolved in water, conced. to a viscous mass, cooled and kept in a desiccator after breaking up any lumps; |


| Conditions | Yield |
|---|---|
| at 80℃; | 100% |
| In nitric acid aq. HNO3; Eu2O3 treated with concd. HNO3; excess HNO3 evapd.; | |
| In nitric acid aq. HNO3; dissolving metal oxide in concd. HNO3, heating; evapn. on water bath, dissolving in water; |


| Conditions | Yield |
|---|---|
| at 80℃; | 100% |
| In water react. metal oxide with 6N HNO3; evapn. at 100°C; | |
| In nitric acid aq. HNO3; by treating the metal oxide with dil. HNO3; the soln. was evapd. on a steam bath; the residue was dissolved in water, conced. to a viscous mass, cooled and kept in a desiccator after breaking up any lumps; | |
| In nitric acid aq. HNO3; dissolving of Y2O3 in excess amt. of aq. nitric acid; |


| Conditions | Yield |
|---|---|
| In water Electrolysis; Cu-cathode, in presence of H2SO4;; | 100% |
| With aluminium In water at elevated pressure;; | 0% |
| With aluminium In water only small amounts of NH3 in dild. HNO3 (5%-20%) at atmospheric pressure;; |

| Conditions | Yield |
|---|---|
| With sulfur trioxide pyridine complex at 25 - 100℃; for 180h; Sealed tube; | 100% |
| In nitric acid in presence of air; | |
| In nitric acid byproducts: N-oxide; by heating; in presence of air; |

| Conditions | Yield |
|---|---|
| at 80℃; | 100% |


| Conditions | Yield |
|---|---|
| In nitric acid aq. HNO3; PbWO4 dissolved in aq. HNO3 (10 wt %) at 75°C; isothermal holdingtime was 2 h; ppt. filtered off; washed (aq. HNO3); calcined at 700°C (WO3 was obtained); to filtrate contg. Pb(NO3)2 added aq. NH4OH with stirring; final pH was 8.9; ppt. diltered off; dried; calcined at 800°C; | A 99.9% B 93.6% |


| Conditions | Yield |
|---|---|
| In water byproducts: NO2; Sb powder was covered with concd. nitric acid, heated under an open hood and over a bunsen burner flame, solid was filtered, washed with water,left to dry, heated in open beaker over a bunsen burner flame; | 99.729% |

| Conditions | Yield |
|---|---|
| In water byproducts: oxalic acid; between 15 and 65°C;; pure NaNO3;; | 99% |

| Conditions | Yield |
|---|---|
| In nitric acid byproducts: H2SO4, NO, CO; stirring for 0.5 h at 25°C; condensing silane in a trap cooled with solid CO2, treating aq. layer with NaOH, evapn. to dryness, IR; | A n/a B 99% |


| Conditions | Yield |
|---|---|
| In further solvent(s) the mixt. of NaBiO3 and Am(3+) in 0.1 M nitric acid was agitated at roomtemp.; detected by UV spectra and γ-ray spectroscopy; | 99% |


| Conditions | Yield |
|---|---|
| In further solvent(s) the mixt. of NaBiO3 and Am(3+) in 0.1 M nitric acid was heated at 80°C; detected by UV spectra and γ-ray spectroscopy; | 99% |


| Conditions | Yield |
|---|---|
| In water High Pressure; Bi(NO3)3, HIO3, HNO3 were heated in autoclave to 200°C, held for 1 week, cooled slowly, 6°C/h, to room temp.; filtered, washed with distilled water; | 99% |


| Conditions | Yield |
|---|---|
| With water; ammonium carbonate In nitric acid solution of Bi in HNO3 (36 Be); vigorous reaction, repeated addition of H2O;; filtration through an asbestos filter and evaporation of the filtrate; crystallisation and washing with H2O (containing a small amount of HNO3); drying at 25°C; addition of (NH4)2CO3 to the end liquors and formation of Bi nitrate with HNO3;; | 98% |
| With H2O; (NH4)2CO3 In nitric acid solution of Bi in HNO3 (36 Be); vigorous reaction, repeated addition of H2O;; filtration through an asbestos filter and evaporation of the filtrate; crystallisation and washing with H2O (containing a small amount of HNO3); drying at 25°C; addition of (NH4)2CO3 to the end liquors and formation of Bi nitrate with HNO3;; | 98% |
| With water; oxygen In nitric acid byproducts: N oxides; addition of warm Bi grains under reflux to HNO3 and O2; absorption of the formed N oxides after oxidation in H2O and returning back to the reaction mixture;; |
-
-
126-33-0
sulfolane

-
-
314-41-0
1,2,3,5-tetrafluoro-4-nitrobenzene

-
-
7697-37-2
nitric acid

-
-
20002-14-6
2,4,5,6-Tetrafluoro-1,3-dinitrobenzene

| Conditions | Yield |
|---|---|
| With boron trifluoride at 65-70°C 7 d; | 98% |
| With BF3 at 65-70°C 7 d; | 98% |


| Conditions | Yield |
|---|---|
| Stage #1: barium(II) nitrate; 4C12H28N(1+)*4H2O*V4O12(4-); dimethyl sulfoxide at 70℃; for 4h; Stage #2: nitric acid at 20℃; for 2h; | 98% |


| Conditions | Yield |
|---|---|
| In benzene-d6 at 20℃; for 0.0833333h; Glovebox; | 98% |

| Conditions | Yield |
|---|---|
| In water HNO3 added to Nd/H(+)=2, Tl salt added dropwise at 25°C with vigorous stirring, kept for 24 h; ppt. filtered, washed (cold water), dried air; elem. anal.; | 97.78% |

Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View




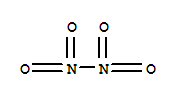











 C,
C, O
O